Enhertu (fam-trastuzumab deruxtecan-nxki) injection, powder, lyophilized, for solution
WARNING: INTERSTITIAL LUNG DISEASE and EMBRYO-FETAL TOXICITY
- Interstitial Lung Disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and the need to immediately report symptoms [see Dosage and Administration (2.3), Warnings and Precautions (5.1)].
- Embryo-Fetal Toxicity: Exposure to ENHERTU during pregnancy can cause embryo-fetal harm. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.4), Use in Specific Populations (8.1, 8.3)].
WARNING: INTERSTITIAL LUNG DISEASE and EMBRYO-FETAL TOXICITY
See full prescribing information for complete boxed warning.
- Interstitial lung disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and to immediately report symptoms. (2.3, 5.1)
- Exposure to ENHERTU during pregnancy can cause embryo-fetal harm. Advise patients of these risks and the need for effective contraception. (5.4, 8.1, 8.3)
1 INDICATIONS AND USAGE
ENHERTU is a HER2-directed antibody and topoisomerase inhibitor conjugate indicated:
HER2-Positive Metastatic Breast Cancer
- in combination with pertuzumab as first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test. (1.1)
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy. (1.1)
HER2-Low and HER2-Ultralow Metastatic Breast Cancer
- as monotherapy for the treatment of adult patients with unresectable or metastatic hormone receptor (HR)-positive, HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting. (1.2)
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting; or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy. (1.2)
HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer
- as monotherapy for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy* (1.3)
HER2-Positive Locally Advanced or Metastatic Gastric Cancer
- as monotherapy for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction adenocarcinoma who have received a prior trastuzumab-based regimen. (1.4)
HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options* (1.5)
* These indications are approved under accelerated approval based on objective response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. (14.3, 14.5)
1.1 HER2-Positive Metastatic Breast Cancer
- ENHERTU, in combination with pertuzumab, is indicated for the first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test [see Dosage and Administration (2.1)].
- ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy.
1.2 HER2-Low and HER2-Ultralow Metastatic Breast Cancer
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic
- Hormone receptor (HR)-positive HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting [see Dosage and Administration (2.1)].
- HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy [see Dosage and Administration (2.1)].
1.3 HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy.
This indication is approved under accelerated approval based on objective response rate and duration of response [see Clinical Studies (14.3)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
1.4 HER2-Positive Locally Advanced or Metastatic Gastric Cancer
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction (GEJ) adenocarcinoma who have received a prior trastuzumab-based regimen.
1.5 HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on objective response rate and duration of response [see Clinical Studies (14.5)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
2 DOSAGE AND ADMINISTRATION
- Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine. (2.2, 2.4)
- For intravenous infusion only. Do not administer as an intravenous push or bolus. DO NOT use Sodium Chloride Injection, USP. (2.4)
- Premedicate for prevention of chemotherapy-induced nausea and vomiting. (2.2)
- HER2-positive, HER2-low, or HER2-ultralow breast cancer, HER2-mutant NSCLC, and HER2-positive (IHC 3+) solid tumors: ENHERTU 5.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. (2.2, 2.3)
- HER2-Positive First-line Metastatic Breast Cancer ENHERTU 5.4 mg/kg every 3 weeks (in combination with pertuzumab until disease progression or unacceptable toxicity.
- HER2-positive gastric cancer: 6.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. (2.2, 2.3)
- Management of adverse reactions (ILD, neutropenia, thrombocytopenia, or left ventricular dysfunction) may require temporary interruption, dose reduction, or discontinuation of ENHERTU. (2.3)
2.1 Patient Selection
HER2-Positive Metastatic Breast Cancer
Select patients for treatment of unresectable or metastatic HER2-positive breast cancer with ENHERTU in combination with pertuzumab based on confirmed HER2-positive status or HER2 gene amplification (IHC 3+ or ISH+) [see Clinical Studies (14.1)].
HER2-Low or HER2-Ultralow Unresectable or Metastatic Breast Cancer
Select patients for treatment of unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer with ENHERTU based on HER2 expression [see Clinical Studies (14.2)].
HER2-Mutant Unresectable or Metastatic NSCLC
Select patients for the treatment of unresectable or metastatic HER2-mutant NSCLC with ENHERTU based on the presence of activating HER2 (ERBB2) mutations in tumor or plasma specimens [see Clinical Studies (14.3)]. If no mutation is detected in a plasma specimen, test tumor tissue.
HER2-Positive Locally Advanced or Metastatic Gastric Cancer
Select patients with locally advanced or metastatic HER2-positive gastric cancer based on HER2 protein overexpression or HER2 gene amplification (IHC 3+ or IHC 2+/ISH+). Reassess HER2 status if it is feasible to obtain a new tumor specimen after prior trastuzumab-based therapy and before treatment with ENHERTU.
HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors
Select patients for treatment of unresectable or metastatic solid tumors with ENHERTU based on HER2-positive (IHC 3+) specimens [see Clinical Studies (14.5)]. An FDA-approved test for the detection of HER2-positive (IHC 3+) solid tumors for treatment with ENHERTU is not currently available.
Additional Patient Selection Information
Information on FDA-approved tests for the detection of HER2 protein expression, HER2 gene amplification, and activating HER2 mutations is available at: http://www.fda.gov/CompanionDiagnostics.
2.2 Recommended Dosage and Schedules
Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine.
Slow or interrupt the infusion rate if the patient develops infusion-related symptoms.
Permanently discontinue ENHERTU in case of severe infusion reactions.
Premedication
ENHERTU is highly emetogenic [see Adverse Reactions (6.1)], which includes delayed nausea and/or vomiting. Administer prophylactic antiemetic medications per local institutional guidelines for prevention of chemotherapy-induced nausea and vomiting.
The recommended dosages for ENHERTU as monotherapy and ENHERTU in combination with pertuzumab are presented in Table 1. Administer ENHERTU as an intravenous infusion [see Dosage and Administration (2.4)].
2.3 Dosage Modifications
Management of adverse reactions may require temporary interruption, dose reduction, or treatment discontinuation of ENHERTU as described in Tables 2 and 3. Refer to the Prescribing Information for pertuzumab for dose modification recommendations. Pertuzumab is not to be administered as a single agent.
Do not re-escalate the ENHERTU dose after a dose reduction is made.
If a planned dose is delayed or missed, administer as soon as possible; do not wait until the next planned cycle. Adjust the schedule of administration to maintain a 3-week interval between doses. Administer the infusion at the dose and rate the patient tolerated in the most recent infusion.
2.4 Preparation and Administration
In order to prevent medication errors, check the vial labels to ensure that the drug being prepared and administered is ENHERTU (fam-trastuzumab deruxtecan-nxki) and not trastuzumab or ado-trastuzumab emtansine.
Reconstitute and further dilute ENHERTU prior to intravenous infusion. Use appropriate aseptic technique.
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a hazardous drug. Follow applicable special handling and disposal procedures.1
Reconstitution
- Reconstitute immediately before dilution.
- More than one vial may be needed for a full dose. Calculate the dose (mg), the total volume of reconstituted ENHERTU solution required, and the number of vial(s) of ENHERTU needed [see Dosage and Administration (2.2)].
- Reconstitute each 100 mg vial by using a sterile syringe to slowly inject 5 mL of Sterile Water for Injection, USP into each vial to obtain a final concentration of 20 mg/mL.
- Swirl the vial gently until completely dissolved. Do not shake.
- If not used immediately, store the reconstituted ENHERTU vials in a refrigerator at 2ºC to 8ºC (36ºF to 46ºF) for up to 24 hours from the time of reconstitution, protect the vial from light. Do not freeze.
- The product does not contain a preservative. Discard unused ENHERTU after 24 hours refrigerated.
Dilution
- Withdraw the calculated amount from the vial(s) using a sterile syringe. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. The solution should be clear and colorless to light yellow. Do not use if visible particles are observed or if the solution is cloudy or discolored.
- Dilute the calculated volume of reconstituted ENHERTU in an intravenous infusion bag containing 100 mL of 5% Dextrose Injection, USP. DO NOT use Sodium Chloride Injection, USP. ENHERTU is compatible with an infusion bag made of polyvinylchloride or polyolefin (copolymer of ethylene and polypropylene).
- Gently invert the infusion bag to thoroughly mix the solution. Do not shake.
- Cover the infusion bag to protect from light.
- Discard any unused portion left in the vials.
Administration
- If not used immediately, store the diluted ENHERTU in a refrigerator at 2ºC to 8ºC (36ºF to 46ºF) for up to 24 hours or at room temperature between 20ºC to 25ºC (68ºF to 77 ºF) for up to 4 hours including preparation and infusion time.
- Protect from light. Do not freeze.
- The maximum time from reconstitution of the vial through the end of administration should not exceed 24 hours.
- If the prepared infusion solution was stored refrigerated (2ºC to 8ºC [36ºF to 46ºF]), allow the solution to reach room temperature prior to administration. Cover the infusion bag to protect from light.
- Administer ENHERTU as an intravenous infusion only with an infusion set made of polyolefin or polybutadiene.
- Administer ENHERTU with a 0.20 or 0.22 micron in-line polyethersulfone (PES) or polysulfone (PS) filter.
- Do NOT administer as an intravenous push or bolus.
- Cover the infusion bag to protect from light during administration.
- Do not mix ENHERTU with other drugs or administer other drugs through the same intravenous line.
- First infusion: Administer infusion over 90 minutes.
- Subsequent infusions: Administer over 30 minutes if prior infusions were well tolerated.
3 DOSAGE FORMS AND STRENGTHS
For injection: 100 mg of fam-trastuzumab deruxtecan-nxki as a white to yellowish white lyophilized powder in a single-dose vial for reconstitution and further dilution
For injection: 100 mg lyophilized powder in a single-dose vial (3)
5 WARNINGS AND PRECAUTIONS
- Neutropenia: Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Manage through treatment interruption or dose reduction. (2.3, 5.2)
- Left Ventricular Dysfunction: Assess left ventricular ejection fraction (LVEF) prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage through treatment interruption or discontinuation. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF). (2.3, 5.3)
5.1 Interstitial Lung Disease/Pneumonitis
Severe, life-threatening, or fatal interstitial lung disease (ILD), including pneumonitis, can occur in patients treated with ENHERTU monotherapy or ENHERTU in combination with pertuzumab [see Adverse Reactions (6.1)]. A higher incidence of Grade 1 and 2 ILD/pneumonitis has been observed in patients with moderate renal impairment.
Advise patients to immediately report cough, dyspnea, fever, and/or any new or worsening respiratory symptoms. Monitor patients for signs and symptoms of ILD. Promptly investigate evidence of ILD. Evaluate patients with suspected ILD by radiographic imaging. Consider consultation with a pulmonologist. For asymptomatic (Grade 1) ILD, consider corticosteroid treatment (e.g., ≥0.5 mg/kg/day prednisolone or equivalent). Withhold ENHERTU until recovery [see Dosage and Administration (2.3)]. In cases of symptomatic ILD (Grade 2 or greater), promptly initiate systemic corticosteroid treatment (e.g., ≥1 mg/kg/day prednisolone or equivalent) and continue for at least 14 days followed by gradual taper for at least 4 weeks. Permanently discontinue ENHERTU in patients who are diagnosed with symptomatic (Grade 2 or greater) ILD [see Dosage and Administration (2.3)].
HER2-Positive, HER2-Low, and HER2-Ultralow Metastatic Breast Cancer, HER2-Mutant NSCLC, and Solid Tumors (Including IHC 3+) (5.4 mg/kg)
ENHERTU as Monotherapy
In patients treated with ENHERTU 5.4 mg/kg, ILD occurred in 12% of patients. Median time to first onset was 5.5 months (range: 0.9 to 31.5). Fatal outcomes due to ILD and/or pneumonitis occurred in 0.9% of patients treated with ENHERTU.
ENHERTU in Combination with Pertuzumab
In patients treated with ENHERTU 5.4 mg/kg in combination with pertuzumab (N=431), ILD occurred in 12% of patients. Median time to first onset was 8.0 months (range: 0.6 to 33.8). Fatal outcomes due to ILD and/or pneumonitis occurred in 0.5% of patients treated with ENHERTU in combination with pertuzumab.
HER2-Positive Locally Advanced or Metastatic Gastric Cancer (6.4 mg/kg)
In patients with locally advanced or metastatic HER2-positive gastric or GEJ adenocarcinoma treated with ENHERTU 6.4 mg/kg, ILD occurred in 10% of patients. Median time to first onset was 2.8 months (range: 1.2 to 21).
5.2 Neutropenia
Severe neutropenia, including febrile neutropenia, can occur in patients treated with ENHERTU monotherapy or ENHERTU in combination with pertuzumab.
Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Based on the severity of neutropenia, ENHERTU may require dose interruption or reduction [see Dosage and Administration (2.3)].
HER2-Positive, HER2-Low, and HER2-Ultralow Metastatic Breast Cancer, HER2-Mutant NSCLC, and Solid Tumors (Including IHC 3+) (5.4 mg/kg)
ENHERTU as Monotherapy
In patients treated with ENHERTU 5.4 mg/kg, a decrease in neutrophil count was reported in 65% of patients. Nineteen percent had Grade 3 or 4 decreased neutrophil count. Median time to first onset of decreased neutrophil count was 22 days (range: 2 to 939). Febrile neutropenia was reported in 1.2% of patients.
ENHERTU in Combination with Pertuzumab
In patients treated with ENHERTU 5.4 mg/kg in combination with pertuzumab (N=431), decreased neutrophil count occurred in 79% of patients. Median time to first onset was 22 days (range: 5 to 994). Twenty-nine percent had Grades 3 or 4 decreased neutrophil count. Febrile neutropenia was reported in 2.6% of patients.
HER2-Positive Locally Advanced or Metastatic Gastric Cancer (6.4 mg/kg)
In patients with locally advanced or metastatic HER2-positive gastric or GEJ adenocarcinoma treated with ENHERTU 6.4 mg/kg, a decrease in neutrophil count was reported in 72% of patients. Fifty-one percent had Grade 3 or 4 decreased neutrophil count. Median time to first onset of decreased neutrophil count was 16 days (range: 4 to 187). Febrile neutropenia was reported in 4.8% of patients.
5.3 Left Ventricular Dysfunction
Patients treated with ENHERTU may be at increased risk of developing left ventricular dysfunction. Left ventricular ejection fraction (LVEF) decrease has been observed with anti-HER2 therapies, including ENHERTU.
Assess LVEF prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage LVEF decrease through treatment interruption. Permanently discontinue ENHERTU if LVEF of less than 40% or absolute decrease from baseline of greater than 20% is confirmed. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF) [see Dosage and Administration (2.3)].
Treatment with ENHERTU has not been studied in patients with a history of clinically significant cardiac disease or LVEF less than 50% prior to initiation of treatment.
HER2-Positive, HER2-Low, and HER2-Ultralow Metastatic Breast Cancer, HER2-Mutant NSCLC, and Solid Tumors (Including IHC 3+) (5.4 mg/kg)
ENHERTU as Monotherapy
In patients treated with ENHERTU 5.4 mg/kg, LVEF decrease was reported in 4.6% of patients, of which 0.6% were Grade 3 or 4.
ENHERTU in Combination with Pertuzumab
In patients treated with ENHERTU 5.4 mg/kg in combination with pertuzumab (N=431), LVEF decrease was reported in 11% of patients, of which 2.1% were Grade 3 or 4.
HER2-Positive Locally Advanced or Metastatic Gastric Cancer (6.4 mg/kg)
In patients with locally advanced or metastatic HER2-positive gastric or GEJ adenocarcinoma treated with ENHERTU 6.4 mg/kg, no clinical adverse events of heart failure were reported; however, on echocardiography, 8% were found to have asymptomatic Grade 2 decrease in LVEF.
5.4 Embryo-Fetal Toxicity
Based on its mechanism of action, ENHERTU can cause fetal harm when administered to a pregnant woman. In postmarketing reports, use of a HER2-directed antibody during pregnancy resulted in cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death. Based on its mechanism of action, the topoisomerase inhibitor component of ENHERTU, DXd, can also cause embryo-fetal harm when administered to a pregnant woman because it is genotoxic and targets actively dividing cells [see Use in Specific Populations (8.1), Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. Advise patients of the potential risks to a fetus.
Verify the pregnancy status of females of reproductive potential prior to the initiation of ENHERTU. Advise females of reproductive potential to use effective contraception during treatment and for 7 months after the last dose of ENHERTU. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for 4 months after the last dose of ENHERTU [see Use in Specific Populations (8.1, 8.3)].
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1)]
- Neutropenia [see Warnings and Precautions (5.2)]
- Left Ventricular Dysfunction [see Warnings and Precautions (5.3)]
The most common adverse reactions (≥20%), including laboratory abnormalities, in patients with:
- HER2-positive, HER2-low, and HER2-ultralow metastatic breast cancer, HER2-mutant NSCLC, and HER2-positive (including IHC 3+) solid tumors are decreased white blood cell count, nausea, decreased hemoglobin, decreased neutrophil count, decreased lymphocyte count, fatigue, decreased platelet count, increased aspartate aminotransferase, increased alanine aminotransferase, increased blood alkaline phosphatase, vomiting, alopecia, constipation, decreased blood potassium, decreased appetite, diarrhea, and musculoskeletal pain. (6.1)
- HER2-positive metastatic breast cancer in treatment with ENHERTU in combination with pertuzumab are decreased white blood cell count, decreased hemoglobin, decreased neutrophil count, nausea, increased alanine aminotransferase, diarrhea, increased aspartate aminotransferase, decreased lymphocyte count, decreased platelet count, increased blood alkaline phosphatase, decreased blood potassium, fatigue, alopecia, vomiting, upper respiratory tract infection, constipation, decreased appetite, decreased weight, COVID-19, musculoskeletal pain, increased blood bilirubin, and abdominal pain. (6.1)
- HER2-positive gastric cancer are decreased hemoglobin, decreased white blood cell count, decreased neutrophil count, decreased lymphocyte count, decreased platelet count, nausea, decreased appetite, increased aspartate aminotransferase, fatigue, increased blood alkaline phosphatase, increased alanine aminotransferase, diarrhea, decreased blood potassium, vomiting, constipation, increased blood bilirubin, pyrexia, and alopecia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Daiichi Sankyo, Inc. at 1-877-437-7763 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
HER2-Positive, HER2-Low, and HER2-Ultralow Metastatic Breast Cancer, HER2-Mutant NSCLC, and Solid Tumors (Including IHC 3+) (5.4 mg/kg)
The pooled safety population described in WARNINGS and PRECAUTIONS reflects exposure to ENHERTU 5.4 mg/kg intravenously every 3 weeks in 2233 patients in Study DS8201-A-J101 (NCT02564900), DESTINY-Breast01, DESTINY- Breast02, DESTINY-Breast03, DESTINY-Breast04, DESTINY-Breast06, DESTINY-Lung01, DESTINY-Lung02, DESTINY-CRC02, and DESTINY-PanTumor02. Among these patients, 67% were exposed for greater than 6 months and 38% were exposed for greater than 12 months. In this pooled safety population, the most common (≥20%) adverse reactions (including laboratory abnormalities) were decreased white blood cell count (73%), nausea (72%), decreased hemoglobin (67%), decreased neutrophil count (65%), decreased lymphocyte count (60%), fatigue (55%), decreased platelet count (48%), increased aspartate aminotransferase (46%), increased alanine aminotransferase (44%), increased blood alkaline phosphatase (39%), vomiting (38%), alopecia (37%), constipation (32%), decreased blood potassium (32%), decreased appetite (31%), diarrhea (30%), and musculoskeletal pain (24%).
ENHERTU in Combination with Pertuzumab
The pooled safety population described in WARNINGS and PRECAUTIONS reflects exposure to ENHERTU 5.4 mg/kg in combination with pertuzumab intravenously every 3 weeks in 431 patients in DESTINY-Breast07 (n=50), and DESTINY-Breast09 (n=381). Among these patients, 86% were exposed for greater than 6 months and 73% were exposed for greater than 12 months. In this pooled safety population, the most common (≥20%) adverse reactions (including laboratory abnormalities) were decreased white blood cell count (86%), decreased hemoglobin (80%), decreased neutrophil count (79%), nausea (74%), increased alanine aminotransferase (65%), diarrhea (64%), increased aspartate aminotransferase (63%), decreased lymphocyte count (61%), decreased platelet count (55%), increased blood alkaline phosphatase (54%), decreased blood potassium (54%), fatigue (53%), alopecia (48%), vomiting (46%), upper respiratory tract infection (32%), constipation (31%), decreased appetite (31%), decreased weight (28%), musculoskeletal pain (23%), increased blood bilirubin (23%), and abdominal pain (22%).
HER2-Positive Locally Advanced or Metastatic Gastric Cancer (6.4 mg/kg)
The data described in WARNINGS and PRECAUTIONS reflect exposure to ENHERTU 6.4 mg/kg intravenously every 3 weeks in 125 patients in DESTINY-Gastric01.
HER2-Positive Metastatic Breast Cancer
DESTINY-Breast09
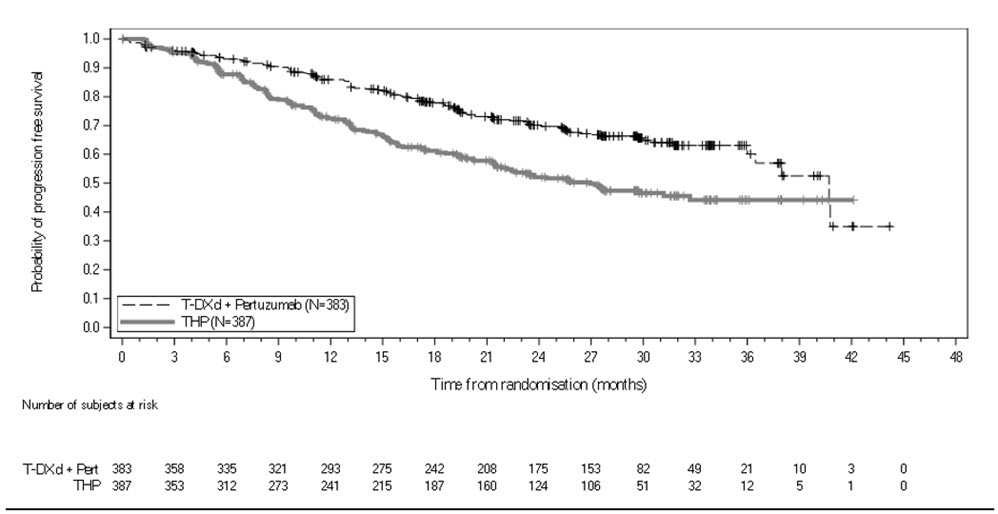
The safety of ENHERTU 5.4 mg/kg in combination with pertuzumab was evaluated in DESTINY-Breast09, a randomized, three-arm, multicenter study including 763 patients with HER2-positive (IHC 3+ or ISH+) unresectable or metastatic breast cancer [see Clinical Studies (14.1)]. Three hundred eighty-one patients received ENHERTU in combination with pertuzumab and 382 patients received THP (taxane [docetaxel or paclitaxel], trastuzumab, and pertuzumab). Among patients who received ENHERTU in combination with pertuzumab, the median duration of treatment was 22 months (range: 0.3 months to 44.5 months).
Serious adverse reactions occurred in 27% of patients receiving ENHERTU in combination with pertuzumab. Serious adverse reactions in >1% of patients were diarrhea, pneumonia, febrile neutropenia, hypokalemia, vomiting, ILD, pulmonary embolism, and sepsis.
Fatalities due to adverse reactions occurred in 3.4% of patients including pneumonia (n=3), ILD (n=2), sepsis (n=2), pulmonary embolism, septic shock, acute kidney injury, dyspnea, febrile neutropenia, and intestinal ischemia (one patient each). ENHERTU was discontinued for adverse reactions in 21% of patients. The most frequent adverse reactions (>2%) associated with permanent discontinuation was ILD/pneumonitis (6.6%).
Dose interruptions due to adverse reactions occurred in 69% of patients. The most frequent adverse reactions (>2%) associated with dose interruption were COVID-19, neutropenia, upper respiratory tract infection, fatigue, anemia, hypokalemia, ILD/pneumonitis, thrombocytopenia, pneumonia, diarrhea, transaminase increased, leukopenia, cough, pyrexia, decreased appetite, and blood bilirubin increased.
Dose reductions occurred in 46% of patients treated with ENHERTU in combination with pertuzumab. The most frequent adverse reactions (>2%) associated with dose reduction were fatigue, neutropenia, nausea, diarrhea, ILD/pneumonitis, thrombocytopenia, vomiting, transaminases increased, decreased weight, febrile neutropenia, and hypokalemia.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were decreased white blood cell count, decreased hemoglobin, decreased neutrophil count, nausea, increased alanine aminotransferase, diarrhea, increased aspartate aminotransferase decreased lymphocyte count, decreased platelet count, increased blood alkaline phosphatase, decreased blood potassium, fatigue, alopecia, vomiting, upper respiratory tract infection, constipation, decreased appetite, decreased weight, COVID-19, musculoskeletal pain, increased blood bilirubin, and abdominal pain.
Tables 4 and 5 summarize common adverse reactions and laboratory abnormalities observed in DESTINY-Breast09.
Other clinically relevant adverse reactions reported in less than 10% of patients treated with ENHERTU in combination with pertuzumab were:
- Blood and Lymphatic System Disorders: febrile neutropenia (2.9%)
- Eye Disorders: blurred vision (4.2%)
- Gastrointestinal Disorders: abdominal distension (6%), gastritis (3.9%), flatulence (2.4%)
- Injury, poisoning, and procedural complications: infusion related reaction (1.8%) [including hypersensitivity and infusion-related reactions
- Investigations: increased blood bilirubin (9%), increased blood creatinine (3.1%)
- Metabolism and Nutrition Disorders: dehydration (2.9%)
- Respiratory, Thoracic, and Mediastinal Disorders: epistaxis (8%), dyspnea (6%)
- Skin and Subcutaneous Tissue Disorders: skin hyperpigmentation (6%) [including skin hyperpigmentation, skin discoloration, and pigmentation disorder]
DESTINY-Breast03
The safety of ENHERTU was evaluated in 257 patients with unresectable or metastatic HER2-positive breast cancer who received at least one dose of ENHERTU 5.4 mg/kg in DESTINY-Breast03 [see Clinical Studies (14.1)]. ENHERTU was administered by intravenous infusion once every three weeks. The median duration of treatment was 14 months (range: 0.7 to 30) for patients who received ENHERTU and 7 months (range: 0.7 to 25) for patients who received ado-trastuzumab emtansine.
Serious adverse reactions occurred in 19% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were vomiting, interstitial lung disease, pneumonia, pyrexia, and urinary tract infection. Fatalities due to adverse reactions occurred in 0.8% of patients including COVID-19 and sudden death (one patient each).
ENHERTU was permanently discontinued in 14% of patients, of which ILD/pneumonitis accounted for 8%. Dose interruptions due to adverse reactions occurred in 44% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose interruption were neutropenia, leukopenia, anemia, thrombocytopenia, pneumonia, nausea, fatigue, and ILD/pneumonitis. Dose reductions occurred in 21% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were nausea, neutropenia, and fatigue.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were nausea, decreased white blood cell count, decreased neutrophil count, increased aspartate aminotransferase, decreased hemoglobin, decreased lymphocyte count, increased alanine aminotransferase, decreased platelet count, fatigue, vomiting, increased blood alkaline phosphatase, alopecia, decreased blood potassium, constipation, musculoskeletal pain, diarrhea, decreased appetite, headache, respiratory infection, abdominal pain, increased blood bilirubin, and stomatitis.
Tables 6 and 7 summarize common adverse reactions and laboratory abnormalities observed in DESTINY-Breast03.
Other clinically relevant adverse reactions reported in less than 10% of patients in the ENHERTU-treated group were:
- Respiratory, Thoracic, and Mediastinal Disorders: dyspnea (8%)
- Skin and Subcutaneous Tissue Disorders: pruritus (8%) and skin hyperpigmentation (6%) [including skin hyperpigmentation, skin discoloration, and pigmentation disorder]
- Nervous System Disorders: dysgeusia (6%)
- Metabolism and Nutrition Disorders: dehydration (4.3%)
- Eye Disorders: blurred vision (3.5%)
- Cardiac Disorders: asymptomatic left ventricular ejection fraction decrease (2.7%) [see Warnings and Precautions (5.3)]
- Injury, Poisoning, and Procedural Complications: infusion-related reactions (2.3%) [including hypersensitivity and infusion-related reactions]
- Blood and Lymphatic System Disorders: febrile neutropenia (0.8%)
DESTINY-Breast02
The safety of ENHERTU was evaluated in 404 patients with unresectable or metastatic HER2-positive breast cancer who received at least one dose of ENHERTU 5.4 mg/kg in DESTINY-Breast02 [see Clinical Studies (14.1)]. ENHERTU was administered by intravenous infusion once every three weeks. The median duration of treatment was 11 months (range: 0.7 to 45) for patients who received ENHERTU.
Serious adverse reactions occurred in 26% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were COVID-19, ILD, pneumonia, vomiting, fatigue, and nausea. Fatalities due to adverse reactions occurred in 2.5% of patients including pneumonitis (2 patients), acute myeloid leukemia, brain edema, COVID-19, hemorrhage, hepatitis B, malignant pleural effusion, pneumonia, and vasogenic cerebral edema (one patient each).
ENHERTU was permanently discontinued in 20% of patients, of which ILD accounted for 9%. Dose interruptions due to adverse reactions occurred in 45% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose interruption were neutropenia, COVID-19, anemia, fatigue, leukopenia, upper respiratory tract infection, and thrombocytopenia. Dose reductions occurred in 25% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were fatigue, nausea, neutropenia, and vomiting.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were nausea, decreased white blood cell count, decreased hemoglobin, decreased neutrophil count, fatigue, decreased lymphocyte count, decreased platelet count, increased alanine aminotransferase, vomiting, increased aspartate aminotransferase, alopecia, increased blood alkaline phosphatase, constipation, decreased appetite, decreased blood potassium, diarrhea, musculoskeletal pain, increased blood bilirubin, abdominal pain, and headache.
Tables 8 and 9 summarize common adverse reactions and laboratory abnormalities observed in DESTINY-Breast02.
Other clinically relevant adverse reactions reported in less than 10% of patients in the ENHERTU-treated group were:
- Respiratory, Thoracic, and Mediastinal Disorders: dyspnea (8%) and epistaxis (8%)
- Skin and Subcutaneous Tissue Disorders: rash (8%) [including rash, pustular rash, maculo-papular rash, and pruritic rash], pruritus (5%), skin hyperpigmentation (5%) [including skin hyperpigmentation and pigmentation disorder]
- Nervous System Disorders: dizziness (8%) and dysgeusia (8%)
- Cardiac Disorders: asymptomatic left ventricular ejection fraction decrease (4.2%) [see Warnings and Precautions (5.3)]
- Eye Disorders: dry eye (6%) and blurred vision [including blurred vision and visual impairment] (3%)
- Metabolism and Nutrition Disorders: dehydration (2.7%)
- Injury, Poisoning, and Procedural Complications: infusion-related reactions (1.2%)
- Blood and Lymphatic System Disorders: febrile neutropenia (0.3%)
DESTINY-Breast01 and Study DS8201-A-J101
The safety of ENHERTU was evaluated in a pooled analysis of 234 patients with unresectable or metastatic HER2-positive breast cancer who received at least one dose of ENHERTU 5.4 mg/kg in DESTINY-Breast01 and Study DS8201-A-J101 (NCT02564900) [see Clinical Studies (14.1)]. ENHERTU was administered by intravenous infusion once every three weeks. The median duration of treatment was 7 months (range: 0.7 to 31).
In the pooled 234 patients, the median age was 56 years (range: 28-96), 74% of patients were <65 years, 99.6% of patients were female, and the majority were White (51%) or Asian (42%). Patients had an Eastern Cooperative Oncology Group (ECOG) performance status of 0 (58%) or 1 (42%) at baseline. Ninety-four percent had visceral disease, 31% had bone metastases, and 13% had brain metastases.
Serious adverse reactions occurred in 20% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were interstitial lung disease, pneumonia, vomiting, nausea, cellulitis, decreased blood potassium, and intestinal obstruction. Fatalities due to adverse reactions occurred in 4.3% of patients including interstitial lung disease (2.6%), and the following events occurred in one patient each (0.4%): acute hepatic failure/acute kidney injury, general physical health deterioration, pneumonia, and hemorrhagic shock.
ENHERTU was permanently discontinued in 9% of patients, of which ILD accounted for 6%.
Dose interruptions due to adverse reactions occurred in 33% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose interruption were neutropenia, anemia, thrombocytopenia, leukopenia, upper respiratory tract infection, fatigue, nausea, and ILD.
Dose reductions occurred in 18% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were fatigue, nausea, and neutropenia.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were nausea, decreased white blood cell count, decreased hemoglobin, decreased neutrophil count, fatigue, vomiting, alopecia, increased aspartate aminotransferase, increased alanine aminotransferase, decreased platelet count, constipation, decreased appetite, diarrhea, decreased blood potassium, and cough.
Tables 10 and 11 summarize common adverse reactions and laboratory abnormalities observed in ENHERTU-treated patients in DESTINY-Breast01 and Study DS8201-A-J101.
Other clinically relevant adverse reactions reported in less than 10% of patients were:
- Injury, Poisoning, and Procedural Complications: infusion-related reactions (2.6%)
- Blood and Lymphatic System Disorders: febrile neutropenia (1.7%)
HER2-Low and HER2-Ultralow Metastatic Breast Cancer
DESTINY-Breast06
The safety of ENHERTU was evaluated in 434 patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer who received ENHERTU 5.4 mg/kg in DESTINY-Breast06 [see Clinical Studies (14.2)]. ENHERTU was administered by intravenous infusion once every three weeks. The median duration of treatment was 11 months (range: 0.4 to 39.6) for patients who received ENHERTU.
Serious adverse reactions occurred in 20% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were interstitial lung disease (ILD)/pneumonitis, COVID-19, febrile neutropenia, and hypokalemia. Fatalities due to adverse reactions occurred in 2.8% of patients including ILD (0.7%); sepsis (0.5%); and COVID-19 pneumonia, bacterial meningoencephalitis, neutropenic sepsis, peritonitis, cerebrovascular accident, general physical health deterioration (0.2% each).
ENHERTU was permanently discontinued in 14% of patients. The most frequent adverse reactions (>2%) associated with permanent discontinuation was ILD/pneumonitis.
Dose interruptions due to adverse reactions occurred in 48% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose interruption were COVID-19, decreased neutrophil count, anemia, pyrexia, pneumonia, decreased white blood cell count, and ILD.
Dose reductions occurred in 25% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were nausea, fatigue, decreased platelet count, and decreased neutrophil count.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were decreased white blood cell count, decreased neutrophil count, nausea, decreased hemoglobin, decreased lymphocyte count, fatigue, decreased platelet count, alopecia, increased alanine aminotransferase, increased blood alkaline phosphatase, increased aspartate aminotransferase, decreased blood potassium, diarrhea, vomiting, constipation, decreased appetite, COVID-19, and musculoskeletal pain.
Tables 12 and 13 summarize common adverse reactions and laboratory abnormalities observed in DESTINY-Breast06.
Other clinically relevant adverse reactions reported in less than 10% of patients in the ENHERTU-treated group were:
- Nervous System Disorders: dizziness (9%)
- Investigations: decreased weight (7%)
- Eye Disorders: dry eye (7%), and blurred vision (5%)
- Respiratory, Thoracic, and Mediastinal Disorders: dyspnea (6%)
- Gastrointestinal Disorders: abdominal distension (4.8%), flatulence (2.3%), and gastritis (0.7%)
- Skin and Subcutaneous Tissue Disorders: pruritus (3.9%), and skin hyperpigmentation (0.9%)
- Metabolism and Nutrition Disorders: dehydration (1.6%)
- Blood and lymphatic system disorders: febrile neutropenia (1.2%)
- Injury, Poisoning, and Procedural Complications: infusion related reaction (1.2%)
DESTINY-Breast04
The safety of ENHERTU was evaluated in 371 patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer who received ENHERTU 5.4 mg/kg in DESTINY-Breast04 [see Clinical Studies (14.2)]. ENHERTU was administered by intravenous infusion once every three weeks. The median duration of treatment was 8 months (range: 0.2 to 33) for patients who received ENHERTU.
Serious adverse reactions occurred in 28% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were ILD/pneumonitis, pneumonia, dyspnea, musculoskeletal pain, sepsis, anemia, febrile neutropenia, hypercalcemia, nausea, pyrexia, and vomiting. Fatalities due to adverse reactions occurred in 4.0% of patients including ILD/pneumonitis (3 patients); sepsis (2 patients); and ischemic colitis, disseminated intravascular coagulation, dyspnea, febrile neutropenia, general physical health deterioration, pleural effusion, and respiratory failure (1 patient each).
ENHERTU was permanently discontinued in 16% of patients, of which ILD/pneumonitis accounted for 8%. Dose interruptions due to adverse reactions occurred in 39% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose interruption were neutropenia, fatigue, anemia, leukopenia, COVID-19, ILD/pneumonitis, increased transaminases, and hyperbilirubinemia. Dose reductions occurred in 23% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were fatigue, nausea, thrombocytopenia, and neutropenia.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were nausea, decreased white blood cell count, decreased hemoglobin, decreased neutrophil count, decreased lymphocyte count, fatigue, decreased platelet count, alopecia, vomiting, increased aspartate aminotransferase, increased alanine aminotransferase, constipation, increased blood alkaline phosphatase, decreased appetite, musculoskeletal pain, diarrhea, and decreased blood potassium.
Tables 14 and 15 summarize common adverse reactions and laboratory abnormalities observed in DESTINY-Breast04.
Other clinically relevant adverse reactions reported in less than 10% of patients treated with ENHERTU:
- Nervous System Disorders: dysgeusia (10%)
- Respiratory, Thoracic and Mediastinal Disorders: cough (10%)
- Gastrointestinal Disorders: abdominal distension (5%), gastritis (2.7%), flatulence (2.4%)
- Eye Disorders: blurred vision (4.9%) [including blurred vision and visual impairment]
- Skin and Subcutaneous Tissue Disorders: pruritus (3.2%) and skin hyperpigmentation (2.7%) [including skin hyperpigmentation, skin discoloration, and pigmentation disorder]
- Metabolism and Nutrition Disorders: dehydration (1.9%)
- Blood and Lymphatic System Disorders: febrile neutropenia (1.1%)
- Injury, Poisoning, and Procedural Complications: infusion-related reactions (0.5%) [including injection-site reaction and chills]
HER2-Mutant Unresectable or Metastatic NSCLC
DESTINY-Lung02 evaluated two dose levels (5.4 mg/kg [n=101] and 6.4 mg/kg [n=50]); however, only the results for the recommended dose of 5.4 mg/kg intravenously every 3 weeks are described below due to increased toxicity observed with the higher dose in patients with NSCLC, including ILD/pneumonitis.
The safety of ENHERTU was evaluated in 101 patients in DESTINY-Lung02 [see Clinical Studies (14.3)]. Patients received ENHERTU 5.4 mg/kg intravenously once every three weeks until disease progression or unacceptable toxicity. Nineteen percent of patients were exposed for greater than 6 months. The median age was 59 years (range 30 to 83); 64% were female; 23% were White, 64% were Asian, and 14% were other races.
Serious adverse reactions occurred in 30% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were ILD/pneumonitis, thrombocytopenia, dyspnea, nausea, pleural effusion, and increased troponin I. Fatality occurred in 1 patient with suspected ILD/pneumonitis (1%).
ENHERTU was permanently discontinued due to an adverse reaction in 8% of patients. Adverse reactions which resulted in permanent discontinuation of ENHERTU were ILD/pneumonitis, diarrhea, decreased blood potassium, hypomagnesemia, myocarditis, and vomiting.
Dose interruptions of ENHERTU due to adverse reactions occurred in 23% of patients. Adverse reactions which required dose interruption (>2%) included neutropenia and ILD/pneumonitis.
Dose reductions due to an adverse reaction occurred in 11% of patients.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were nausea, decreased white blood cell count, decreased hemoglobin, decreased neutrophil count, decreased lymphocyte count, decreased platelet count, decreased albumin, increased aspartate aminotransferase, increased alanine aminotransferase, fatigue, constipation, decreased appetite, vomiting, increased alkaline phosphatase, and alopecia.
Tables 16 and 17 summarize common adverse reactions and laboratory abnormalities observed in DESTINY-Lung02.
Other clinically relevant adverse reactions reported in less than 10% of patients were:
- Respiratory, Thoracic and Mediastinal Disorders: interstitial lung disease (6%) [including interstitial lung disease that was adjudicated as drug-induced ILD including pneumonitis, interstitial lung disease, pulmonary toxicity, and respiratory failure], dyspnea (5%), and epistaxis (3%)
- Gastrointestinal Disorders: abdominal pain (9%) [including abdominal discomfort, abdominal pain, and upper abdominal pain]
- Skin and Subcutaneous Disorders: rash (3%) [including rash and maculo-papular rash]
- Infections and Infestations: upper respiratory tract infection (4%) [including upper respiratory tract infection, pharyngitis, and laryngitis]
- Nervous System Disorders: headache (4%) [including headache and migraine]
HER2-Positive Locally Advanced or Metastatic Gastric Cancer
The safety of ENHERTU was evaluated in 187 patients with locally advanced or metastatic HER2-positive gastric or GEJ adenocarcinoma in DESTINY-Gastric01 [see Clinical Studies (14.4)]. Patients intravenously received at least one dose of either ENHERTU (N=125) 6.4 mg/kg once every three weeks or either irinotecan (N=55) 150 mg/m2 biweekly or paclitaxel (N=7) 80 mg/m2 weekly for 3 weeks. The median duration of treatment was 4.6 months (range: 0.7 to 22.3) in the ENHERTU group and 2.8 months (range: 0.5 to 13.1) in the irinotecan/paclitaxel group.
Serious adverse reactions occurred in 44% of patients receiving ENHERTU 6.4 mg/kg. Serious adverse reactions in >2% of patients who received ENHERTU were decreased appetite, ILD, anemia, dehydration, pneumonia, cholestatic jaundice, pyrexia, and tumor hemorrhage. Fatalities due to adverse reactions occurred in 2.4% of patients: disseminated intravascular coagulation, large intestine perforation, and pneumonia occurred in one patient each (0.8%).
ENHERTU was permanently discontinued in 15% of patients, of which ILD accounted for 6%.
Dose interruptions due to adverse reactions occurred in 62% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose interruption were neutropenia, anemia, decreased appetite, leukopenia, fatigue, thrombocytopenia, ILD, pneumonia, lymphopenia, upper respiratory tract infection, diarrhea, and decreased blood potassium.
Dose reductions occurred in 32% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were neutropenia, decreased appetite, fatigue, nausea, and febrile neutropenia.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were decreased hemoglobin, decreased white blood cell count, decreased neutrophil count, decreased lymphocyte count, decreased platelet count, nausea, decreased appetite, increased aspartate aminotransferase, fatigue, increased blood alkaline phosphatase, increased alanine aminotransferase, diarrhea, decreased blood potassium, vomiting, constipation, increased blood bilirubin, pyrexia, and alopecia.
Tables 18 and 19 summarize adverse reactions and laboratory abnormalities observed in patients receiving ENHERTU 6.4 mg/kg in DESTINY-Gastric01.
Other clinically relevant adverse reactions reported in less than 10% of patients were:
- Cardiac Disorders: asymptomatic left ventricular ejection fraction decrease (8%) [see Warnings and Precautions (5.3)]
- Infections and Infestations: pneumonia (6%)
- Injury, Poisoning, and Procedural Complications: infusion-related reactions (1.6%)
HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors
The safety of ENHERTU was evaluated in 347 adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who received ENHERTU 5.4 mg/kg in DESTINY-Breast01, DESTINY-PanTumor02, DESTINY-Lung01, and DESTINY-CRC02 [see Clinical Studies (14.1 and 14.5)]. ENHERTU was administered by intravenous infusion once every three weeks. The median duration of treatment was 8.3 months (range 0.7 to 30.2).
The median age was 60 years (range 23 to 96); 74% were female; 51% were White, 42% were Asian, 2.9% were Black or African American, 3.5% were of Hispanic or Latino ethnicity; and 40% had an ECOG performance status 0 and 41% had an ECOG performance status of 1.
Serious adverse reactions occurred in 34% of patients receiving ENHERTU. Serious adverse reactions in >1% of patients who received ENHERTU were sepsis, pneumonia, vomiting, urinary tract infection, abdominal pain, nausea, pneumonitis, pleural effusion, hemorrhage, COVID-19, fatigue, acute kidney injury, anemia, cellulitis, and dyspnea. Fatalities due to adverse reactions occurred in 6.3% of patients including ILD/pneumonitis (2.3%), cardiac arrest (0.6%), COVID-19 (0.6%), and sepsis (0.6%). The following events occurred in one patient each (0.3%): acute kidney injury, cerebrovascular accident, general physical health deterioration, pneumonia, and hemorrhagic shock.
ENHERTU was permanently discontinued in 15% of patients, of which ILD/pneumonitis accounted for 10%.
Dose interruptions due to adverse reactions occurred in 48% of patients. The most frequent adverse reactions (>2%) associated with dose interruption were decreased neutrophil count, anemia, COVID-19, fatigue, decreased white blood cell count, and ILD/pneumonitis.
Dose reductions occurred in 27% of patients treated with ENHERTU. The most frequent adverse reactions (>2%) associated with dose reduction were fatigue, nausea, decreased neutrophil count, ILD/pneumonitis, and diarrhea.
The most common (≥20%) adverse reactions, including laboratory abnormalities, were decreased white blood cell count, nausea, decreased hemoglobin, decreased neutrophil count, fatigue, decreased lymphocyte count, decreased platelet count, increased aspartate aminotransferase, increased alanine aminotransferase, increased blood alkaline phosphatase, vomiting, decreased appetite, alopecia, diarrhea, decreased blood potassium, constipation, decreased sodium, stomatitis, and upper respiratory tract infection.
Tables 20 and 21 summarize the common adverse reactions and laboratory abnormalities in DESTINY-PanTumor02, DESTINY-Lung01, DESTINY-Breast01, and DESTINY-CRC02.
Other clinically relevant adverse reactions reported in less than 10% of patients were:
- Respiratory, Thoracic, and Mediastinal Disorders: epistaxis (9%)
- Nervous System Disorders: dizziness (9%) [including dizziness, postural dizziness, and vertigo] and dysgeusia (6%)
- Skin and Subcutaneous Disorders: pruritus (5%) and skin hyperpigmentation (4.3%) [including skin hyperpigmentation, skin discoloration, pigmentation disorder]
- Eye Disorders: blurred vision (4%) [including blurred vision, visual impairment]
- Metabolism and Nutrition Disorders: dehydration (3.2%)
- Gastrointestinal Disorders: abdominal distension (2.6%), flatulence (1.7%) and gastritis (0.9%)
- Blood and Lymphatic System Disorders: febrile neutropenia (1.7%)
- Injury, Poisoning, and Procedural Complications: infusion-related reactions (1.4%) [including administration related reaction, anaphylactic reaction, hypersensitivity, infusion-related reaction and infusion-related hypersensitivity reaction]
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on its mechanism of action, ENHERTU can cause fetal harm when administered to a pregnant woman. There are no available data on the use of ENHERTU in pregnant women. In postmarketing reports, use of a HER2-directed antibody during pregnancy resulted in cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death (see Data ). Based on its mechanism of action, the topoisomerase inhibitor component of ENHERTU, DXd, can also cause embryo-fetal harm when administered to a pregnant woman because it is genotoxic and targets actively dividing cells [see Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. Advise patients of the potential risks to a fetus.
There are clinical considerations if ENHERTU is used in pregnant women, or if a patient becomes pregnant within 7 months after the last dose of ENHERTU (see Clinical Considerations ).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Monitor women who received ENHERTU during pregnancy or within 7 months prior to conception for oligohydramnios. If oligohydramnios occurs, perform fetal testing that is appropriate for gestational age and consistent with community standards of care.
Data
Human Data
There are no available data on the use of ENHERTU in pregnant women. In postmarketing reports in pregnant women receiving a HER2-directed antibody, cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death have been reported. These case reports described oligohydramnios in pregnant women who received a HER2-directed antibody either alone or in combination with chemotherapy. In some case reports, amniotic fluid index increased after use of a HER2-directed antibody was stopped.
Animal Data
There were no animal reproductive or developmental toxicity studies conducted with fam-trastuzumab deruxtecan-nxki.
8.2 Lactation
Risk Summary
There is no data regarding the presence of fam-trastuzumab deruxtecan-nxki in human milk, the effects on the breastfed child, or the effects on milk production. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with ENHERTU and for 7 months after the last dose.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiation of ENHERTU.
Contraception
Females
ENHERTU can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with ENHERTU and for 7 months after the last dose.
Males
Because of the potential for genotoxicity, advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for 4 months after the last dose [see Nonclinical Toxicology (13.1)].
Infertility
Based on findings in animal toxicity studies, ENHERTU may impair male reproductive function and fertility [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
Safety and effectiveness of ENHERTU have not been established in pediatric patients.
Animal Data
Juvenile animal studies have not been conducted with fam-trastuzumab deruxtecan-nxki. In a six-week repeat-dose toxicity study in rats, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in incisor tooth toxicity including single cell necrosis in the base area (e.g., ameloblasts, odontoblasts) and degeneration of the enamel at ≥60 mg/kg (approximately ≥9 times the human recommended dose of 5.4 mg/kg based on AUC), abnormal formation or hypoplasia of the dentin, hemorrhage in the sub-enamel organ tissue, and focal lack of the cementum at 197 mg/kg (approximately 19 times the human recommended dose of 5.4 mg/kg based on AUC). Degeneration of the enamel organ, abnormal dentin formation, hemorrhage in the sub-enamel organ tissue, focal lack of the cementum, and root fracture were observed at 197 mg/kg following a 9-week recovery period.
8.5 Geriatric Use
ENHERTU as Monotherapy
Of the 2355 patients with HER2-positive, HER2-low, or HER2-ultralow breast cancer treated with ENHERTU 5.4 mg/kg, 23% were 65 years or older and 5% were 75 years or older. No overall differences in efficacy within clinical studies were observed between patients ≥65 years of age compared to younger patients. There was a higher incidence of Grade 3-4 adverse reactions observed in patients aged 65 years or older (55%) as compared to younger patients (50%).
Of the 101 patients with HER2-mutant unresectable or metastatic NSCLC treated with ENHERTU 5.4 mg/kg, 40% were 65 years or older and 8% were 75 years or older. No overall differences in efficacy or safety were observed between patients ≥65 years of age compared to younger patients.
Of the 125 patients with HER2-positive locally advanced or metastatic gastric or GEJ adenocarcinoma treated with ENHERTU 6.4 mg/kg in DESTINY-Gastric01, 56% were 65 years or older and 14% were 75 years or older. No overall differences in efficacy or safety were observed between patients ≥65 years of age compared to younger patients.
Of the 192 patients with HER2-positive (IHC 3+) unresectable or metastatic solid tumors treated with ENHERTU 5.4 mg/kg in DESTINY-PanTumor02, DESTINY-Lung01 or DESTINY-CRC02, 39% were 65 years or older and 9% were 75 years or older. No overall differences in efficacy or safety were observed between patients ≥65 years of age compared to younger patients.
ENHERTU in Combination with Pertuzumab
In patients with HER2-positive unresectable or metastatic breast cancer treated with ENHERTU 5.4 mg/kg in combination with pertuzumab (N=431), 17% were 65 years or older and 3% were 75 years or older. No overall differences in efficacy or safety were observed between patients ≥65 years of age compared to younger patients.
8.6 Renal Impairment
No dose adjustment of ENHERTU is required in patients with mild (creatinine clearance [CLcr] ≥60 and <90 mL/min) or moderate (CLcr ≥30 and <60 mL/min) renal impairment [see Clinical Pharmacology (12.3)]. A higher incidence of Grade 1 and 2 ILD/pneumonitis has been observed in patients with moderate renal impairment [see Warnings and Precautions (5.1)]. Monitor patients with moderate renal impairment more frequently. The recommended dosage of ENHERTU has not been established for patients with severe renal impairment (CLcr <30 mL/min) [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dose adjustment of ENHERTU is required in patients with mild (total bilirubin ≤ULN and any AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST) or moderate (total bilirubin >1.5 to 3 times ULN and any AST) hepatic impairment. In patients with moderate hepatic impairment, due to potentially increased exposure, closely monitor for increased toxicities related to the topoisomerase inhibitor, DXd [see Dosage and Administration (2.3)]. The recommended dosage of ENHERTU has not been established for patients with severe hepatic impairment (total bilirubin >3 times ULN and any AST) [see Clinical Pharmacology (12.3)].
11 DESCRIPTION
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody and topoisomerase inhibitor conjugate. Fam-trastuzumab deruxtecan-nxki is an antibody-drug conjugate (ADC) composed of three components: 1) a humanized anti-HER2 IgG1 monoclonal antibody (mAb), covalently linked to 2) a topoisomerase inhibitor, via 3) a tetrapeptide-based cleavable linker. Deruxtecan is composed of a protease-cleavable maleimide tetrapeptide linker and the topoisomerase inhibitor, DXd, which is an exatecan derivative.
The antibody is produced in Chinese hamster ovary cells by recombinant DNA technology, and the topoisomerase inhibitor and linker are produced by chemical synthesis. Approximately 8 molecules of deruxtecan are attached to each antibody molecule. Fam-trastuzumab deruxtecan-nxki has the following structure:
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a sterile, white to yellowish white, preservative-free lyophilized powder in single-dose vials. Each vial delivers 100 mg of fam-trastuzumab deruxtecan-nxki, L-histidine (4.45 mg), L-histidine hydrochloride monohydrate (20.2 mg), polysorbate 80 (1.5 mg), and sucrose (450 mg). Following reconstitution with 5 mL of Sterile Water for Injection, USP, the resulting concentration of fam-trastuzumab deruxtecan-nxki is 20 mg/mL with a pH of 5.5. The resulting solution is administered by intravenous infusion following dilution.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody-drug conjugate. The antibody is a humanized anti-HER2 IgG1. The small molecule, DXd, is a topoisomerase I inhibitor attached to the antibody by a cleavable linker. Following binding to HER2 on tumor cells, fam-trastuzumab deruxtecan-nxki undergoes internalization and intracellular linker cleavage by lysosomal enzymes. Upon release, the membrane-permeable DXd causes DNA damage and apoptotic cell death.
12.2 Pharmacodynamics
Exposure-Response Relationships
Exposure relationship for efficacy has not been fully characterized. Higher systemic exposure to fam-trastuzumab deruxtecan-nxki was associated with a higher incidence rate of any grade ILD.
Cardiac Electrophysiology
The administration of multiple doses of ENHERTU 6.4 mg/kg every 3 weeks did not show a large mean effect (i.e., >20 ms) on the QTc interval in an open-label, single-arm study in 51 patients with metastatic HER2-positive cancer.
12.3 Pharmacokinetics
The pharmacokinetics of fam-trastuzumab deruxtecan-nxki was evaluated in patients with cancer. Following a single dose, exposures (Cmax and AUC) of fam-trastuzumab deruxtecan-nxki and released topoisomerase inhibitor (DXd) increased proportionally over a dose range of 3.2 mg/kg to 8 mg/kg (approximately 0.6 to 1.5 times the recommended dose in breast cancer, NSCLC, and HER2-positive (IHC 3+) solid tumors and 0.5 to 1.25 times the recommended dose in gastric cancer).
At the recommended dosage of ENHERTU for patients with metastatic breast cancer, NSCLC, and HER2-positive (IHC 3+) solid tumors, the geometric mean (coefficient of variation [CV]%) Cmax of fam-trastuzumab deruxtecan-nxki and DXd were 132 µg/mL (20%) and 4.7 ng/mL (48%), respectively, and the AUC of fam-trastuzumab deruxtecan-nxki and DXd were 772 µg∙day/mL (27%) and 29 ng∙day/mL (48%), respectively. Accumulation of fam-trastuzumab deruxtecan-nxki was approximately 35% at steady-state (Cycle 3).
At the recommended dosage of ENHERTU for patients with HER2-positive gastric cancer, the geometric mean Cmax,ss of fam-trastuzumab deruxtecan-nxki and DXd were 126 µg/mL (18%) and 5.2 ng/mL (42%), respectively, and the AUCss of fam-trastuzumab deruxtecan-nxki and DXd were 743 µg∙day/mL (26%) and 33 ng∙day/mL (43%), respectively. Accumulation of fam-trastuzumab deruxtecan-nxki was approximately 39% at steady-state (Cycle 3).
Distribution
The estimated volume of distribution of the central compartment (Vc) of fam-trastuzumab deruxtecan-nxki was 2.68 L.
DXd plasma protein binding is approximately 97% and the blood-to-plasma ratio is approximately 0.6, in vitro.
Elimination
The median elimination half-life (t1/2) of fam-trastuzumab deruxtecan-nxki is 5.4-5.7 days. The estimated systemic clearance of fam-trastuzumab deruxtecan-nxki was 0.41 L/day.
The median elimination half-life (t1/2) of DXd is 5.4-6.1 days. The estimated systemic clearance of DXd was 18.3 L/h.
Metabolism
The humanized HER2 IgG1 monoclonal antibody is expected to be degraded into small peptides and amino acids via catabolic pathways in the same manner as endogenous IgG.
In vitro, DXd is primarily metabolized by CYP3A4.
Specific Populations
No clinically significant differences in the pharmacokinetics of fam-trastuzumab deruxtecan-nxki or DXd were observed for age (20-96 years); race (Asian vs Non-Asian), including White, and Black or African American; sex; body weight (27.3-125.4 kg); tumor types; mild hepatic impairment; mild or moderate renal impairment.
The pharmacokinetics of fam-trastuzumab deruxtecan-nxki or DXd in patients with moderate to severe hepatic impairment or severe renal impairment is unknown.
Drug Interaction Studies
Clinical Studies
Effect of CYP3A Inhibitors on DXd: Coadministration of itraconazole, a strong CYP3A inhibitor, with multiple doses of ENHERTU increased steady state AUC0-17 days of fam-trastuzumab deruxtecan-nxki by 11% and DXd by 18%. The impact of these changes is not clinically meaningful.
Effect of OATP Inhibitors on DXd: Coadministration of ritonavir, a dual inhibitor of OATP1B/CYP3A, with multiple doses of ENHERTU increased steady state AUC0-17 days of fam-trastuzumab deruxtecan-nxki by 19% and DXd by 22%. The impact of these changes is not clinically meaningful.
In Vitro Studies
Effects of DXd on CYP Enzymes: DXd does not inhibit CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A nor induce CYP1A2, CYP2B6, or CYP3A.
Effects of DXd on Transporters: At clinically relevant concentrations (steady-state Cmax of ~0.2 μmol/L), DXd has a low potential to inhibit OAT1 (IC50 value of 12.7 μmol/L), OAT3, OCT1, OCT2, OATP1B1 (IC50 value of 14.4 μmol/L), OATP1B3, MATE1, MATE2-K, P-gp, BCRP, or BSEP transporters.
Effects of Other Drugs on DXd: DXd is a substrate of OATP1B1, OATP1B3, MATE2-K, P-gp, MRP1, and BCRP.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of fam-trastuzumab deruxtecan-nxki or of other fam-trastuzumab deruxtecan products.
Among patients who received ENHERTU as a single agent over a 6 to 9 month treatment period in 13 clinical trials, anti-fam-trastuzumab deruxtecan-nxki antibodies developed in 2.2% (49/2,231) of patients who received ENHERTU 5.4 mg/kg every three weeks and in 2.6% (21/793) of patients who received ENHERTU 6.4 mg/kg every three weeks. Among patients who received ENHERTU (5.4 mg/kg every three weeks) in combination with pertuzumab for a median of 16 months in 2 clinical trials, anti-fam-trastuzumab deruxtecan-nxki antibodies developed in 7.7% (33/426) of patients.
These anti-drug antibodies have no clinically significant effect on the pharmacokinetics or safety of fam-trastuzumab deruxtecan-nxki. Because of the low occurrence of anti-drug antibodies, the effect of antibodies on the effectiveness of fam-trastuzumab deruxtecan-nxki products is unknown.
Among patients with anti-drug antibodies, neutralizing antibodies against fam-trastuzumab deruxtecan-nxki were detected in 6% (4/70) of patients who received ENHERTU as a single agent (5.4 mg/kg or 6.4 mg/kg every 3 weeks) and in 18% (6/33) of patients who received ENHERTU (5.4 mg/kg every 3 weeks) in combination with pertuzumab.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with fam-trastuzumab deruxtecan-nxki.
The topoisomerase inhibitor component of fam-trastuzumab deruxtecan-nxki, DXd, was clastogenic in both an in vivo rat bone marrow micronucleus assay and an in vitro Chinese hamster lung chromosome aberration assay and was not mutagenic in an in vitro bacterial reverse mutation assay.
Fertility studies have not been conducted with fam-trastuzumab deruxtecan-nxki. In a six-week repeat-dose toxicity study in rats, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in spermatid retention at 20 mg/kg and 60 mg/kg (approximately 4 and 9 times the human recommended dose of 5.4 mg/kg based on AUC, respectively). Decreased testes and epididymides weights, tubular atrophy/degeneration in testes, and reduced sperm count in epididymides were observed at a dose of 197 mg/kg (19 times the human recommended dose of 5.4 mg/kg based on AUC). In a three-month repeat-dose toxicity study in monkeys, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in decreased numbers of round spermatids in the testes at seminiferous tubule stages V to VI at ≥30 mg/kg (≥7 times the human recommended dose of 5.4 mg/kg based on AUC). Evidence of reversibility was observed in monkeys by the end of a three-month recovery period.
14 CLINICAL STUDIES
14.1 HER2-Positive Metastatic Breast Cancer
DESTINY-Breast09
The efficacy of ENHERTU in combination with pertuzumab was evaluated in DESTINY-Breast09 (NCT04784715), a randomized, three-arm, multicenter, global study that enrolled 1157 adult patients with HER2-positive advanced, or metastatic breast cancer who had not received prior chemotherapy or HER2-targeted therapy or had received neoadjuvant or adjuvant HER2-targeted therapy more than 6 months before the diagnosis of advanced or metastatic disease. A single line of prior endocrine therapy was permitted for advanced or metastatic breast cancer. HER2 expression was confirmed at a central laboratory using PATHWAY anti-HER-2/neu (4B5) Rabbit Monoclonal Primary Antibody and HER2 Dual ISH DNA Probe Cocktail with HER2 positivity defined as HER2 IHC 3+ or ISH positive.
Patients were excluded for a history of ILD/pneumonitis requiring treatment with steroids or ILD/pneumonitis at screening, patients with symptomatic brain metastases, or ECOG performance status >1, and patients with a history of clinically significant cardiac disease.
Patients were randomized 1:1:1 to receive either, ENHERTU 5.4 mg/kg plus pertuzumab (N=383), THP (taxane [docetaxel or paclitaxel], trastuzumab, and pertuzumab) (N=387), or an investigational therapy (N=387) by intravenous infusion every 3 weeks until unacceptable toxicity or disease progression. Patients with hormone receptor (HR) positive disease (N=416) were allowed to receive concurrent endocrine therapy after 6 cycles of ENHERTU or after discontinuation of the taxane in the THP arm; this occurred in 13.5% of patients in the ENHERTU in the combination with pertuzumab arm and in 38.3% of patients in the THP arm. Randomization was stratified by prior treatment status (de novo vs recurrent), hormone receptor status, and PIK3CA mutation status.
The major efficacy outcome was progression-free survival (PFS) as assessed by blinded independent central review (BICR) based on Response Evaluation Criteria in Solid Tumors v1.1. Additional efficacy outcome measures were overall survival (OS) and confirmed objective response rate (ORR) assessed by BICR.
The median age was 54 years (range: 20-88); 82% were <65 years; 100% were female; 50% of the patients were Asian, 37% were White, 2% were American Indian or Alaska Native, and 3% were Black or African American; 14% were Hispanic or Latino; 8% other or race unknown; 52% had de novo disease and 48% had recurrent disease; 53% were HR-positive and 47% HR-negative; and 31% of patients had a PIK3CA mutation.
The trial demonstrated a statistically significant improvement in PFS for patients randomized to ENHERTU in combination with pertuzumab compared to THP. Efficacy results are summarized in Table 22 and Figure 1. At the time of the PFS analysis, OS data was not mature with 126 (16%) of patients who died across both study arms in the overall population.
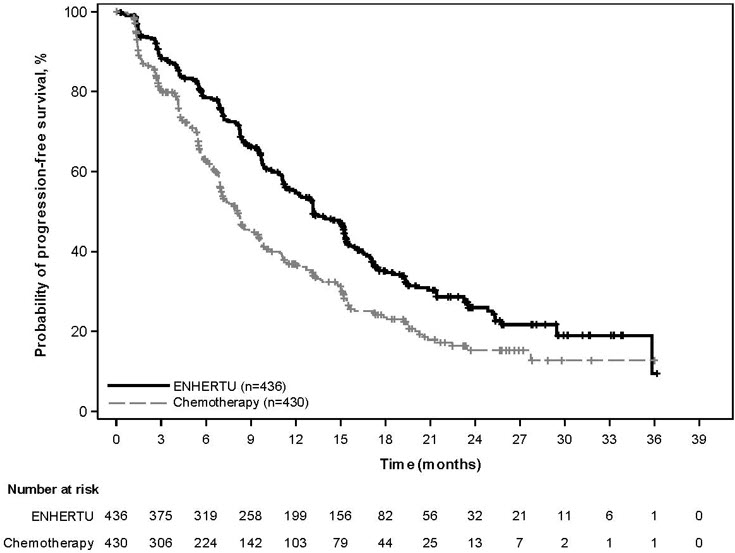
DESTINY-Breast03
The efficacy of ENHERTU was evaluated in study DESTINY-Breast03 (NCT03529110), a multicenter, open-label, randomized trial that enrolled 524 patients with HER2-positive, unresectable and/or metastatic breast cancer who received prior trastuzumab and taxane therapy for metastatic disease or developed disease recurrence during or within 6 months of completing adjuvant therapy. HER2 expression was based on archival tissue tested at a central laboratory prior to enrollment with HER2 positivity defined as HER2 IHC 3+ or ISH positive. Patients were excluded for a history of ILD/pneumonitis requiring treatment with steroids, ILD/pneumonitis at screening, or clinically significant cardiac disease. Patients were also excluded for untreated and symptomatic brain metastases, ECOG performance status >1, or prior treatment with an anti-HER2 antibody-drug conjugate in the metastatic setting.
Patients were randomized 1:1 to receive either ENHERTU 5.4 mg/kg (N=261) or ado-trastuzumab emtansine 3.6 mg/kg (N=263) by intravenous infusion every 3 weeks until unacceptable toxicity or disease progression. Randomization was stratified by hormone receptor status, prior treatment with pertuzumab, and visceral versus non-visceral disease. Tumor imaging was obtained every 6 weeks and CT/MRI of the brain was mandatory for all patients at baseline. The major efficacy outcomes were progression-free survival (PFS) as assessed by blinded independent central review (BICR) based on Response Evaluation Criteria in Solid Tumors (RECIST) v.1.1 and overall survival (OS). Confirmed objective response rate (ORR) was an additional outcome measure.
The median age was 54 years (range: 20-83); 80% were <65 years; 99.6% were female; 60% were Asian, 27% were White, and 3.6% were Black; 11% of patients were of Hispanic/Latino ethnicity. Patients had an ECOG performance status of 0 (63%) or 1 (37%) at baseline. Seventy-three percent had visceral disease, 16% had brain metastases at baseline, 52% were hormone receptor positive (HR+), and 48% of patients had received one line of prior systemic therapy in the metastatic setting. The percentage of patients who had not received prior treatment for metastatic disease was 10%.
Efficacy results are summarized in Table 23 and Figures 2 and 3.
DESTINY-Breast02
The efficacy of ENHERTU was evaluated in study DESTINY-Breast02 (NCT03523585), a multicenter, open-label, randomized study that enrolled 608 patients with HER2-positive, unresectable and/or metastatic breast cancer who were previously treated with ado-trastuzumab emtansine. HER2 expression was based on archival tissue tested at a central laboratory prior to enrollment with HER2 positivity defined as HER2 IHC 3+ or ISH positive. Patients were randomized 2:1 to receive either ENHERTU 5.4 mg/kg (N=406) by intravenous infusion every 3 weeks or treatment of physician's choice (TPC) (N=202, trastuzumab plus capecitabine or lapatinib plus capecitabine) until unacceptable toxicity or disease progression. Randomization was stratified by hormone receptor status, prior treatment with pertuzumab, and visceral versus non-visceral disease. The major efficacy outcomes were PFS as assessed by BICR based on RECIST v1.1 and OS.
The median age was 54 years (range: 22 to 88); 80% were <65 years; 99% were female; 63% were White, 29% were Asian, and 3% were Black or African American; 11% of patients were of Hispanic/Latino ethnicity. Patients had an ECOG performance status of 0 (57%) or 1 (42%) at baseline. Seventy-eight percent had visceral disease, 18% had brain metastases at baseline, 59% were hormone receptor positive (HR+), and 5% of patients had received one line of prior systemic therapy in the metastatic setting.
Efficacy results are summarized in Table 24 and Figures 4 and 5.
DESTINY-Breast01
The efficacy of ENHERTU was evaluated in study DESTINY-Breast01 (NCT03248492), a multicenter, single-arm, trial that enrolled 184 female patients with HER2-positive, unresectable and/or metastatic breast cancer who had received two or more prior anti-HER2 therapies. Patients were excluded for a history of treated ILD or current ILD at screening. Patients were also excluded for history of clinically significant cardiac disease, active brain metastases, and ECOG performance status >1. HER2 expression was based on archival tissue tested at a central laboratory prior to enrollment with HER2 positivity defined as HER2 IHC 3+ or ISH positive.
Patients received ENHERTU 5.4 mg/kg by intravenous infusion every 3 weeks until unacceptable toxicity or disease progression. Tumor imaging was obtained every 6 weeks and CT/MRI of the brain was mandatory for patients with brain metastases at baseline. The major efficacy outcomes were confirmed objective response rate (ORR) assessed by independent central review (ICR) using RECIST v1.1 and duration of response (DOR).
The median age was 55 years (range: 28-96); 76% of patients were <65 years. All 184 patients were female, and the majority were White (55%) or Asian (38%). Patients had an ECOG performance status of 0 (55%) or 1 (44%) at baseline. Ninety-two percent had visceral disease, 29% had bone metastases, and 13% had brain metastases. Fifty-three percent were HR+. Sum of diameters of target lesions were <5 cm in 42%, and ≥5 cm in 50% (not evaluable by central review in 8% of patients).
The median number of prior cancer regimens in the locally advanced/metastatic setting was 5 (range: 2-17). All patients received prior trastuzumab, ado-trastuzumab emtansine, and 66% had prior pertuzumab.
Efficacy results are summarized in Table 25.
14.2 HER2-Low and HER2-Ultralow Metastatic Breast Cancer
DESTINY-Breast06
The efficacy of ENHERTU was evaluated in study DESTINY-Breast06 (NCT04494425), a randomized (1:1), multicenter, open-label study that randomized 866 adult patients with advanced or metastatic HR+ breast cancer with HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) expression as determined by Ventana's PATHWAY anti-HER-2/neu (4B5) Rabbit Monoclonal Primary Antibody assay and evaluated at a central laboratory. HER2-ultralow is defined as membrane staining that is seen in >0 and ≤10% of tumor cells. Patients were eligible if they had disease progression on (a) at least 2 lines of endocrine therapy in the metastatic setting or (b) one line of endocrine therapy in the metastatic setting and demonstrated progression within 24 months of the start of adjuvant endocrine therapy, or within 6 months of starting first line endocrine therapy in combination with a CDK 4/6 inhibitor in the metastatic setting. Patients with prior chemotherapy in the neoadjuvant or adjuvant setting were eligible if they had a disease-free interval greater than 12 months. The study excluded patients with prior chemotherapy for advanced or metastatic disease, patients with a history of ILD/pneumonitis requiring treatment with steroids or ILD/pneumonitis at screening, uncontrolled or significant cardiovascular disease, untreated and symptomatic brain metastases, or ECOG performance status >1.
Patients were randomized to receive either ENHERTU 5.4 mg/kg (N=436) by intravenous infusion every three weeks or physician's choice of single agent chemotherapy (N=430, capecitabine 60%, nab-paclitaxel 24%, or paclitaxel 16%). Randomization was stratified by prior CDK4/6 inhibitor use (yes or no), prior taxane use in the non-metastatic setting (yes or no), and HER2 IHC status of tumor samples (IHC 2+/ISH- vs IHC 1+ vs IHC 0 with membrane staining). Treatment with ENHERTU was administered until disease progression, death, withdrawal of consent, or unacceptable toxicity.
The major efficacy outcome measure was PFS in patients with HER2-low breast cancer assessed by BICR based on RECIST v1.1. Additional efficacy outcome measures were PFS assessed by BICR based on RECIST v1.1 in the overall population, OS in HER2-low patients, and OS in the overall population.
The median age was 57 years (range: 28 to 87); 31% were age 65 or older; 99.9% were female; 53% were White, 35% were Asian, 0.8% were Black or African American, 0.1% were American Indian or Alaska Native, and 7% were of Hispanic/Latino ethnicity. Patients had an ECOG performance status of 0 (59%) or 1 (39%) at baseline; 18% were IHC 0 with membrane staining, 55% were IHC 1+, 27% were IHC 2+/ISH-; 67% had liver metastases, 32% had lung metastases, 8% had brain metastases, and 3% had bone-only metastases. Patients had a median of 2 prior lines of endocrine therapy in the metastatic setting (range: 1 to 5) with 17% having 1 and 68% having 2. Eighty-nine percent of patients had prior endocrine therapy in combination with CDK4/6i treatment in the metastatic setting, 41% had prior taxane use in the non-metastatic setting.
Efficacy results are summarized in Table 26 and Figure 6. At the time of the PFS final analysis, overall survival (OS) data was immature, and a total of 335 (39%) of patients had died across both study arms in the overall population.
In an exploratory analysis of the HER2-ultralow subgroup (n=153), median PFS was 15.1 months (95% CI: 10.0, 17.3) in patients randomized to ENHERTU and 8.3 months (95% CI: 5.8, 15.2) in patients randomized to chemotherapy with a hazard ratio of 0.76 (95% CI: 0.49, 1.17). Confirmed ORR (BICR) was 65.7% (95% CI: 53.1, 76.8) and 30.8% (95% CI: 19.9, 43.4) in patients randomized to ENHERTU and chemotherapy and with measurable disease at baseline, respectively. Median DOR was 14.3 months (95% CI: 11.8, not estimable) and 14.1 months (95% CI: 5.9, not estimable) in patients randomized to ENHERTU and chemotherapy, respectively.
DESTINY-Breast04
The efficacy of ENHERTU was evaluated in study DESTINY-Breast04 (NCT03734029), a randomized, multicenter, open-label study that enrolled 557 adult patients with unresectable or metastatic HER2-low breast cancer. The study included 2 cohorts: 494 hormone receptor-positive (HR+) patients and 63 hormone receptor-negative (HR-) patients. HER2-low expression was defined as IHC 1+ or IHC 2+/ISH-, as determined at a central laboratory using Ventana's PATHWAY anti-HER-2/neu (4B5) Rabbit Monoclonal Primary Antibody assay. Patients must have received chemotherapy in the metastatic setting or have developed disease recurrence during or within 6 months of completing adjuvant chemotherapy. Patients who were HR+ must have received at least one endocrine therapy or be ineligible for endocrine therapy. Patients were randomized 2:1 to receive either ENHERTU 5.4 mg/kg (N=373) by intravenous infusion every 3 weeks or physician's choice of chemotherapy (N=184, eribulin, capecitabine, gemcitabine, nab paclitaxel, or paclitaxel). Randomization was stratified by HER2 IHC status of tumor samples (IHC 1+ or IHC 2+/ISH-), number of prior lines of chemotherapy in the metastatic setting (1 or 2), and HR status/prior CDK4/6i treatment (HR+ with prior CDK4/6 inhibitor treatment, HR+ without prior CDK4/6 inhibitor treatment, or HR-). Treatment was administered until disease progression, death, withdrawal of consent, or unacceptable toxicity. The study excluded patients with a history of ILD/pneumonitis requiring treatment with steroids or ILD/pneumonitis at screening and clinically significant cardiac disease. Patients were also excluded for untreated or symptomatic brain metastases or ECOG performance status >1.
The major efficacy outcome measure was PFS in patients with HR+ breast cancer assessed by BICR based on RECIST v1.1. Additional efficacy outcome measures were PFS assessed by BICR based on RECIST v1.1 in the overall population (all randomized HR+ and HR- patients), OS in HR+ patients, and OS in the overall population.
The median age was 57 years (range: 28 to 81); 24% were age 65 or older; 99.6% were female; 48% were White, 40% were Asian, and 2% were Black or African American; 3.8% of patients were of Hispanic/Latino ethnicity. Patients had an ECOG performance status of 0 (55%) or 1 (45%) at baseline; 58% were IHC 1+, 42% were IHC 2+/ISH-; 70% had liver metastases, 33% had lung metastases, and 6% had brain metastases. In the metastatic setting, patients had a median of 3 prior lines of systemic therapy (range: 1 to 9) with 58% having 1 and 41% having 2 prior chemotherapy regimens; 3.9% were early progressors (progression in the neo/adjuvant setting). In HR+ patients, the median number of prior lines of endocrine therapy was 2 (range: 0 to 9) and 70% had prior CDK4/6i treatment.
Efficacy results are summarized in Table 27 and Figures 7 and 8.
14.3 HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer
ENHERTU was evaluated in DESTINY-Lung01 (NCT03505710) and at two dose levels in DESTINY-Lung02 (NCT04644237). Patients were prospectively selected for treatment with ENHERTU based on the presence of activating HER2 (ERBB2) mutations by local testing using tissue. Samples from DESTINY-Lung01 were retrospectively tested using Oncomine™ Dx Target Test (Life Technologies Corporation, Tissue-test) and Guardant360® CDx test (Guardant Health Inc., Plasma test). Demographic and baseline disease characteristics were similar for patients in DESTINY-Lung01 and DESTINY-Lung02, except for race (34% Asian vs 79% Asian, respectively). Response rates were consistent across dose levels. Increased rates of ILD/pneumonitis were observed at the higher dose. The approved recommended dose of 5.4 mg/kg intravenously every 3 weeks in the DESTINY-Lung02 study is described below [see Adverse Reactions (6.1)].
The efficacy of ENHERTU was evaluated in DESTINY-Lung02, a multicenter, multicohort, randomized, blinded, dose-optimization trial. Eligible patients were required to have unresectable or metastatic HER2-mutant non-squamous NSCLC with disease progression after one prior systemic therapy. Patients with a history of steroid dependent ILD/pneumonitis, clinically significant cardiac disease, clinically active brain metastases, and ECOG performance status >1 were excluded. Patients received ENHERTU 5.4 mg/kg by intravenous infusion every 3 weeks until disease progression or unacceptable toxicity. Tumor imaging was obtained every 6 weeks and CT/MRI of the brain was mandatory for patients with stable brain metastases at baseline.
Results from an interim efficacy analysis in a pre-specified patient cohort are described below. The major efficacy outcomes were confirmed ORR as assessed by BICR using RECIST v1.1 and DOR.
The median age was 58 years (range 30 to 78); 69% were female; 79% were Asian, 12% were White, and 10% were other races; 29% had an ECOG performance status of 0 and 71% had 1; 33% had stable brain metastases; 94% had a mutation in the ERBB2 kinase domain and 6% had a mutation in the extracellular domain. The median number of prior regimens was 2 (range: 1 to 12); 100% of patients received prior platinum therapy, 71% received prior immunotherapy, and 44% received both in combination. Fifty percent of patients were never-smokers and 50% were former smokers; 96% of patients had adenocarcinoma histology.
Efficacy results are provided in Table 28.
14.4 HER2-Positive Locally Advanced or Metastatic Gastric Cancer
The efficacy of ENHERTU was evaluated in study DESTINY-Gastric01 (NCT03329690), a multicenter, open-label, randomized trial conducted in Japan and South Korea that enrolled 188 adult patients with HER2-positive (IHC 3+ or IHC 2+/ISH positive), locally advanced or metastatic gastric or GEJ adenocarcinoma who had progressed on at least two prior regimens including trastuzumab, a fluoropyrimidine- and a platinum-containing chemotherapy. HER2 expression was determined by a central lab on tissue obtained either before or after prior trastuzumab treatment. Patients were excluded for a history of treated or current ILD, a history of clinically significant cardiac disease, active brain metastases, or ECOG performance status >1.
Patients were randomized 2:1 to receive ENHERTU (N=126) 6.4 mg/kg intravenously every 3 weeks or physician's choice of chemotherapy: irinotecan monotherapy (N=55) 150 mg/m2 intravenously every 2 weeks or paclitaxel monotherapy (N=7) 80 mg/m2 intravenously weekly. Randomization was stratified by HER2 status (IHC 3+ or IHC 2+/ISH+), ECOG performance status (0 or 1), and region (Japan or South Korea). Tumor imaging assessments were performed at screening and every 6 weeks from the first treatment dose. Treatment was administered until unacceptable toxicity or disease progression. The major efficacy outcomes were ORR assessed by ICR according to RECIST v1.1 and OS in the intent-to-treat population. Additional efficacy outcomes were PFS and DOR.
The median age was 66 years (range 28 to 82); 76% were male; and 100% were Asian. All patients received a trastuzumab product. Patients had an ECOG performance status of either 0 (49%) or 1 (51%); 87% had gastric adenocarcinoma and 13% had GEJ adenocarcinoma; 76% were IHC 3+ and 23% were IHC 2+/ISH+; 65% had inoperable advanced cancer; 35% had postoperative recurrent cancer; 54% had liver metastases; 29% had lung metastases; 45% had three or more prior regimens in the locally advanced or metastatic setting. A total of 30% of patients were identified as HER2-positive using tissue obtained following prior treatment with a trastuzumab product.
Efficacy results are summarized in Table 29, and the Kaplan-Meier curve for OS is shown in Figure 9.
14.5 HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors
The efficacy of ENHERTU was evaluated in 192 adult patients with previously treated unresectable or metastatic HER2-positive (IHC 3+) solid tumors who were enrolled in one of three multicenter trials: DESTINY-PanTumor02, DESTINY-Lung01, and DESTINY-CRC02. All three studies excluded patients with a history of ILD/pneumonitis requiring treatment with steroids or ILD/pneumonitis at screening and clinically significant cardiac disease. Patients were also excluded for active brain metastases or ECOG performance status >1. Patients received ENHERTU 5.4 mg/kg by intravenous infusion every three weeks. Treatment was administered until disease progression, death, withdrawal of consent, or unacceptable toxicity. The major efficacy outcome measure in all three of the studies was confirmed objective response rate (ORR) and an additional efficacy outcome measure was duration of response (DOR). All outcomes were assessed by independent central review (ICR) based on RECIST v1.1.
DESTINY-PanTumor02
DESTINY-PanTumor02 (NCT04482309) was a multicenter, multicohort, open-label trial that included 111 adult patients with locally advanced, unresectable, or metastatic HER2-positive (IHC 3+ by either local or central assessment) solid tumors that progressed following at least one prior systemic regimen in the advanced/metastatic setting or that had no satisfactory alternative treatment option.
The median age was 64 years (range 23 to 85); 59% were female; 58% were White, 34% were Asian, and 4% were Black or African American; 3% of patients were of Hispanic/Latino ethnicity. Patients had an ECOG performance status of either 0 (49%) or 1 (51%) at baseline. The median number of prior regimens in any treatment setting was 2.
DESTINY-Lung01
DESTINY-Lung01 (NCT03505710) was a multicenter, open-label, 2-cohort trial that included 17 patients with previously treated, unresectable, or metastatic, centrally confirmed HER2-positive (IHC 3+) NSCLC. Patients must have relapsed from or be refractory to standard treatment or have no available standard treatment.
The median age was 59 years (range 31 to 74); 59% were male; 65% were White, 18% were Asian, and 12% were Black or African American. Patients had an ECOG performance status of either 0 (12%) or 1 (88%) at baseline. The median number of prior regimens in any treatment setting was 3.
DESTINY-CRC02
DESTINY-CRC02 (NCT04744831) was a multicenter, randomized, 2-arm trial that included 64 patients with previously treated, unresectable or metastatic centrally confirmed HER2-positive (IHC 3+) colorectal cancer (CRC). Unless contraindicated, patients must have received fluoropyrimidine, oxaliplatin and irinotecan. If clinically indicated, patients must have received anti-EGFR treatment, anti-VEGF treatment and anti-PDL1 therapy.
The median age was 58 years (range 25 to 78); 53% were male; 55% were Asian and 41% were White; 1.6% of patients were of Hispanic/Latino ethnicity. Patients had an ECOG performance status of either 0 (58%) or 1 (42%) at baseline. The median number of prior regimens in any treatment setting was 4.
Efficacy results are summarized in Table 30 and Table 31.
15 REFERENCES
- OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied/Storage
ENHERTU (fam-trastuzumab deruxtecan-nxki) for injection is a white to yellowish white lyophilized powder supplied as:
Store vials in a refrigerator at 2ºC to 8ºC (36ºF to 46ºF) in the original carton to protect from light until time of reconstitution. Do not freeze. Do not shake the reconstituted or diluted solution [see Dosage and Administration (2.4)].
Special Handling
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a hazardous drug. Follow applicable special handling and disposal procedures.1
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Interstitial Lung Disease
- Inform patients of the risks of severe or fatal ILD. Advise patients to contact their healthcare provider immediately for any of the following: cough, shortness of breath, fever, or other new or worsening respiratory symptoms [see Warnings and Precautions (5.1)].
Neutropenia
- Advise patients of the possibility of developing neutropenia and to immediately contact their healthcare provider should they develop a fever, particularly in association with any signs of infection [see Warnings and Precautions (5.2)].
Left Ventricular Dysfunction
- Advise patients to contact their healthcare provider immediately for any of the following: new onset or worsening shortness of breath, cough, fatigue, swelling of ankles/legs, palpitations, sudden weight gain, dizziness, loss of consciousness [see Warnings and Precautions (5.3)].
Embryo-Fetal Toxicity
- Inform female patients of the potential risk to a fetus. Advise female patients to contact their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
- Advise females of reproductive potential to use effective contraception during treatment with ENHERTU and for 7 months after the last dose [see Use in Specific Populations (8.3)].
- Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for 4 months after the last dose [see Use in Specific Populations (8.3)].
Lactation
- Advise women not to breastfeed during treatment and for 7 months after the last dose of ENHERTU [see Use in Specific Populations (8.2)].
Infertility
- Advise males of reproductive potential that ENHERTU may impair fertility [see Use in Specific Populations (8.3)].
Manufactured by:
Daiichi Sankyo, Inc., Basking Ridge, NJ 07920
U.S. License No. 2128
Marketed by:
Daiichi Sankyo, Inc., Basking Ridge, NJ 07920 and AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850
ENHERTU® is a registered trademark of Daiichi Sankyo Company, Ltd.
© 2025 Daiichi Sankyo Co., Ltd.
USPI-ENH-C19.1+DB09-1225-r010
PRINCIPAL DISPLAY PANEL - 100 mg Vial Carton
NDC 65597-406-01
Rx only
ENHERTU®
(fam-trastuzumab deruxtecan-nxki)
For Injection
100 mg per vial
For Intravenous Infusion Only
Dispense the enclosed Medication Guide to each patient.
Reconstitute and Dilute prior to administration
Single-Dose Vial
Discard Unused Portion
Hazardous Drug
KEEP REFRIGERATED
1 vial
Daiichi-Sankyo
AstraZeneca
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.