Capmist DM (Dextromethorphan Hydrobromide, Guaifenesin and Pseudoephedrine Hydrochloride) tablet
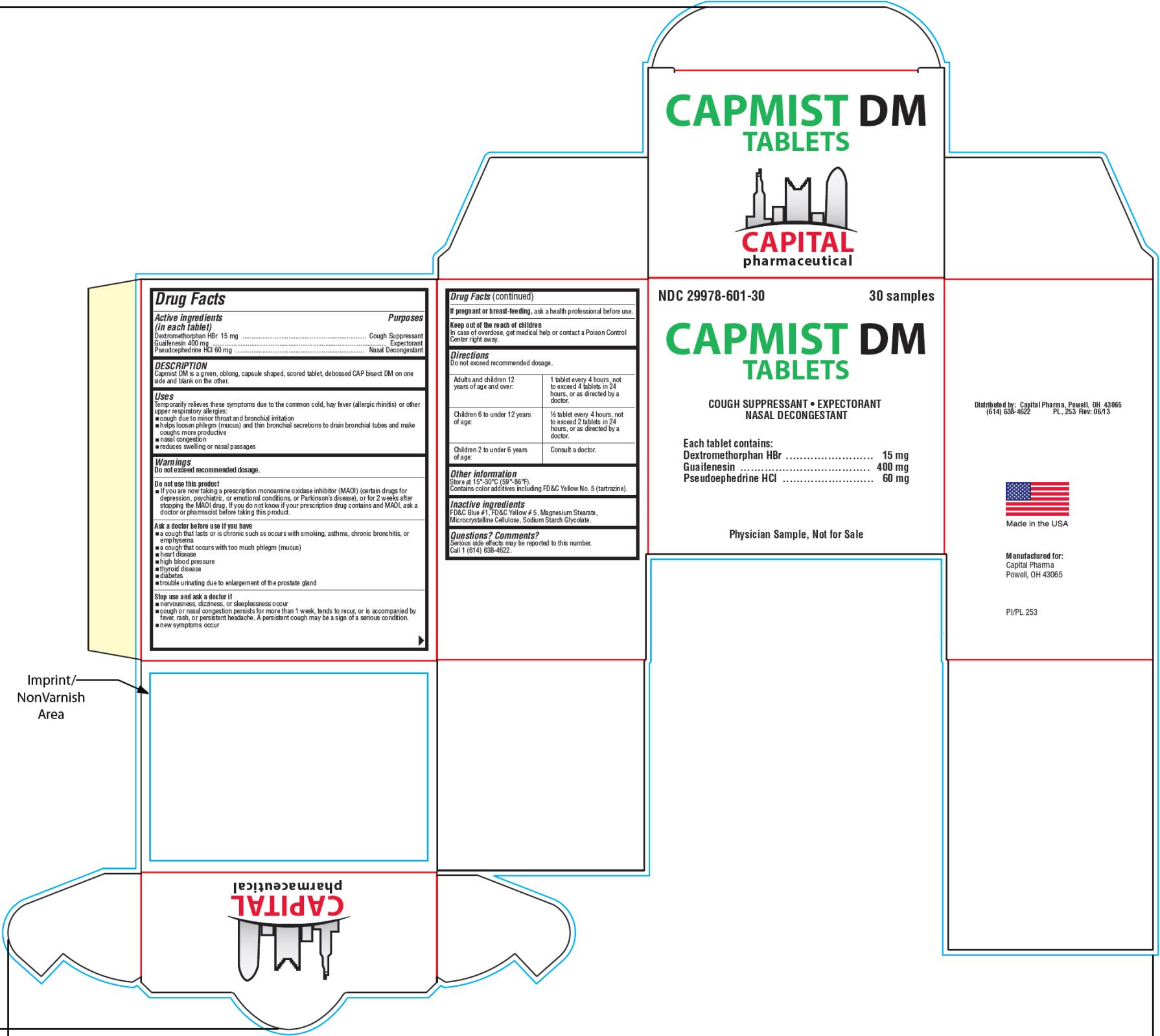
Drug Facts
Active ingredients
(in each tablet)
Dextromethorphan HBr 15mg
Purpose
Cough Suppressant
Active ingredients
(in each tablet)
Guaifenesin 400mg
Purpose
Expectorant
Active ingredients
(in each tablet)
Pseudoephedrine HCl 60mg
Purpose
Nasal Decongestant
Uses
Temporarily relieves these symptoms due to the common cold, hay fever (allergic rhinitis) or other upper respiratory allergies:
- cough due to minor throat and bronchial irritation
- helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes and make coughs more productive
- nasal congestion
- reduces swelling of nasal passages
Warnings
Do not exceed recommended dosage.
Do not use this product
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- a cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- a cough that occurs with too much phlegm (mucus)
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to enlargement of the prostate gland
Stop use and ask a doctor if:
- nervousness, dizziness, or sleeplessness occur
- cough or nasal congestion persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache. A persistent cough may be a sign of a serious condition.
- new symptoms occur
If pregnant or breast feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
Other information
Store at 15°-30°C (59°-86°F).
Capmist DM is a light green, oblong, capsule shaped, scored tablet, debossed “CAP” bisect “DM” on one side and blank on the other.
Contains color additives including FD&C Yellow No. 5 (tartrazine).
Supplied in a tight, light-resistant container with a child-resistant cap.
Inactive ingredients
FD&C Blue #1, FD&C Yellow #5, Magnesium Stearate, Microcrystalline Cellulose, Sodium Starch Glycolate.
PRINCIPAL DISPLAY PANEL
NDC
29978-601-90
CAPMIST
DM Tablets
Cough Suppressant ● Expectorant
Nasal Decongestant
90 tablets
PRINCIPAL DISPLAY PANEL
NDC
29978-601-30
CAPMIST
DM Tablets
Cough Suppressant ●
Expectorant
Nasal Decongestant
3
0
samples
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.