WINREVAIR (SOTATERCEPT-CSRK) kit
1 INDICATIONS AND USAGE
WINREVAIR™ is indicated for the treatment of adults with pulmonary arterial hypertension (PAH, Group 1 pulmonary hypertension) to improve exercise capacity and World Health Organization (WHO) functional class (FC), and reduce the risk of clinical worsening events including hospitalization for PAH, lung transplantation and death [see Clinical Studies (14.1)].
WINREVAIR is an activin signaling inhibitor indicated for the treatment of adults with pulmonary arterial hypertension (PAH, WHO Group 1 pulmonary hypertension) to improve exercise capacity and WHO functional class (FC), and reduce the risk of clinical worsening events, including hospitalization for PAH, lung transplantation and death. (1)
2 DOSAGE AND ADMINISTRATION
- The recommended starting dose is 0.3 mg/kg by subcutaneous injection. (2.1)
- The recommended target dose is 0.7 mg/kg every 3 weeks by subcutaneous injection. (2.2)
- Dosage modifications due to increased hemoglobin (Hgb) and decreased platelets may be necessary. Check Hgb and platelets before each dose for the first 5 doses, or longer if values are unstable, and monitor periodically thereafter. (2.3)
- See full prescribing information for preparation and administration instructions. (2.4)
2.1 Recommended Starting Dosage
WINREVAIR is administered once every 3 weeks by subcutaneous injection according to patient body weight. The starting dose of WINREVAIR is 0.3 mg/kg.
Obtain hemoglobin (Hgb) and platelet count prior to the first dose of WINREVAIR. Do not initiate treatment if platelet count is <50,000/mm3 (<50 x 109/L) [see Dosage and Administration (2.3)].
Injection volume for starting dose is calculated based on patient weight as follows:
Injection volume should be rounded to the nearest 0.1 mL.
For example: (70 kg x 0.3 mg/kg) ÷ 50 mg/mL = 0.42 mL, rounds to 0.4 mL
See Table 1 for selecting the appropriate kit based on calculated injection volume for starting dose.
2.2 Recommended Target Dosage
After verifying acceptable Hgb and platelet count, increase to the target dose of 0.7 mg/kg. Continue treatment at 0.7 mg/kg every 3 weeks unless dosage adjustments are required [see Dosage and Administration (2.3)].
Injection volume for target dose is calculated based on patient weight as follows:
Injection volume should be rounded to the nearest 0.1 mL.
For example: (70 kg x 0.7 mg/kg) ÷ 50 mg/mL = 0.98 mL, rounds to 1 mL
See Table 2 for selecting the appropriate kit based on calculated injection volume for target dose.
Missed Dose, Overdose, and Underdose
If a dose of WINREVAIR is missed, administer as soon as possible. If the missed dose of WINREVAIR is not administered within 3 days of the scheduled date, adjust the schedule to maintain 3-week dosing intervals. In case of an overdose, monitor for erythrocytosis [see Overdosage (10)].
2.3 Dosage Modifications Due to Hemoglobin Increase or Platelet Count Decrease
Check Hgb and platelet count before each dose for the first 5 doses, or longer if values are unstable. Thereafter, monitor Hgb and platelet count periodically [see Warnings and Precautions (5.1, 5.2)].
Delay treatment for at least 3 weeks if any of the following occur:
- Hgb increases >2.0 g/dL from the previous dose and is above ULN.
- Hgb increases >4.0 g/dL from baseline.
- Hgb increases >2.0 g/dL above ULN.
- Platelet count decreases to <50,000/mm3 (<50 x 109/L).
Recheck Hgb and platelet count before reinitiating treatment. For treatment delays lasting >9 weeks, restart treatment at 0.3 mg/kg, and escalate to 0.7 mg/kg after verifying acceptable Hgb and platelet count.
2.4 Preparation and Administration
Administration is subject to monitoring of hemoglobin and platelet count [see Dosage and Administration (2.3), Warnings and Precautions (5.1, 5.2)].
WINREVAIR is intended for use under the guidance of a healthcare professional. Patients and caregivers may administer WINREVAIR when considered appropriate and when they receive training and follow-up from the healthcare provider (HCP) on how to reconstitute, prepare, measure, and inject WINREVAIR [see Patient Counseling Information (17)].
Confirm at subsequent visits that the patient and/or caregiver can correctly prepare and administer WINREVAIR, particularly if the dose changes or the patient requires a different kit [see Warnings and Precautions (5.1)].
Refer to the Instructions for Use (IFU) for detailed instructions on the proper preparation and administration of WINREVAIR.
Selecting the Appropriate Product Kit
If a patient’s body weight requires the use of two 45 mg vials or two 60 mg vials of lyophilized product, use a 2-vial kit instead of two individual 1-vial kits. A 2-vial kit includes instructions to combine the contents of two vials, which aids in measuring the proper dosage and eliminates the need for multiple injections [see How Supplied/Storage and Handling (16.1)].
Reconstitution Instructions
- Remove the injection kit from the refrigerator and wait 15 minutes to allow the prefilled syringe(s) and drug product to come to room temperature prior to preparation.
- Attach the vial adapter to the vial.
- Visually inspect the pre-filled syringe for any damage or leaks and the Sterile Water for Injection inside to ensure there are no visible particles.
- Snap off the cap of the pre-filled syringe and attach the syringe to the vial adapter.
- Inject all of the Sterile Water for Injection from the attached syringe into the vial containing the lyophilized powder. This will provide a final concentration of 50 mg/mL.
- Gently swirl the vial to reconstitute the drug product. DO NOT shake or vigorously agitate.
- Allow the vial to stand for up to 3 minutes to allow bubbles to disappear.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- When properly mixed, WINREVAIR should be clear to opalescent and colorless to slightly brownish-yellow and does not have clumps or powder.
- If prescribed a 2-vial presentation, repeat the steps within this section to prepare the second vial.
- Use the reconstituted solution as soon as possible, but no later than 4 hours after reconstitution. Discard unused reconstituted solution.
Syringe Preparation
- Turn the syringe and vial upside-down and withdraw the appropriate volume for injection, based on the patient’s weight.
- If the dose amount requires the use of two vials, withdraw the entire contents of the first vial and slowly transfer full contents into the second vial.
- Turn the syringe and vial upside-down and withdraw the required amount of drug product.
- If necessary, remove excess drug product.
- If necessary, remove excess air from the syringe.
Administration Instructions
WINREVAIR is for subcutaneous injection.
- Select the injection site on the abdomen (at least 2 inches away from navel), upper thigh, or upper arm, and swab with an alcohol wipe. Select a new site for each injection that is not scarred, tender, or bruised.
- For administration by the patient or caregiver, use only the abdomen and upper thigh (see IFU).
- Perform subcutaneous injection.
3 DOSAGE FORMS AND STRENGTHS
- For injection: 45 mg white to off-white lyophilized cake or powder appearance in a single-dose vial.
- For injection: 60 mg white to off-white lyophilized cake or powder appearance in a single-dose vial.
5 WARNINGS AND PRECAUTIONS
- Erythrocytosis: If severe, may increase the risk of thromboembolic events and hyperviscosity syndrome. Monitor Hgb before each dose for the first 5 doses, or longer if values are unstable, and periodically thereafter to determine if dose adjustments are required. (5.1)
- Severe Thrombocytopenia: May increase the risk of bleeding. Monitor platelets before each dose for the first 5 doses, or longer if values are unstable, and periodically thereafter to determine if dose adjustments are required. (5.2)
- Serious Bleeding: Serious bleeding events were reported and were more likely with concomitant prostacyclin and/or antithrombotic agents, or with low platelet counts. Do not administer WINREVAIR if the patient is experiencing serious bleeding. (5.3)
- Embryo-Fetal Toxicity: May cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.4, 8.1, 8.3)
- Impaired Fertility: May impair female and male fertility. (5.5, 8.3, 13.1)
5.1 Erythrocytosis
WINREVAIR may increase hemoglobin. Severe erythrocytosis may increase the risk of thromboembolic events or hyperviscosity syndrome. In clinical studies, moderate elevations in Hgb (>2 g/dL above ULN) occurred in 15% of patients taking WINREVAIR while no elevations ≥4 g/dL above ULN were observed. Monitor Hgb before each dose for the first 5 doses, or longer if values are unstable, and periodically thereafter, to determine if dose adjustments are required [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
5.2 Severe Thrombocytopenia
WINREVAIR may decrease platelet count. Severe thrombocytopenia may increase the risk of bleeding. In clinical studies, severe thrombocytopenia (platelet count <50,000/mm3 [<50 x 109/L]) occurred in 3% to 6% of patients taking WINREVAIR. Thrombocytopenia occurred more frequently in patients also receiving prostacyclin infusion.
Do not initiate treatment if platelet count is <50,000/mm3 [see Dosage and Administration (2.3)].
Monitor platelets before each dose for the first 5 doses, or longer if values are unstable, and periodically thereafter to determine whether dose adjustments are required. [see Dosage and Administration (2.3), Adverse Reactions (6.1)].
5.3 Serious Bleeding
In clinical studies, serious bleeding (e.g., gastrointestinal, intracranial hemorrhage) was reported in 4% vs 1% (STELLAR) and 7% vs 5% (ZENITH) of patients taking WINREVAIR vs placebo, respectively [see Clinical Studies (14.1)]. Patients with serious bleeding were more likely to be on prostacyclin background therapy and/or antithrombotic agents, or have low platelet counts. Advise patients about signs and symptoms of blood loss. Evaluate and treat bleeding accordingly. Do not administer WINREVAIR if the patient is experiencing serious bleeding [see Warnings and Precautions (5.2), Adverse Reactions (6.1)].
5.4 Embryo-Fetal Toxicity
Based on findings in animal reproduction studies, WINREVAIR may cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of WINREVAIR to pregnant rats and rabbits during organogenesis resulted in adverse developmental outcomes, including increased embryo-fetal mortality, alterations to growth, and structural variations at exposures 4-fold and 0.6-fold (based on area under the curve [AUC]) those occurring at the maximum recommended human dose (MRHD), respectively. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use an effective method of contraception during treatment with WINREVAIR and for at least 4 months after the final dose [see Use in Specific Populations (8.1, 8.3)].
5.5 Impaired Fertility
Based on findings in animals, WINREVAIR may impair female and male fertility. Advise patients on the potential effects on fertility [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)].
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Erythrocytosis [see Warnings and Precautions (5.1)]
- Severe Thrombocytopenia [see Warnings and Precautions (5.2)]
- Serious Bleeding [see Warnings and Precautions (5.3)]
- Embryo-Fetal Toxicity [see Warnings and Precautions (5.4)]
- Impaired Fertility [see Warnings and Precautions (5.5)]
The most common (≥10% in patients receiving WINREVAIR and 5% more than placebo) adverse reactions were infections, epistaxis, telangiectasia, diarrhea, headache, rash, increased hemoglobin, dizziness, erythema, and gingival bleeding. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
STELLAR
The following data reflect exposure to WINREVAIR in the STELLAR trial. Adult PAH patients with WHO FC II or III (n=323) were randomized in a 1:1 ratio to receive WINREVAIR or placebo in combination with background standard of care therapies. Patients received a starting dose of 0.3 mg/kg via SC injection and the dose was increased to the target dose of 0.7 mg/kg administered once every 3 weeks for 24 weeks. After completing the primary 24-week treatment phase, patients continued into a long-term double-blind (LTDB) treatment period, maintaining their randomized treatment assignment, until all patients completed the primary treatment period. The median duration of treatment was 273 days in the placebo group and 313 days in the WINREVAIR group [see Clinical Studies (14.1)].
The most common adverse reactions occurring in STELLAR (≥10% for WINREVAIR and at least 5% more than placebo) are shown in Table 3.
Increased Hemoglobin
Increases in Hgb were managed by dose delays (10%), dose reductions (6%), or both (5%). Shifts in Hgb from normal to above normal levels occurred in 87 (53%) patients receiving WINREVAIR and in 23 (14%) patients receiving placebo.
Thrombocytopenia
Decreases in platelets were managed by dose delays (2%), dose reductions (2%), or both (2%). Shifts in platelet count from normal to below normal occurred in 40 (25%) patients receiving WINREVAIR and in 26 (16%) patients receiving placebo.
Telangiectasia
In patients exposed to WINREVAIR who experienced telangiectasia, the median time to onset was 36.1 weeks.
Increased Blood Pressure
In patients taking WINREVAIR, mean systolic/diastolic blood pressure increased from baseline by 2.2/4.9 mmHg at 24 weeks. In patients taking placebo, the change from baseline in mean blood pressure was -1.6/-0.6 mmHg.
Treatment Discontinuation
The incidences of treatment discontinuations due to an adverse reaction were 4% in the WINREVAIR group and 7% in the placebo group. No specific adverse reactions causing treatment discontinuations occurred with a frequency greater than 1% and more often in the WINREVAIR group.
ZENITH
The following data reflect exposure to WINREVAIR in the ZENITH trial. Adult PAH patients with WHO FC III or IV at high risk of mortality (n=172) were randomized in a 1:1 ratio to treatment with WINREVAIR or placebo in combination with background standard of care therapies. Patients who did not experience a primary endpoint event remained in the Double-Blind Placebo-Controlled (DBPC) Treatment Period, while patients who experienced an event of PAH worsening-related hospitalization of ≥24 hours were eligible to enroll into the open-label, long-term follow-up (LTFU) study SOTERIA. The median duration of exposure was longer in the WINREVAIR group (435 days) than in the placebo group (268 days) [see Clinical Studies (14.1)].
The overall incidences of adverse reactions in both arms were higher in the ZENITH trial than in the STELLAR trial. Severe reduction in platelet count <50,000/mm3 (<50.0 x 109/L) occurred in 6% of patients taking WINREVAIR. In the WINREVAIR group, 1 patient (1%) discontinued study intervention due to an adverse event, compared with 4 patients (5%) in the placebo group.
Uncontrolled Long-term Safety Data
PULSAR was a multicenter, randomized, double-blind, phase 2 trial that included a 24-week placebo-controlled treatment period, followed by an 18-month active-drug extension period. The safety profile in the long-term uncontrolled extension period of the PULSAR study was generally similar to that observed in the STELLAR study. Patients were treated with WINREVAIR 0.3 mg/kg or 0.7 mg/kg (n=104) and had a mean duration of exposure of 151 weeks (maximum 218 weeks).
Intrapulmonary Right-to-Left Shunting: Cases of intrapulmonary right-to-left shunting have been reported in a clinical trial with WINREVAIR. In SOTERIA, an ongoing open-label study of the long-term safety and efficacy of WINREVAIR, right-to-left intrapulmonary shunting has been reported in 2 participants (<0.5%) who developed worsening hypoxemia despite improved PAH hemodynamics. Post-marketing cases have also been reported.
Partial to complete improvement in oxygenation has been observed following discontinuation of WINREVAIR.
6.2 Post-marketing Experience
The following adverse reaction has been reported during post-approval use of WINREVAIR. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac disorders: pericardial effusion
8 USE IN SPECIFIC POPULATIONS
Lactation: Breastfeeding not recommended. (8.2)
8.1 Pregnancy
Risk Summary
Based on findings in animal reproduction studies, WINREVAIR may cause fetal harm when administered to a pregnant woman. There are risks to the mother and the fetus associated with pulmonary arterial hypertension in pregnancy (see Clinical Considerations). There are no available data on WINREVAIR use in pregnant women to inform a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
In animal reproduction studies, administration of WINREVAIR to pregnant rats and rabbits during the period of organogenesis resulted in adverse developmental outcomes, including embryo-fetal mortality, alterations to growth, and structural variations at exposures 4-fold and 0.6-fold (based on area under the curve [AUC]) above those occurring at the maximum recommended human dose (MRHD), respectively (see Data). Advise pregnant women of the potential risk to a fetus [see Use in Specific Populations (8.3)].
The background risk of major birth defects and miscarriage for the indicated population is not known. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Report exposure during pregnancy or lactation to the Merck Sharp & Dohme, LLC Adverse Event reporting line at 1-877-888-4231.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
In patients with pulmonary arterial hypertension, pregnancy is associated with an increased rate of maternal and fetal morbidity and mortality, including spontaneous abortion, intrauterine growth restriction, and premature labor.
Data
Animal Data
In embryo-fetal developmental toxicity studies, pregnant animals were dosed subcutaneously with sotatercept-csrk during the period of organogenesis. Sotatercept-csrk was administered to rats on gestation days 6 and 13 at doses of 5, 15, or 50 mg/kg and to rabbits on gestation days 7 and 14 at doses of 0.5, 1.5, or 5 mg/kg. Effects in both species included reductions in numbers of live fetuses and fetal body weights, delays in ossification, and increases in resorptions and post-implantation losses. In rats and rabbits, these effects were observed at exposures (based on area under the curve [AUC]) approximately 4-fold and 0.6-fold the maximum recommended human dose (MRHD), respectively. In rats only, skeletal variations (increased number of supernumerary ribs and changes in the number of thoracic or lumbar vertebrae) occurred at an exposure 15-fold the human exposure at the MRHD.
In a prenatal and postnatal development study in rats, sotatercept-csrk was administered subcutaneously at doses of 1.5 and 5 mg/kg on gestation days 6 and 13, or at dosages of 1.5, 5, or 10 mg/kg during lactation on days 1, 8, and 15. There were no adverse effects in first filial generation (F1) pups from dams dosed during gestation at estimated exposures up to 2-fold the MRHD. In F1 pups from dams dosed during lactation, decreases in pup weight correlated with delays in sexual maturation at estimated exposures (based on AUC) ≥2-fold the MRHD.
8.2 Lactation
Risk Summary
There are no data on the presence of sotatercept-csrk in human milk, the effects on the breastfed infant, or the effects on milk production. Because of the potential for serious adverse reactions in the breastfed child, advise patients that breastfeeding is not recommended during treatment with WINREVAIR, and for 4 months after the final dose.
8.3 Females and Males of Reproductive Potential
WINREVAIR may cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)].
Pregnancy Testing
Pregnancy testing is recommended for females of reproductive potential before starting WINREVAIR treatment.
Contraception
Females
Advise female patients of reproductive potential to use effective contraception during treatment with WINREVAIR and for at least 4 months after the final dose if treatment is discontinued [see Use in Specific Populations (8.1)].
Infertility
Based on findings in animals, sotatercept-csrk may impair female and male fertility [see Nonclinical Toxicology (13.1)]. In male rats, although adverse histologic changes in reproductive organs were not reversible after a 13-week period, functional fertility demonstrated reversibility.
8.4 Pediatric Use
The safety and effectiveness of WINREVAIR have not been established in patients less than 18 years of age.
8.5 Geriatric Use
A total of 127 patients ≥65 years of age participated in clinical studies for PAH, of which 99 (78%) were treated with WINREVAIR. No differences in efficacy of WINREVAIR were observed between the <65-year-old and ≥65-year-old subgroups.
With the exception of bleeding events (a collective group of adverse events of clinical interest), there were no differences in safety between the <65-year-old and ≥65-year-old subgroups. Bleeding events occurred more commonly in the older WINREVAIR subgroup, but with no imbalance between age subgroups for any specific bleeding event.
Clinical studies of WINREVAIR did not include sufficient numbers of patients aged 75 and older to determine whether they respond differently from younger patients.
10 OVERDOSAGE
In healthy volunteers, WINREVAIR dosed at 1 mg/kg resulted in increases in Hgb associated with hypertension; both improved with phlebotomy. In the event of overdose, monitor closely for increases in Hgb and blood pressure, and provide supportive care as appropriate. WINREVAIR is not dialyzable.
11 DESCRIPTION
Sotatercept-csrk is a homodimeric recombinant fusion protein consisting of the extracellular domain of the human activin receptor type IIA (ActRIIA) linked to the human IgG1 Fc domain. The molecular weight based on the amino acid sequence of sotatercept-csrk is approximately 78 kDa as a homodimer.
Sotatercept-csrk for injection is a sterile, preservative-free, white to off-white lyophilized cake or powder appearance in single-dose vials for subcutaneous administration after reconstitution.
Each 45 mg single-dose vial provides 45 mg of sotatercept-csrk and citric acid monohydrate (0.40 mg), polysorbate 80 (0.18 mg), sodium citrate (1.84 mg), and sucrose (72 mg) at pH 5.8. After reconstitution with 1 mL Sterile Water for Injection, the resulting concentration is 50 mg/mL of sotatercept-csrk and the nominal deliverable volume is 0.9 mL.
Each 60 mg single-dose vial provides 60 mg of sotatercept-csrk and citric acid monohydrate (0.53 mg), polysorbate 80 (0.24 mg), sodium citrate (2.45 mg), and sucrose (96 mg) at pH 5.8. After reconstitution with 1.3 mL Sterile Water for Injection, the resulting concentration is 50 mg/mL of sotatercept-csrk and the nominal deliverable volume is 1.2 mL.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Sotatercept-csrk, a recombinant activin receptor type IIA-Fc (ActRIIA-Fc) fusion protein, is an activin signaling inhibitor that binds to activin A and other TGF- β superfamily ligands. As a result, sotatercept-csrk improves the balance between the pro-proliferative (ActRIIA/Smad2/3-mediated) and anti-proliferative (BMPRII/Smad1/5/8-mediated) signaling to modulate vascular proliferation. In rat models of PAH, a sotatercept-csrk analog reduced inflammation and inhibited proliferation of endothelial and smooth muscle cells in diseased vasculature. These cellular changes were associated with thinner vessel walls, partial reversal of right ventricular remodeling, and improved hemodynamics.
12.2 Pharmacodynamics
STELLAR
A greater decrease from baseline in pulmonary vascular resistance (PVR) was observed in the WINREVAIR group compared to the placebo group. The median treatment difference in PVR between sotatercept-csrk and placebo was -235 dynes*sec/cm5 (95% CI: -288, -181). Sotatercept-csrk steady state exposure at 0.7 mg/kg dose was associated with near maximal reduction in PVR based on exposure-response analysis.
A greater decrease from baseline in NT-proBNP was observed in the WINREVAIR group compared to the placebo group. The median treatment difference in NT-proBNP between the sotatercept-csrk and placebo was -442 pg/mL (95% CI: -574, -310).
ZENITH
The median treatment difference in change in PVR from baseline between the sotatercept and placebo groups after 24 weeks was -340 dynes*sec/cm5 (95% CI: -511, -168). The median treatment difference in change in NT-proBNP from baseline between the sotatercept-csrk and placebo groups after 24 weeks was -2339 pg/mL (95% CI: -3379, -1299). The median treatment difference in change from baseline in mean pulmonary artery pressure (mPAP) between the sotatercept-csrk and placebo groups after 24 weeks was -21.2 mm Hg (95% CI: -27.8, -14.6).
12.3 Pharmacokinetics
Following subcutaneous administration of 0.7 mg/kg WINREVAIR every three weeks to PAH patients (PULSAR, SPECTRA, and STELLAR), the steady state geometric mean (%CV) area under the time concentration curve (AUC) is 171.3 mcg×d/mL (34.2%), and peak concentration (Cmax) is 9.7 mcg/mL (30%). Sotatercept-csrk AUC and Cmax increased proportionally with dose. Steady state is achieved after approximately 15 weeks following initiation of multiple dosing. The accumulation ratio of sotatercept-csrk AUC is approximately 2.2. Sotatercept-csrk pharmacokinetics were similar in PAH participants in ZENITH.
Absorption
Following subcutaneous administration, the absolute bioavailability of sotatercept-csrk is approximately 66%. The sotatercept-csrk median time to peak drug concentration (Tmax) is approximately 7 days (range from 2 to 8 days) following multiple SC administration every 4 weeks.
Distribution
The population PK model estimated volume of distribution (%CV) of sotatercept-csrk at steady state is approximately 5.3 L (27.3%) in patients with PAH.
Elimination
The sotatercept-csrk effective half-life is approximately 24 days and its clearance is approximately 0.18 L/day.
Metabolism
Sotatercept-csrk is expected to be metabolized into small peptides by catabolic pathways.
Specific Populations
No clinically significant differences in sotatercept-csrk pharmacokinetics (PK) were observed based on age (18 to 81 years of age), sex, race, mild to moderate (eGFR ranging from 30 to 89 mL/min) renal impairment (PAH patients), or end-stage kidney disease (eGFR <15 mL/min) with dialysis. Based on limited data, severe renal impairment (eGFR ranging from 15 to 30 mL/min, n=3) had no impact on the PK of sotatercept-csrk. Sotatercept-csrk is not dialyzable. The effect of hepatic impairment on the PK of sotatercept-csrk has not been studied.
Body Weight
The clearance (CL) and central volume of distribution (Vc) increase with increase in body weight. This effect is not clinically significant when sotatercept-csrk is administered using weight-based dosing as recommended.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of WINREVAIR or of other sotatercept-csrk products.
During the 24-week treatment period in STELLAR, 27% (44/163) of sotatercept-csrk-treated patients developed anti-sotatercept-csrk antibodies (ADA). Among the 44 ADA-positive patients, 12 (27%) tested positive for neutralizing antibodies against sotatercept-csrk.
During the ZENITH trial, with a median sotatercept treatment duration of 435 days, 43% (36/84) of patients developed anti-sotatercept antibodies. Among these 36 patients, 20 (56%) tested positive for neutralizing antibodies against sotatercept.
There were no identified clinical effects of anti-sotatercept-csrk antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of sotatercept-csrk over the treatment duration of 24 weeks at the recommended dosage in these studies.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity or mutagenicity studies have been conducted with sotatercept-csrk.
In a fertility and early embryonic development study in female rats, sotatercept-csrk was administered SC once weekly at doses of 5, 15, and 50 mg/kg beginning 2 weeks prior to mating and through gestation day 7. At doses ≥15 mg/kg (≥9 fold the MRHD, based on estimated AUC), pregnancy rates were decreased and there were increases in preimplantation and postimplantation loss and reductions in live litter size. Increased estrous cycle duration occurred at 50 mg/kg only (21-fold the MRHD, based on estimated AUC).
In a fertility study in male rats, sotatercept-csrk was administered SC once weekly at doses of 0.3, 3, and 30 mg/kg for 13 weeks (beginning 10 weeks prior to mating). A subset of animals was examined after a 13-week recovery period. At ≥0.3 mg/kg (0.5-fold the MRHD, based on estimated AUC) there were non-reversible histologic changes in the efferent ducts, testes, and epididymides. Reversible decreases in functional fertility endpoints occurred at 30 mg/kg (20-fold the MRHD, based on estimated AUC).
14 CLINICAL STUDIES
14.1 Pulmonary Arterial Hypertension
STELLAR
The efficacy of WINREVAIR was evaluated in adult patients with PAH in the STELLAR trial (NCT04576988). STELLAR was a global, double-blind, placebo-controlled, multicenter, parallel-group clinical trial in which 323 patients with PAH (WHO Group 1, FC II or III) were randomized 1:1 to WINREVAIR (target dose 0.7 mg/kg) (n=163) or placebo (n=160) administered subcutaneously once every 3 weeks.
Participants were: 79% female; had a median age of 48 years (range: 18 to 82 years), and median body weight of 68 kg (range 38 to 141 kg); and 89% White, 2% Black/African American, 2% Asian, 0.3% American Indian or Alaska Native, 0.3% Native Hawaiian or Other Pacific Islander, 6% Missing/Other races. The most common PAH etiologies were idiopathic PAH (59%), heritable PAH (18%), and PAH associated with connective tissue diseases (CTD) (15%). STELLAR excluded patients with human immunodeficiency virus (HIV)-associated PAH, PAH associated with portal hypertension, schistosomiasis-associated PAH, and pulmonary veno occlusive disease. The mean time from PAH diagnosis to screening was 8.8 years. Most participants were receiving either three (61%) or two (35%) background drugs for PAH, and 40% were receiving prostacyclin infusions. Patients had a WHO FC II (49%) or III (51%) at baseline.
The primary efficacy endpoint was the change from baseline at Week 24 in 6-Minute Walk Distance (6 MWD). In the WINREVAIR group, the placebo-adjusted median increase in 6 MWD was 41 meters (95% CI: 28, 54; p<0.001). Figure 1 displays placebo-adjusted changes in 6 MWD at Week 24 in relevant subgroups.
Treatment with WINREVAIR led to an improvement from baseline by at least 1 WHO FC at Week 24 in 29% of patients compared to 14% of patients treated with placebo (p<0.001).
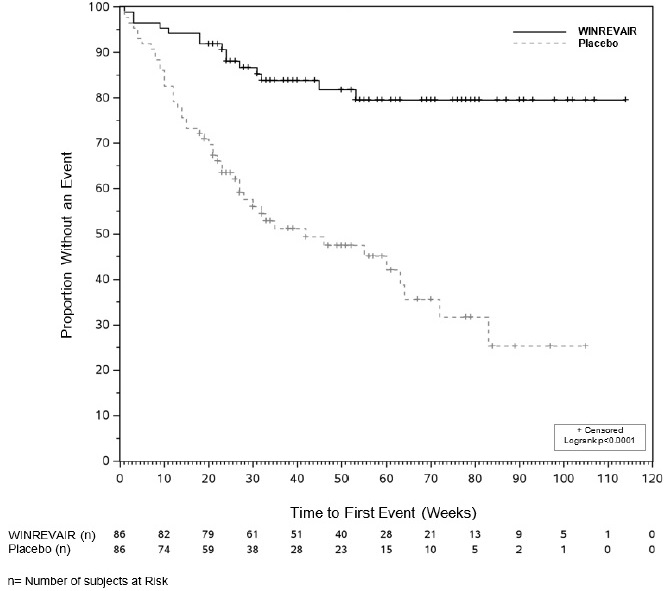
Treatment with WINREVAIR resulted in an 84% reduction in the occurrence of death from any cause or PAH clinical worsening events compared to placebo (see Table 5 and Figure 2). These outcomes were captured until the last patient completed the Week 24 visit (data up to the data cutoff; median duration of exposure 33.6 weeks).
ZENITH
The efficacy of WINREVAIR was evaluated in adult PAH patients with WHO FC III or IV at high risk of mortality in the ZENITH trial (NCT04896008). ZENITH was a global, double-blind, placebo-controlled, multicenter, parallel-group clinical trial in which 172 patients were randomized 1:1 to WINREVAIR (target dose 0.7 mg/kg) (n=86) or placebo (n=86) administered subcutaneously once every 3 weeks. Efficacy was evaluated at the pre-specified interim analysis which occurred when 61 patients experienced a primary endpoint event and median patient time on study was 273 days.
The demographic and baseline clinical characteristics were similar between the WINREVAIR and placebo groups. Participants were: 77% female; had a median age of 58 years (range: 18 to 75 years); and 87% White, 5% Black/African American, 4% Asian, 1% American Indian or Alaska Native, 3% Missing/Other race, 74% were FC III and 26% were FC IV. The most common PAH etiologies were idiopathic PAH (50%), PAH associated with connective tissue diseases (CTD) (28%), and heritable PAH (11%). The mean time since PAH diagnosis to screening was 8 years. Participants were on background PAH treatment, 72% on triple therapy, 28% on double therapy, and 59% on prostacyclin infusion therapy. The REVEAL Lite 2 risk score was <9 for 2% of participants, 9 to 10 for 67% of participants, and ≥11 for 30% of participants. The ZENITH trial excluded patients diagnosed with human immunodeficiency virus (HIV)-associated PAH, PAH associated with portal hypertension, pulmonary veno-occlusive disease, or pulmonary capillary hemangiomatosis or overt signs of capillary and/or venous involvement.
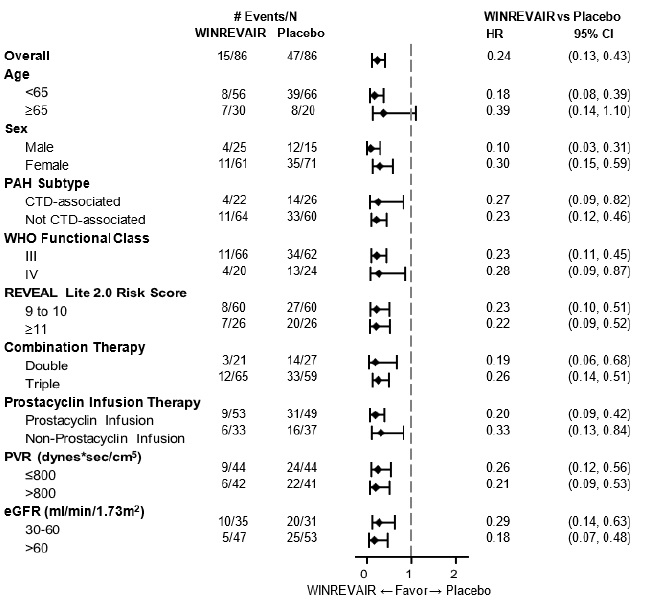
The primary efficacy endpoint was time to first event of all-cause death, lung transplantation, or PAH worsening-related hospitalization of ≥24 hours. In the WINREVAIR treatment group, the risk of a first event of all-cause death, lung transplantation, or PAH worsening-related hospitalization of ≥24 hours was 76% lower compared with the placebo group (HR: 0.24; 95% CI: 0.13, 0.43; p<0.0001) (see Table 6). Fewer participants in the WINREVAIR group (15 [17%]) than in the placebo group (47 [55%]) had a primary endpoint event as of the data cutoff. Based on the primary endpoint result, the study was stopped for favorable efficacy at the interim analysis.
The treatment effect of WINREVAIR was consistent across the prespecified subgroups (see Figure 4).
The secondary endpoint of overall survival (OS) included all deaths up to the data cutoff, except for those occurring after lung transplantation or enrollment in a long-term follow-up study. Twenty OS events were observed (7 deaths in the WINREVAIR treatment group and 13 deaths in the placebo group). The point estimate for the OS HR favored the WINREVAIR treatment group over the placebo group.
Subsequent secondary endpoints were not eligible to be tested due to the hierarchical testing strategy.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
WINREVAIR (sotatercept-csrk) for injection is a white to off-white lyophilized cake or powder appearance supplied in single-dose vials (45 mg or 60 mg) packaged in kits that contain one measuring syringe and one safety needle. Each kit also contains Sterile Water for Injection in prefilled syringes necessary to reconstitute the product, vial adapter(s), and alcohol pads, as shown in Table 7.
16.2 Storage and Handling
Store vials refrigerated at 2°C to 8°C (36°F to 46°F) in original carton to protect from light. Do not freeze.
The kit should remain in the refrigerator until ready for use. The unused kit can be out of the refrigerator for (up to 25°C/77°F) up to 24 hours. For additional information on temperature excursions, call Merck Sharp & Dohme LLC at 1-800-672-6372.
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Patient Information and IFU).
Discuss the following with patients prior to and during treatment with WINREVAIR.
Erythrocytosis
Caution patients that WINREVAIR may raise Hgb to levels that increase their risk of thrombotic events. Inform patients that Hgb levels will be assessed before at least the first 5 doses and then periodically, as dosage may need to be adjusted [see Warnings and Precautions (5.1)].
Severe Thrombocytopenia
Caution patients that WINREVAIR may cause platelet count to decrease, which if severe could cause bleeding. Inform patients that platelet count will be assessed before at least the first 5 doses and then periodically, as dosage may need to be adjusted [see Warnings and Precautions (5.2)].
Serious Bleeding
Inform patients of the possibility of serious bleeding, which is more likely to occur if they have low platelet counts or while on prostacyclin background therapy and/or antithrombotic agents. Advise patients to notify their healthcare provider about signs and symptoms of bleeding [see Warnings and Precautions (5.3)].
Embryo-Fetal Toxicity
Advise females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception while receiving WINREVAIR and for at least 4 months after the final dose. Advise females to contact their healthcare provider if they become pregnant, or if pregnancy is suspected during treatment with WINREVAIR [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)]. Report exposure during pregnancy or lactation to the Merck Sharp & Dohme, LLC Adverse Event reporting line at 1-877-888-4231.
Lactation
Advise females not to breastfeed during treatment with WINREVAIR and for 4 months after the final dose [see Use in Specific Populations (8.2)].
Females and Males of Reproductive Potential
Advise females and males of reproductive potential that WINREVAIR may impair fertility [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)].
Administration by Patient or Caregiver
Review the IFU with the patient or caregiver step-by-step. Provide training to the patient or caregiver regarding proper preparation and administration of WINREVAIR and decide whether a patient or caregiver is capable of preparing and administering WINREVAIR independently [see Dosage and Administration (2.4)].
Make sure the patient or caregiver can do the following correctly:
- reconstitute the medicine,
- measure the correct amount of medicine according to the patient’s prescription,
- select and prepare a proper injection site, and
- inject the medicine subcutaneously.
Incorrect Dose or Missed Dose
Inform patients to call their healthcare provider for further instruction if they take more than or less than the correct dose. Advise them about signs/symptoms to monitor for and what to do if any of these signs/symptoms should occur. Advise them that additional laboratory tests may be required prior to the next scheduled dose to ensure that the next dose can be safely administered.
Instruct the patient that if they miss the prescribed dose of WINREVAIR, they should take it within 3 days and maintain the original schedule for the next dose. If not taken within 3 days, instruct them to call their healthcare provider for guidance [see Dosage and Administration (2.2)].
Manufactured by:
Merck Sharp & Dohme LLC, Rahway, NJ 07065, USA
U.S. license number 0002
For patent information: www.msd.com/research/patent
Copyright © 2024-2025 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
uspi-mk7962-i-2512r004
In this booklet:
Important information for patients or caregivers
Get to know the parts of your kit
Important information you need to know before injecting WINREVAIR
Before you prepare and inject WINREVAIR, you must be:
- trained by a healthcare provider following this Instructions for Use booklet step-by-step, and
- capable of independent administration of WINREVAIR as determined by a healthcare provider
Before using WINREVAIR, make sure you are trained to do the following correctly:
- Reconstitute (mix) the medicine
- Measure the correct amount of medicine according to your prescription
- Select and prepare a proper injection site
- Inject the medicine subcutaneously
Get to know the parts of your kit
Important information you need to know before injecting WINREVAIR
Get started
Before you can prepare and inject WINREVAIR, you must first be trained and determined to be capable of independent administration of WINREVAIR by a healthcare provider.
1 Check WINREVAIR injection kit and expiration date
2 Let your kit come to room temperature, gather supplies, and wash your hands
Mix powdered medicine into liquid form
Start with the Top Tray
3 Remove medicine vial, prefilled syringe with sterile water for injection, alcohol wipes, and vial adapter from the kit.
4a Check medicine vial
4b Remove plastic cap and clean vial
5a Align vial adapter to vial
5b Attach vial adapter to vial
5c Clean vial adapter
6 Check prefilled syringe
7 Snap off prefilled syringe white cap
8 Connect prefilled syringe to vial adapter
9 Transfer sterile water from prefilled syringe to vial
10 Swirl to mix medicine
11 Wait for large bubbles in the vial to go away
12 Prepare vial by removing extra air
13 Remove prefilled syringe from vial
Withdraw your prescribed dose
For the next steps, you will need:
-
- Mixed vial of medicine -
- Items from the Bottom Tray (needle, dosing syringe for injection, and alcohol wipes).
14 Clean top of vial adapter
15 Remove empty dosing syringe from its package
16 Pull air into the dosing syringe
17 Connect dosing syringe to the vial
18 Push air into vial, then flip vial upside down
19 Pull plunger back to withdraw your prescribed dose
20 Check for large air bubbles and air pockets
21 Remove air bubbles and air pockets from syringe
22 Compare amount to prescribed dose
23 Confirm your prescribed dose
24 Remove the dosing syringe from the vial and set dosing syringe aside
25 Attach injection needle
26 Choose and clean your injection site
Now, you are ready to inject the medicine.
Inject your medicine
27 Inject your medicine
Frequently asked questions
What should I do if I am bleeding at the injection site?
Place a cotton ball or bandage on your skin right away and apply a small amount of pressure. If the bleeding does not stop, call your healthcare provider right away.
Where can I find my prescribed dose?
Your prescribed dose in ‘mL’ is on your prescription label on your kit. Contact your healthcare provider if you cannot find your prescribed dose.
What should I do if I accidentally get some medicine on my skin or my work surface?
Wash the area thoroughly with soap and water right away.
What should I do if I am not sure I administered my prescribed dose correctly?
Call your pharmacy or healthcare provider.
What should I do if the plunger of my dosing syringe moves automatically when I try to withdraw medicine from the vial?
Do not worry if your plunger moves slightly on its own when you are filling your dosing syringe with medicine.
With one hand, hold the plunger in place to stop the plunger from moving. With the other hand, unscrew the vial from the dosing syringe.
After unscrewed, it is safe to let go of the plunger.
You can avoid this automatic plunger movement by pushing air into the vial before filling your dosing syringe with medicine. Refer to Steps 16 to 23 for detailed instructions.
What should I do if my kit parts are damaged or are discolored, cloudy, or have particles?
Do not use your kit. Call your pharmacy to get a new kit.
What should I do if my medicine does not turn clear after mixing and swirling?
Do not use the medicine if you have swirled the medicine vial for about 2 minutes and let it stand for another 3 minutes but your medicine vial remains cloudy or has clumps, powder, or foreign particles. Call your pharmacy to get a new kit.
What should I do if the sterile water will not come out of the prefilled syringe?
Check that the vial adapter is attached to the vial securely. If not, hold the vial and press the vial adapter down firmly to make sure the vial adapter punctures the vial rubber stopper.
What should I do if I dropped my kit components?
Do not use if any items are damaged. If you are unsure, call your pharmacy to get a new kit.
Can I use my kit that has been left out of the refrigerator?
Store the injection kit in the refrigerator until ready to use it. If the unused injection kit has been out of the refrigerator up to 77°F (25°C) for more than 24 hours, please contact your pharmacy or healthcare provider.
Do I need to use mixed medicine right away?
We recommend you inject the medicine right away after mixing but no later than 4 hours after mixing. If it has been more than 4 hours, throw away unused mixed medicine. If you have questions, please contact your pharmacy or healthcare provider.
How can I get help preparing and giving my injection?
If you have questions about how to give WINREVAIR the correct way or need more information, you can call your pharmacy or healthcare provider.
Call your doctor for advice about side effects.
To report side effects or product quality complaints, please call Merck Sharp & Dohme LLC at 1-800-672-6372. You can also report side effects to FDA at 1-800-FDA-1088.
Manufactured by:
Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
U.S. License Number 0002
Copyright © 2024 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
usifu1-mk7962-i-2403r000
This Instructions for Use has been approved by the U.S. Food and Drug Administration
Approved: March 2024
In this booklet:
Important information for patients or caregivers
Get to know the parts of your kit
Important information you need to know before injecting WINREVAIR
Before you prepare and inject WINREVAIR, you must be:
- trained by a healthcare provider following this Instructions for Use booklet step-by-step, and
- capable of independent administration of WINREVAIR as determined by a healthcare provider
Before using WINREVAIR, make sure you are trained to do the following correctly:
- Reconstitute (mix) the medicine
- Combine medicine from both vials
- Measure the correct amount of medicine according to your prescription
- Select and prepare a proper injection site
- Inject the medicine subcutaneously
How to use WINREVAIR
Before injecting WINREVAIR, make sure you read the full instructions in this booklet to get your prescribed dose.
Proceed to the next pages for step-by-step directions.
Get to know the parts of your kit
Important information you need to know before injecting WINREVAIR
Get started
1 Check WINREVAIR injection kit and expiration date
2 Let your kit come to room temperature, gather supplies, and wash your hands
Mix powdered medicine into liquid form
Start with the Top Tray
3 Remove medicine vial 1, prefilled syringe with sterile water for injection 1, alcohol wipes, and vial adapter 1 from the kit.
4a Check medicine vial
4b Remove plastic cap and clean vial
5a Align vial adapter to vial
5b Attach vial adapter to vial
5c Clean vial adapter
6 Check prefilled syringe
7 Snap off prefilled syringe white cap
8 Connect prefilled syringe to vial adapter
9 Transfer sterile water from prefilled syringe to vial
10 Prepare vial 2, prefilled syringe 2, and vial adapter 2
11 Swirl to mix medicine
12 Wait for large bubbles in the vials to go away
13 Prepare vials by removing extra air
14 Remove prefilled syringes from vials
Combine medicine from both vials
For the next steps, you will need:
-
- Mixed vial 1 and vial 2 -
- Items from the Bottom Tray (needle, dosing syringe for injection, and alcohol wipes).
15 Clean top of both vial adapters
16 Remove empty dosing syringe from its package
17 Pull air into the dosing syringe
18 Connect dosing syringe to one of the vials
19 Push air into vial, then flip upside down
20 Withdraw all the medicine from 1st vial
21 Remove 1st vial from the dosing syringe
22 Connect the dosing syringe to the 2nd vial
23 Push all the medicine into the 2nd vial, then flip vial upside down
Withdraw your prescribed dose
24 Pull plunger back to withdraw your prescribed dose
25 Check for large bubbles and air pockets
26 Remove air bubbles and air pockets from syringe
27 Compare amount to prescribed dose
28 Confirm your prescribed dose
29 Remove the dosing syringe from the vial and set dosing syringe aside
30 Attach injection needle to dosing syringe
31 Choose and clean your injection site
Now, you are ready to inject the medicine.
Inject your medicine
32 Inject your medicine
Frequently asked questions
What should I do if I am bleeding at the injection site?
Place a cotton ball or bandage on your skin right away and apply a small amount of pressure. If the bleeding does not stop, call your healthcare provider right away.
Where can I find my prescribed dose?
Your prescribed dose in ‘mL’ is on your prescription label on your kit. Contact your healthcare provider if you cannot find your prescribed dose.
What should I do if I accidentally get some medicine on my skin or my work surface?
Wash the area thoroughly with soap and water right away.
What should I do if I am not sure I administered my prescribed dose correctly?
Call your pharmacy or healthcare provider.
What should I do if the plunger of my dosing syringe moves automatically when I try to withdraw medicine from the vial?
Do not worry if your plunger moves slightly on its own when you are filling your dosing syringe with medicine.
With one hand, hold the plunger in place to stop the plunger from moving. With the other hand, unscrew the vial from the dosing syringe.
After unscrewed, it is safe to let go of the plunger.
You can avoid this automatic plunger movement by pushing air into the vial before filling your dosing syringe with medicine. Refer to Steps 17 to 28 for detailed instructions.
What should I do if my kit parts are damaged or are discolored, cloudy, or have particles?
Do not use your kit. Call your pharmacy to get a new kit.
What should I do if my medicine does not turn clear after mixing and swirling?
Do not use the medicine if you have swirled the medicine vial for about 2 minutes and let it stand for another 3 minutes but your medicine vial remains cloudy or has clumps, powder, or foreign particles. Call your pharmacy to get a new kit.
What should I do if the sterile water will not come out of the prefilled syringe?
Check that the vial adapter is attached to the vial securely. If not, hold the vial and press the vial adapter down firmly to make sure the vial adapter punctures the vial rubber stopper.
What should I do if I dropped my kit components?
Do not use if any items are damaged. If you are unsure, call your pharmacy to get a new kit.
Can I use my kit that has been left out of the refrigerator?
Store the injection kit in the refrigerator until ready to use it. If the unused injection kit has been out of the refrigerator up to 77°F (25°C) for more than 24 hours, please contact your pharmacy or healthcare provider.
Do I need to use mixed medicine right away?
We recommend you inject the medicine right away after mixing but no later than 4 hours after mixing. If it has been more than 4 hours, throw away unused mixed medicine. If you have questions, please contact your pharmacy or healthcare provider.
How can I get help preparing and giving my injection?
If you have questions about how to give WINREVAIR the correct way or need more information, you can call your pharmacy or healthcare provider.
Call your doctor for advice about side effects.
To report side effects or product quality complaints, please call Merck Sharp & Dohme LLC at 1-800-672-6372. You can also report side effects to FDA at 1-800-FDA-1088.
Manufactured by:
Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
U.S. License Number 0002
Copyright © 2024 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
usifu2-mk7962-i-2403r000
This Instructions for Use has been approved by the U.S. Food and Drug Administration
Approved: March 2024
PRINCIPAL DISPLAY PANEL - 45 mg Vial Kit Carton
NDC 0006-5090-01
WINREVAIR™
(sotatercept-csrk) for injection
45 mg/vial
Package
contains
1 Vial
Dose
based on
patient
weight
For Subcutaneous Use Only
Must be reconstituted with diluent provided
prior to administration
1 Single-Dose Vial
Discard unused portion
Rx only
PRINCIPAL DISPLAY PANEL - 60 mg Vial Kit Carton
NDC 0006-5091-01
WINREVAIR™
(sotatercept-csrk) for injection
60 mg/vial
Package
contains
1 Vial
Dose
based on
patient
weight
For Subcutaneous Use Only
Must be reconstituted with diluent provided
prior to administration
1 Single-Dose Vial
Discard unused portion
Rx only
PRINCIPAL DISPLAY PANEL - 45 mg 2 Vials Kit Carton
NDC 0006-5087-01
WINREVAIR™
(sotatercept-csrk) for injection
45 mg/vial
Package
contains
2 Vials
Dose
based on
patient
weight
For Subcutaneous Use Only
Must be reconstituted with diluent provided
prior to administration
2 Single-Dose Vials
Discard unused portion
Rx only
PRINCIPAL DISPLAY PANEL - 60 mg 2 Vials Kit Carton
NDC 0006-5088-01
WINREVAIR™
(sotatercept-csrk) for injection
60 mg/vial
Package
contains
2 Vials
Dose
based on
patient
weight
For Subcutaneous Use Only
Must be reconstituted with diluent provided
prior to administration
2 Single-Dose Vials
Discard unused portion
Rx only
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.