CALQUENCE (Acalabrutinib) capsule, gelatin coated
1 INDICATIONS AND USAGE
CALQUENCE is a kinase inhibitor indicated:
-
In combination with bendamustine and rituximab for the treatment of adult patients with previously untreated mantle cell lymphoma (MCL) who are ineligible for autologous hematopoietic stem cell transplantation (HSCT). (1.1) -
For the treatment of adult patients with MCL who have received at least one prior therapy. (1.2) -
For the treatment of adult patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL). (1.3)
1.1 Previously Untreated Mantle Cell Lymphoma
CALQUENCE in combination with bendamustine and rituximab is indicated for the treatment of adult patients with previously untreated mantle cell lymphoma (MCL) who are ineligible for autologous hematopoietic stem cell transplantation (HSCT).
1.2 Previously Treated Mantle Cell Lymphoma
CALQUENCE is indicated for the treatment of adult patients with MCL who have received at least one prior therapy.
1.3 Chronic Lymphocytic Leukemia or Small Lymphocytic Lymphoma
CALQUENCE is indicated for the treatment of adult patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL).
2 DOSAGE AND ADMINISTRATION
Recommended dose is 100 mg orally approximately every 12 hours; swallow whole with water and with or without food. (2.1)
2.1 Recommended Dosage
CALQUENCE Administration Instructions
Advise patients to swallow capsule whole with water. Advise patients not to open, break or chew the capsules. CALQUENCE may be taken with or without food. If a dose of CALQUENCE is missed by more than 3 hours, it should be skipped and the next dose should be taken at its regularly scheduled time. Extra capsules of CALQUENCE should not be taken to make up for a missed dose.
CALQUENCE as Monotherapy
For patients with MCL, CLL, or SLL, the recommended dosage of CALQUENCE is 100 mg taken orally approximately every 12 hours until disease progression or unacceptable toxicity.
CALQUENCE in Combination with Bendamustine and Rituximab
For patients with previously untreated MCL, the recommended dosage of CALQUENCE is 100 mg taken orally approximately every 12 hours until disease progression or unacceptable toxicity.
Start CALQUENCE on Day 1 of Cycle 1 (each cycle is 28 days) and administer until disease progression or unacceptable toxicity. Administer bendamustine 90 mg/m2 on Days 1 and 2 and rituximab 375 mg/m2 on Day 1 of Cycle 1 and continue for a total of 6 cycles. Patients achieving a response (PR or CR) after the first 6 cycles may receive maintenance rituximab on Day 1 of every other cycle for a maximum of 12 additional doses, starting on Cycle 8 up to Cycle 30 [see Clinical Studies (14.1)].
CALQUENCE in Combination with Obinutuzumab
For patients with previously untreated CLL or SLL, the recommended dosage of CALQUENCE is 100 mg taken orally approximately every 12 hours until disease progression or unacceptable toxicity. Start CALQUENCE at Cycle 1 (each cycle is 28 days). Start obinutuzumab at Cycle 2 for a total of 6 cycles and refer to the obinutuzumab prescribing information for recommended dosing. Administer CALQUENCE prior to obinutuzumab when given on the same day.
CALQUENCE in Combination with Venetoclax
For patients with previously untreated CLL or SLL, the recommended dosage of CALQUENCE is 100 mg taken orally approximately every 12 hours until disease progression, unacceptable toxicity or completion of 14 cycles of treatment. Start CALQUENCE at Cycle 1 (each cycle is 28 days). Start venetoclax at Cycle 3 for total of 12 cycles. Start venetoclax at 20 mg daily for first week of treatment and increase weekly as per dosing schedule for 5-week ramp up (up to 400 mg daily) as described in the venetoclax USPI. Refer to the venetoclax USPI for additional details.
2.2 Recommended Dosage for Drug Interactions
Dosage Modifications for Use with CYP3A Inhibitors or Inducers
These are described in Table 1 [see Drug Interactions (7) ].
Concomitant Use with Gastric Acid Reducing Agents
Proton Pump Inhibitors: Avoid concomitant use [see Drug Interactions (7) ].
H2-Receptor Antagonists: Take CALQUENCE 2 hours before taking a H2-receptor antagonist [see Drug Interactions (7) ].
Antacids: Separate dosing by at least 2 hours [see Drug Interactions (7) ].
2.3 Dose Modifications for Adverse Reactions
Recommended dosage modifications are provided in Table 2 and 3.
Refer to the prescribing information of each of the products used in combination with CALQUENCE for additional information for management of toxicities.
3 DOSAGE FORMS AND STRENGTHS
Capsules:100 mg acalabrutinib size 1 hard gelatine capsules, with a yellow body and blue cap, marked in black ink with ‘ACA 100 mg’.
Capsules: 100 mg. (3)
5 WARNINGS AND PRECAUTIONS
-
Serious and Opportunistic Infections: Monitor for signs and symptoms of infection and treat promptly. (5.1) -
Hemorrhage: Monitor for bleeding and manage appropriately. (5.2) -
Cytopenias: Monitor complete blood counts regularly. (5.3) -
Second Primary Malignancies: Other malignancies have occurred, including skin cancers and other solid tumors. Advise patients to use sun protection. (5.4) -
Cardiac Arrhythmias: Monitor for symptoms of arrhythmias and manage. (5.5) -
Hepatotoxicity, Including Drug-Induced Liver Injury: Monitor hepatic function throughout treatment. (5.6)
5.1 Serious and Opportunistic Infections
Fatal and serious infections, including opportunistic infections, have occurred in patients with hematologic malignancies treated with CALQUENCE.
Serious or Grade 3 or higher infections (bacterial, viral, or fungal) occurred in 29% of 2,055 patients exposed to CALQUENCE in clinical trials, most often due to respiratory tract infections (18% of all patients, including pneumonia in 14%) [see Adverse Reactions (6.1)]. These infections predominantly occurred in the absence of Grade 3 or 4 neutropenia, with neutropenic infection reported in 8% of all patients. Opportunistic infections in recipients of CALQUENCE have included, but are not limited to, hepatitis B virus reactivation, fungal pneumonia, Pneumocystis jiroveci pneumonia, Epstein-Barr virus reactivation, cytomegalovirus, and progressive multifocal leukoencephalopathy (PML). Consider prophylaxis in patients who are at increased risk for opportunistic infections. Monitor patients for signs and symptoms of infection and treat promptly.
In an additional cohort of patients receiving CALQUENCE in combination with venetoclax with obinutuzumab (AVO) (an unapproved regimen for previously untreated CLL/SLL in AMPLIFY), serious or Grade 3 or higher infections occurred in 25% receiving AVO compared to 14% in patients receiving AV. Fatal infections occurred in 6% receiving AVO compared to 3.1% of patients receiving AV, most commonly due to COVID-19. The safety and effectiveness of AVO has not been established in patients with previously untreated CLL/SLL [see Clinical Studies (14.3)].
5.2 Hemorrhage
Fatal and serious hemorrhagic events have occurred in patients treated with CALQUENCE. Major hemorrhage (serious or Grade 3 or higher bleeding or any central nervous system bleeding) occurred in 4.7% of patients, with fatal hemorrhage occurring in 0.1% of 2,055 patients exposed to CALQUENCE in clinical trials. Bleeding events of any grade, excluding bruising and petechiae, occurred in 39% of patients [see Adverse Reactions (6.1)].
Use of antithrombotic agents concomitantly with CALQUENCE may further increase the risk of hemorrhage. In clinical trials, major hemorrhage occurred in 5% of patients taking CALQUENCE without antithrombotic agents and 3.2% of patients taking CALQUENCE with antithrombotic agents. Consider the risks and benefits of antithrombotic agents when co-administered with CALQUENCE. Monitor patients for signs of bleeding.
Consider the benefit-risk of withholding CALQUENCE for 3 to 7 days pre- and post-surgery depending upon the type of surgery and the risk of bleeding.
5.3 Cytopenias
CALQUENCE can cause Grade 3 or 4 cytopenias. Grade 3 or 4 cytopenias included absolute neutrophil count decreased (28%), absolute lymphocyte count decreased (10%), hemoglobin decreased (9%), and platelets decreased (9%) in 1,758 patients treated with CALQUENCE alone and in combination with obinutuzumab or venetoclax; Grade 4 neutropenia developed in 14% [see Adverse Reactions (6.1)].
Monitor complete blood counts regularly during treatment. Interrupt treatment, reduce the dose, or discontinue treatment as warranted [see Dosage and Administration (2.3)].
5.4 Second Primary Malignancies
Second primary malignancies, including skin cancers and other solid tumors, occurred in 16% of 2,055 patients exposed to CALQUENCE in clinical trials [see Adverse Reactions (6.1)]. The most frequent second primary malignancy was non-melanoma skin cancer, reported in 9% of patients, followed by other solid tumors in 8% (including melanoma, lung cancer, gastrointestinal cancers, and genitourinary cancers) and hematologic malignancies (1.1%). Fatal second primary malignancies occurred in 0.8% of patients. Monitor patients for the development of second cancers and advise protection from sun exposure.
5.5 Cardiac Arrhythmias
Fatal and serious cardiac arrhythmias have occurred in patients treated with CALQUENCE. Grade 3 or 4 atrial fibrillation or flutter was reported in 2.2% of 2,055 patients treated with CALQUENCE, with all grades of atrial fibrillation or flutter reported in 7% of all patients [see Adverse Reactions (6.1)]. Grade 3 or higher ventricular arrhythmia events were reported in 0.5% of patients, including fatal cases in 0.3% of all patients. The risk of arrhythmias may be increased in patients with cardiac risk factors, hypertension, previous arrhythmias, and acute infection. Monitor for symptoms of arrhythmia (e.g., palpitations, dizziness, syncope, dyspnea) and manage as appropriate.
5.6 Hepatotoxicity, Including Drug-Induced Liver Injury
Hepatotoxicity, including severe, life-threatening, and potentially fatal cases of drug-induced liver injury (DILI), has occurred in patients treated with Bruton tyrosine kinase inhibitors, including CALQUENCE.
Evaluate bilirubin and transaminases at baseline and throughout treatment with CALQUENCE. For patients who develop abnormal liver tests after CALQUENCE, monitor more frequently for liver test abnormalities and clinical signs and symptoms of hepatic toxicity. If DILI is suspected, withhold CALQUENCE. Upon confirmation of DILI, discontinue CALQUENCE.
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
-
Serious and Opportunistic Infections [see Warnings and Precautions (5.1) ] -
Hemorrhage [see Warnings and Precautions (5.2) ] -
Cytopenias [see Warnings and Precautions (5.3) ] -
Second Primary Malignancies [see Warnings and Precautions (5.4) ] -
Cardiac Arrhythmias [see Warnings and Precautions (5.5) ] -
Hepatotoxicity, including DILI [see Warnings and Precautions (5.6) ]
The most common adverse reactions (≥ 30%), excluding laboratory abnormalities, are upper respiratory tract infection, diarrhea, headache, and musculoskeletal pain. The most common Grade 3 or 4 laboratory abnormalities (≥ 10%) are absolute neutrophil count decreased, uric acid increase, absolute lymphocyte count decreased, and platelets decreased. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact AstraZeneca at 1-800-236-9933 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data in the Warnings and Precautions reflect exposure to CALQUENCE 100 mg approximately every 12 hours in 2,055 patients with hematologic malignancies. Treatment includes CALQUENCE monotherapy in 1,258 patients in 9 trials, and CALQUENCE combinations in 797 patients in 4 trials. Among these recipients of CALQUENCE, 89% were exposed for at least 6 months and 82% were exposed for at least one year. In this pooled safety population, adverse reactions in ≥ 30% of 2,055 patients, excluding laboratory abnormalities, were upper respiratory tract infection (37%), diarrhea (36%), headache (35%), and musculoskeletal pain (32%). The most common Grade 3 or 4 laboratory abnormalities (≥ 10%) were absolute neutrophil count decreased (32%), uric acid increased (27%), absolute lymphocyte count decreased (21%), and platelets decreased (10%).
Previously Untreated Mantle Cell Lymphoma
The safety data described below reflect exposure to CALQUENCE (100 mg approximately every 12 hours, with or without BR) in patients with MCL [see Clinical Studies (14.1)].
ECHO
The safety of CALQUENCE in combination with bendamustine and rituximab (CALQUENCE plus BR) was evaluated in 297 patients with previously untreated MCL in ECHO [see Clinical Studies (14.1)]. The trial enrolled patients with previously untreated MCL, ≥ 65 years of age with no intention for transplant, total bilirubin ≤ 1.5 × ULN, AST or ALT ≤ 2.5 × ULN, and estimated creatinine clearance of > 50 mL/min. Patients received 6 cycles (as 28-day cycles) of CALQUENCE 100 mg orally twice daily (n = 297) or placebo (n = 297) in combination with bendamustine and rituximab. Patients then received CALQUENCE 100 mg orally twice daily or placebo continuously until progressive disease or unacceptable toxicity, with 12 additional dosages of rituximab every other cycle up to Cycle 30.
The median duration of treatment with CALQUENCE was 28.6 months. A total of 171 (57.6%) patients were treated with CALQUENCE for ˃ 24 months and 122 (41.1%) patients were treated for ˃ 36 months.
Serious adverse reactions occurred in 69% of patients who received CALQUENCE plus BR. Serious adverse reactions reported in ≥ 2% of patients were pneumonia (23%; includes COVID-19 pneumonia), COVID-19 (20%; includes COVID-19 pneumonia), pyrexia (6%), second primary malignancy (7%), rash (3.4%), febrile neutropenia (3.4%), atrial fibrillation (3%), sepsis (2.7%), and anemia (2.4%). Fatal adverse reactions that occurred within 30 days of the last study treatment were reported in 12% who received CALQUENCE plus BR including COVID-19 (6%; includes COVID-19 pneumonia), pneumonia (1%), sepsis (0.3%), second primary malignancy (0.7%), and pneumonitis (0.3%).
Adverse reactions led to permanent discontinuation of CALQUENCE in 43%, dosage interruptions in 74%, and dosage reductions in 10% of patients. Adverse reactions that resulted in dosage modification in > 10% included infections, cytopenias, rashes, and gastrointestinal toxicity. Adverse reactions which resulted in permanent discontinuation of CALQUENCE in ≥ 4% of patients included COVID-19 (includes COVID-19 pneumonia) and neutropenia.
Table 5 and Table 6 summarize select adverse reactions and laboratory abnormalities observed in patients treated in ECHO.
Clinically relevant adverse reactions in < 15% of patients receiving CALQUENCE plus BR included bruising, abdominal pain, atrial fibrillation or flutter, and tumor lysis syndrome.
Grade 4 laboratory abnormalities in > 15% of patients treated with CALQUENCE plus BR include absolute lymphocyte count decreased (26%), absolute neutrophil count decreased (36%), and uric acid increased (17%).
Previously Treated Mantle Cell Lymphoma
ACE-LY-004
The safety data described in this section reflect exposure to CALQUENCE (100 mg approximately every 12 hours) in 124 patients with previously treated MCL in Trial LY-004 [see Clinical Studies (14.2)]. The median duration of treatment with CALQUENCE was 16.6 (range: 0.1 to 26.6) months. A total of 91 (73.4%) patients were treated with CALQUENCE for ≥ 6 months and 74 (59.7%) patients were treated for ≥ 1 year.
The most common adverse reactions (≥ 20%) of any grade were anemia, thrombocytopenia, headache, neutropenia, diarrhea, fatigue, myalgia, and bruising. Grade 1 severity for the non-hematologic, most common events were as follows: headache (25%), diarrhea (16%), fatigue (20%), myalgia (15%), and bruising (19%). The most common Grade ≥ 3 non-hematological adverse reaction (reported in at least 2% of patients) was diarrhea.
Dose reductions and discontinuation due to any adverse reaction were reported in 1.6% and 6.5% of patients, respectively.
Tables 7 and 8 present the frequency category of adverse reactions observed in patients with MCL treated with CALQUENCE.
Increases in creatinine to 1.5 to 3 times the upper limit of normal (ULN) occurred in 4.8% of patients.
Chronic Lymphocytic Leukemia
The safety data described below reflect exposure to CALQUENCE (100 mg approximately every 12 hours, with or without obinutuzumab) in 511 patients with CLL from two randomized controlled clinical trials [see Clinical Studies (14.3)].
The most common adverse reactions (≥ 30%) of any grade in patients with CLL were anemia, neutropenia, thrombocytopenia, headache, upper respiratory tract infection, and diarrhea.
ELEVATE-TN
The safety of CALQUENCE plus obinutuzumab (CALQUENCE+G), CALQUENCE monotherapy, and obinutuzumab plus chlorambucil (GClb) was evaluated in a randomized, multicenter, open-label, actively controlled trial in 526 patients with previously untreated CLL [see Clinical Studies (14.3)].
Patients randomized to the CALQUENCE+G arm were treated with CALQUENCE and obinutuzumab in combination for six cycles, then with CALQUENCE as monotherapy until disease progression or unacceptable toxicity. Patients initiated obinutuzumab on Day 1 of Cycle 2, continuing for a total of 6 cycles. Patient randomized to CALQUENCE monotherapy received CALQUENCE approximately every 12 hours until disease progression or unacceptable toxicity. The trial required age ≥ 65 years of age or 18 to < 65 years of age with a total Cumulative Illness Rating Scale (CIRS) > 6 or creatinine clearance of 30 to 69 mL/min, hepatic transaminases ≤ 3 times ULN and total bilirubin ≤ 1.5 times ULN, and allowed patients to receive antithrombotic agents other than warfarin or equivalent vitamin K antagonists.
During randomized treatment, the median duration of exposure to CALQUENCE in the CALQUENCE+G and CALQUENCE monotherapy arms was 27.7 months (range 0.3 to 40 months), with 95% and 92% and 89% and 86% of patients with at least 6 months and 12 months of exposure, respectively. In the obinutuzumab and chlorambucil arm the median number of cycles was 6 with 84% of patients receiving at least 6 cycles of obinutuzumab, 70% of patients received at least 6 cycles of chlorambucil. Eighty-five percent of patients in the CALQUENCE+G arm received at least 6 cycles of obinutuzumab.
In the CALQUENCE+G and CALQUENCE monotherapy arms, fatal adverse reactions that occurred in the absence of disease progression and with onset within 30 days of the last study treatment were reported in 2% for each treatment arm, most often from infection. Serious adverse reactions were reported in 39% of patients in the CALQUENCE+G arm and 32% in the CALQUENCE monotherapy arm, most often due to events of pneumonia (2.8% to 7%).
In the CALQUENCE+G arm, adverse reactions led to treatment discontinuation in 11% of patients and a dose reduction of CALQUENCE in 7% of patients. In the CALQUENCE monotherapy arm, adverse reactions led to discontinuation in 10% and dose reduction in 4% of patients.
Tables 9 and 10 present adverse reactions and laboratory abnormalities identified in the ELEVATE-TN trial.
Other clinically relevant adverse reactions (all grades incidence < 15%) in recipients of CALQUENCE (CALQUENCE in combination with obinutuzumab and monotherapy) included:
-
Neoplasms: second primary malignancy (10%), non-melanoma skin cancer (5%) -
Cardiac disorders: atrial fibrillation or flutter (3.6%), hypertension (5%) -
Infection: herpesvirus infection (6%)
Increases in creatinine to 1.5 to 3 times ULN occurred in 3.9% and 2.8% of patients in the CALQUENCE combination arm and monotherapy arm, respectively.
ASCEND
The safety of CALQUENCE in patients with relapsed or refractory CLL was evaluated in a randomized, open-label study (ASCEND) [see Clinical Studies (14.3)]. The trial enrolled patients with relapsed or refractory CLL after at least one prior therapy and required hepatic transaminases ≤ 2 times ULN, total bilirubin ≤ 1.5 times ULN, and an estimated creatinine clearance ≥ 30 mL/min. The trial excluded patients having an absolute neutrophil count < 500/µL, platelet count < 30,000/µL, prothrombin time or activated partial thromboplastin time > 2 times ULN, significant cardiovascular disease, or a requirement for strong CYP3A inhibitors or inducers. Patients were allowed to receive antithrombotic agents other than warfarin or equivalent vitamin K antagonist.
In ASCEND, 154 patients received CALQUENCE (100 mg approximately every 12 hours until disease progression or unacceptable toxicity), 118 received idelalisib (150 mg approximately every 12 hours until disease progression or unacceptable toxicity) with up to 8 infusions of a rituximab product, and 35 received up to 6 cycles of bendamustine and a rituximab product. The median age overall was 68 years (range: 32-90); 67% were male; 92% were white; and 88% had an ECOG performance status of 0 or 1.
In the CALQUENCE arm, serious adverse reactions occurred in 29% of patients. Serious adverse reactions in > 5% of patients who received CALQUENCE included lower respiratory tract infection (6%). Fatal adverse reactions within 30 days of the last dose of CALQUENCE occurred in 2.6% of patients, including from second primary malignancies and infection.
In recipients of CALQUENCE, permanent discontinuation due to an adverse reaction occurred in 10% of patients, most frequently due to second primary malignancies followed by infection. Adverse reactions led to dosage interruptions of CALQUENCE in 34% of patients, most often due to respiratory tract infections followed by neutropenia, and dose reduction in 3.9% of patients.
Selected adverse reactions are described in Table 11 and non-hematologic laboratory abnormalities are described in Table 12. These tables reflect exposure to CALQUENCE with median duration of 15.7 months with 94% of patients on treatment for greater than 6 months and 86% of patients on treatment for greater than 12 months. The median duration of exposure to idelalisib was 11.5 months with 72% of patients on treatment for greater than 6 months and 48% of patients on treatment for greater than 12 months. Eighty-three percent of patients completed 6 cycles of bendamustine and rituximab product.
Other clinically relevant adverse reactions (all grades incidence < 15%) in recipients of CALQUENCE included:
-
Skin and subcutaneous disorders: bruising (10%), rash (9%) -
Neoplasms: second primary malignancy (12%), non-melanoma skin cancer (6%) -
Musculoskeletal and connective tissue disorders: arthralgia (8%) -
Cardiac disorders: atrial fibrillation or flutter (5%), hypertension (3.2%) -
Infection: herpesvirus infection (4.5%)
Increases in creatinine to 1.5 to 3 times ULN occurred in 1.3% of patients who received CALQUENCE.
AMPLIFY
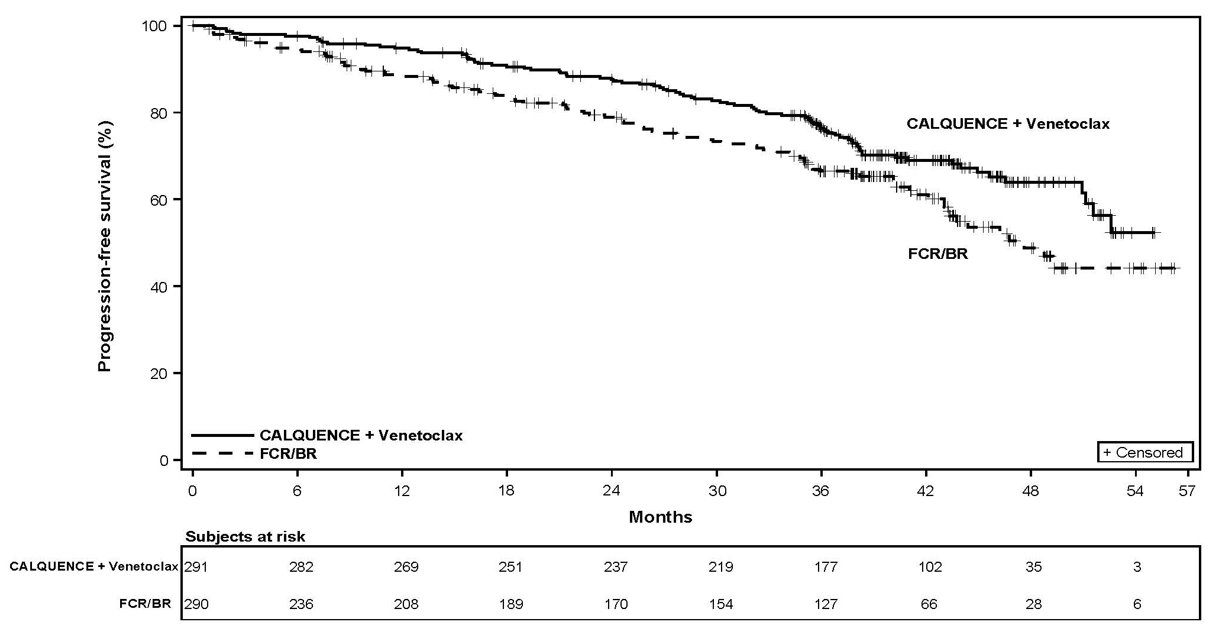
The safety of CALQUENCE in patients with previously untreated CLL was evaluated in a randomized, multicenter, open-label study (AMPLIFY), in which 291 patients received CALQUENCE plus venetoclax (AV), and 259 patients received Investigator’s choice of FCR/BR (fludarabine plus cyclophosphamide plus rituximab or bendamustine plus rituximab) [see Clinical Studies (14.3) ].
Among patients who received AV, 96% were exposed for 6 months or longer and 91% were exposed for greater than one year. The median duration of exposure to CALQUENCE was 12.9 months (range: 1 to 18 months) and to venetoclax was 11.1 months (range: 2 to 14 months).
Serious adverse reactions occurred in 25% of patients receiving AV. The most common serious adverse reactions (≥ 2%) were COVID-19 including COVID-19 pneumonia (9%), second primary malignancies (2.7%), and neutropenia (2.1%). Fatal adverse events occurred in 3.4% of patients. The most common fatal adverse events included COVID-19 and COVID-19 pneumonia.
Treatment discontinuation of CALQUENCE due to adverse reactions occurred in 8% of patients receiving AV. The most common adverse reaction (≥ 2%) leading to treatment discontinuation was COVID-19 pneumonia (2.1%). Dose reduction of CALQUENCE occurred in 6% of patients. Neutropenia was the only adverse reaction leading to dose reduction that occurred in ≥ 1% of patients.
Table 13 and Table 14 summarize select adverse reactions and laboratory abnormalities observed in patients treated in AMPLIFY.
Clinically relevant adverse reactions in < 15% of patients receiving CALQUENCE plus Venetoclax included upper respiratory tract infections, lower respiratory tract infection, arthralgia, pneumonia, hemorrhage, dizziness, constipation, vomiting, second primary malignancy and hypertension.
Grade 4 laboratory abnormalities in > 15% of patients treated with CALQUENCE plus Venetoclax include absolute neutrophil count decreased (15%).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of CALQUENCE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
Cardiac disorders: ventricular arrhythmias -
Hepatobiliary disorders: drug-induced liver injury
7 DRUG INTERACTIONS
-
CYP3A Inhibitors: Avoid co-administration with strong CYP3A inhibitors. Dose adjustments may be recommended. (2.2, 7, 12.3) -
CYP3A Inducers: Avoid co-administration with strong CYP3A inducers. Dose adjustments may be recommended. (2.2, 7, 12.3) -
Gastric Acid Reducing Agents: Avoid co-administration with proton pump inhibitors (PPIs). Stagger dosing with H2-receptor antagonists and antacids. (2.2, 7, 12.3)
7.1 Effect of Other Drugs on CALQUENCE
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on findings in animals, CALQUENCE may cause fetal harm and dystocia when administered to a pregnant woman. There are no available data in pregnant women to inform the drug-associated risk. In animal reproduction studies, administration of acalabrutinib to animals during organogenesis resulted in dystocia in rats and reduced fetal growth in rabbits at maternal exposures (AUC) 2 times exposures in patients at the recommended dose of 100 mg approximately every 12 hours (see Data). Advise pregnant women of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
In a combined fertility and embryo-fetal development study in female rats, acalabrutinib was administered orally at doses up to 200 mg/kg/day starting 14 days prior to mating through gestational day [GD] 17. No effects on embryo-fetal development and survival were observed. The AUC at 200 mg/kg/day in pregnant rats was approximately 9-times the AUC in patients at the recommended dose of 100 mg approximately every 12 hours. The presence of acalabrutinib and its active metabolite were confirmed in fetal rat plasma.
In an embryo-fetal development study in rabbits, pregnant animals were administered acalabrutinib orally at doses up to 200 mg/kg/day during the period of organogenesis (from GD 6-18). Administration of acalabrutinib at doses ≥ 100 mg/kg/day produced maternal toxicity and 100 mg/kg/day resulted in decreased fetal body weights and delayed skeletal ossification. The AUC at 100 mg/kg/day in pregnant rabbits was approximately 2-times the AUC in patients at 100 mg approximately every 12 hours.
In a pre- and postnatal development study in rats, acalabrutinib was administered orally to pregnant animals during organogenesis, parturition and lactation, at doses of 50, 100, and 150 mg/kg/day. Dystocia (prolonged or difficult labor) and mortality of offspring were observed at doses ≥ 100 mg/kg/day. The AUC at 100 mg/kg/day in pregnant rats was approximately 2-times the AUC in patients at 100 mg approximately every 12 hours. Underdeveloped renal papilla was also observed in F1 generation offspring at 150 mg/kg/day with an AUC approximately 5-times the AUC in patients at 100 mg approximately every 12 hours.
8.2 Lactation
Risk Summary
No data are available regarding the presence of acalabrutinib or its active metabolite in human milk, its effects on the breastfed child, or on milk production. Acalabrutinib and its active metabolite were present in the milk of lactating rats. Due to the potential for adverse reactions in a breastfed child from CALQUENCE, advise lactating women not to breastfeed while taking CALQUENCE and for at least 2 weeks after the final dose.
8.3 Females and Males of Reproductive Potential
CALQUENCE may cause embryo-fetal harm and dystocia when administered to pregnant women [see Use in Specific Populations (8.1)].
Pregnancy Testing
Pregnancy testing is recommended for females of reproductive potential prior to initiating CALQUENCE therapy.
Contraception
Females
Advise female patients of reproductive potential to use effective contraception during treatment with CALQUENCE and for at least 1 week following the last dose of CALQUENCE. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be informed of the potential hazard to a fetus.
8.4 Pediatric Use
The safety and efficacy of CALQUENCE in pediatric patients have not been established.
8.5 Geriatric Use
CLL and Previously Treated MCL
Of the 1,758 CALQUENCE-treated patients with B-cell malignancies (excluding previously untreated MCL) in clinical trials, 1,074 (61%) were 65 years of age or older, and 341 (19%) were 75 years of age or older. Among patients 65 years of age or older, 73% had Grade 3 or higher adverse reactions and 55% had serious adverse reactions. Among patients younger than age 65, 58% had Grade 3 or higher adverse reactions and 35% had serious adverse reactions. No clinically relevant differences in efficacy were observed between patients ≥ 65 years and younger.
Of patients that received CALQUENCE in combination with venetoclax in AMPLIFY, 33% (97/291) were ≥ 65 years of age, and 4.5% (13/291) were ≥ 75 years of age. In patients 65 years of age or older and younger than age 65, the fatal adverse reactions were 5% and 2.6% respectively. No clinically relevant differences in efficacy were observed between patients ≥ 65 years of age and younger adults.
Previously Untreated MCL
Of the 297 CALQUENCE-treated patients with previously untreated MCL, 214 (72%) were 65 to 74 years of age and 83 (28%) were 75 years of age and older. No clinically relevant differences in safety or efficacy were observed between patients ages 65 to 74 years and those who were 75 years of age and older.
8.6 Hepatic Impairment
Avoid administration of CALQUENCE in patients with severe hepatic impairment. The safety of CALQUENCE has not been evaluated in patients with moderate or severe hepatic impairment [see Clinical Pharmacology (12.3)].
11 DESCRIPTION
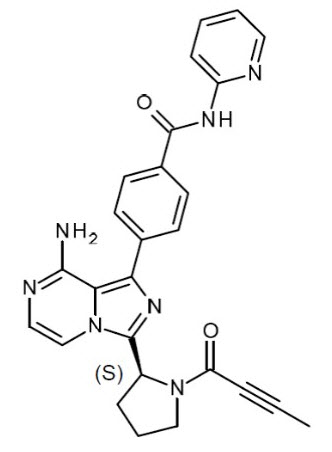
CALQUENCE (acalabrutinib) is an inhibitor of Bruton tyrosine kinase (BTK). The molecular formula for acalabrutinib is C26H23N7O2, and the molecular weight is 465.51. The chemical name is 4-{8-amino-3-[(2S)-1-(but-2-ynoyl)pyrrolidin-2-yl]imidazo[1,5-a]pyrazin-1-yl)}-N-(pyridine-2-yl)benzamide.
The chemical structure of acalabrutinib is shown below:
Acalabrutinib is a white to yellow powder with pH-dependent solubility. It is freely soluble in water at pH values below 3 and practically insoluble at pH values above 6.
CALQUENCE capsules for oral administration contain 100 mg acalabrutinib and the following inactive ingredients: silicified microcrystalline cellulose, partially pregelatinized starch, magnesium stearate, and sodium starch glycolate. The capsule shell contains gelatin, titanium dioxide, yellow iron oxide, FD&C Blue 2 and is imprinted with edible black ink.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Acalabrutinib is a small-molecule inhibitor of BTK. Acalabrutinib and its active metabolite, ACP-5862, form a covalent bond with a cysteine residue in the BTK active site, leading to inhibition of BTK enzymatic activity. BTK is a signaling molecule of the B cell antigen receptor (BCR) and cytokine receptor pathways. In B cells, BTK signaling results in activation of pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion. In nonclinical studies, acalabrutinib inhibited BTK-mediated activation of downstream signaling proteins CD86 and CD69 and inhibited malignant B-cell proliferation and tumor growth in mouse xenograft models.
12.2 Pharmacodynamics
In patients with B-cell malignancies dosed with 100 mg approximately every 12 hours, median steady state BTK occupancy of ≥ 95% in peripheral blood was maintained over 12 hours, resulting in inactivation of BTK throughout the recommended dosing interval.
Cardiac Electrophysiology
The effect of acalabrutinib on the QTc interval was evaluated in a randomized, double-blind, double-dummy, placebo‑ and positive-controlled, 4-way crossover thorough QTc study in 48 healthy adult subjects. Administration of a single dose of acalabrutinib that is the 4-fold maximum recommended single dose did not prolong the QTc interval to any clinically relevant extent (i.e., ≥ 10 ms).
12.3 Pharmacokinetics
Acalabrutinib exhibits dose-proportionality, and both acalabrutinib and its active metabolite, ACP-5862, exposures increase with dose across a dose range of 75 to 250 mg (0.75 to 2.5 times the approved recommended single dose) in patients with B-cell malignancies. At the recommended dose of 100 mg twice daily, the geometric mean (% coefficient of variation [CV]) daily area under the plasma drug concentration over time curve (AUC24h) and maximum plasma concentration (Cmax) for acalabrutinib were 1843 (38%) ngh/mL and 563 (29%) ng/mL, respectively, and for ACP-5862 were 3947 (43%) ngh/mL and 451 (52%) ng/mL, respectively.
Absorption
The geometric mean absolute bioavailability of acalabrutinib was 25%. Median [min, max] time to peak acalabrutinib plasma concentrations (Tmax) was 0.9 [0.5, 1.9] hours, and 1.6 [0.9, 2.7] hour for ACP-5862.
Effect of Food
In healthy subjects, administration of a single 75 mg dose of acalabrutinib (0.75 times the approved recommended single dose) with a high-fat, high-calorie meal (approximately 918 calories, 59 grams carbohydrate, 59 grams fat, and 39 grams protein) did not affect the mean AUC as compared to dosing under fasted conditions. Resulting Cmax decreased by 73% and Tmax was delayed 1-2 hours.
Distribution
Reversible binding to human plasma protein was 97.5% for acalabrutinib and 98.6% for ACP-5862. The in vitro mean blood-to-plasma ratio was 0.8 for acalabrutinib and 0.7 for ACP-5862. The geometric mean (% CV) steady-state volume of distribution (Vss) was approximately 101 (52%) L for acalabrutinib and 67 (32%) L for ACP-5862.
Elimination
The geometric mean (% CV) terminal elimination half-life (t1/2) was 1 (59%) hour for acalabrutinib and 3.5 (24%) hours for ACP-5862. The geometric mean (%CV) apparent oral clearance (CL/F) was 148 (33%) L/hr for acalabrutinib and 19 (23%) L/hr for ACP-5862.
Metabolism
Acalabrutinib is predominantly metabolized by CYP3A enzymes, and to a minor extent, by glutathione conjugation and amide hydrolysis, based on in vitro studies. ACP-5862 was identified as the major active metabolite in plasma with a geometric mean exposure (AUC) that was approximately 2- to 3-fold higher than the exposure of acalabrutinib. ACP-5862 is approximately 50% less potent than acalabrutinib with regard to BTK inhibition.
Excretion
Following administration of a single 100 mg radiolabeled acalabrutinib dose in healthy subjects, 84% of the dose was recovered in the feces and 12% of the dose was recovered in the urine, with less than 2% of the dose excreted as unchanged acalabrutinib in urine and feces.
Specific Populations
Age, Race, and Body Weight
Age (32 to 90 years), sex, race (Caucasian, African American), and body weight (40 to 149 kg) did not have clinically meaningful effects on the PK of acalabrutinib and its active metabolite, ACP-5862.
Renal Impairment
No clinically relevant PK difference was observed in patients with mild or moderate renal impairment (estimated glomerular filtration rate [eGFR] by Modification of Diet in Renal Disease [MDRD] equation: 30 to < 90 mL/min). Acalabrutinib PK has not been evaluated in patients with severe renal impairment (eGFR < 30 mL/min, MDRD) or renal impairment requiring dialysis.
Hepatic Impairment
The AUC of acalabrutinib increased 1.9-fold in subjects with mild hepatic impairment (Child-Pugh class A), 1.5-fold in subjects with moderate hepatic impairment (Child-Pugh class B) and 5.3-fold in subjects with severe hepatic impairment (Child-Pugh class C) compared to subjects with normal liver function. No clinically relevant PK difference in ACP-5862 was observed in subjects with severe hepatic impairment (Child-Pugh Class C) compared to subjects with normal liver function. No clinically relevant PK differences in acalabrutinib and ACP-5862 were observed in patients with mild or moderate hepatic impairment (total bilirubin ≤ 3 x ULN and any AST) relative to patients with normal hepatic function (total bilirubin and AST ≤ ULN).
Drug Interaction Studies
Effect of CYP3A Inhibitors on Acalabrutinib
Co-administration with a strong CYP3A inhibitor (200 mg itraconazole once daily for 5 days) increased the acalabrutinib Cmax by 3.9-fold and AUC by 5.1-fold in healthy subjects.
Physiologically based pharmacokinetic (PBPK) simulations with acalabrutinib and moderate CYP3A inhibitors (erythromycin, fluconazole, diltiazem) showed that co-administration increased acalabrutinib Cmax and AUC approximately 2- to 3-fold.
Effect of CYP3A Inducers on Acalabrutinib
Co-administration with a strong CYP3A inducer (600 mg rifampin once daily for 9 days) decreased acalabrutinib Cmax by 68% and AUC by 77% in healthy subjects.
Gastric Acid Reducing Agents
Acalabrutinib solubility decreases with increasing pH. Co-administration with an antacid (1 g calcium carbonate) decreased acalabrutinib AUC by 53% in healthy subjects. Co-administration with a proton pump inhibitor (40 mg omeprazole for 5 days) decreased acalabrutinib AUC by 43%.
In Vitro Studies
Metabolic Pathways
Acalabrutinib is a weak inhibitor of CYP3A4/5, CYP2C8 and CYP2C9, but does not inhibit CYP1A2, CYP2B6, CYP2C19, CYP2D6, UGT1A1, and UGT2B7. ACP-5862 is a weak inhibitor of CYP2C8, CYP2C9 and CYP2C19, but does not inhibit CYP1A2, CYP2B6, CYP2D6, CYP3A4/5, UGT1A1, and UGT2B7.
Acalabrutinib is a weak inducer of CYP1A2, CYP2B6 and CYP3A4; ACP-5862 weakly induces CYP3A4.
Based on in vitro data and PBPK modeling, no interaction with CYP substrates is expected at clinically relevant concentrations.
Drug Transporter Systems
Acalabrutinib and its active metabolite, ACP-5862, are substrates of P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP). Acalabrutinib is not a substrate of renal uptake transporters OAT1, OAT3, and OCT2, or hepatic transporters OATP1B1, and OATP1B3. ACP-5862 is not a substrate of OATP1B1 or OATP1B3.
Acalabrutinib and ACP-5862 do not inhibit P-gp, OAT1, OAT3, OCT2, OATP1B1, OATP1B3, and MATE2-K at clinically relevant concentrations.
Acalabrutinib may increase exposure to co-administered BCRP substrates (e.g., methotrexate) by inhibition of intestinal BCRP. ACP-5862 does not inhibit BCRP at clinically relevant concentrations. Acalabrutinib does not inhibit MATE1, while ACP-5862 may increase exposure to co-administered MATE1 substrates (e.g., metformin) by inhibition of MATE1.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with acalabrutinib.
Acalabrutinib was not mutagenic in an in vitro bacterial reverse mutation (AMES) assay or clastogenic in an in vitro human lymphocyte chromosomal aberration assay or in an in vivo rat bone marrow micronucleus assay.
In a fertility study in rats, there were no effects of acalabrutinib on fertility in male rats at exposures 11-times, or in female rats at exposures 9-times the AUC observed in patients at the recommended dose of 100 mg twice daily.
14 CLINICAL STUDIES
14.1 Previously Untreated Mantle Cell Lymphoma
ECHO
The efficacy of CALQUENCE in patients with previously untreated MCL was evaluated in a randomized, double-blind, placebo controlled, multicenter study (ECHO; NCT02972840). The study enrolled 598 patients who were ≥ 65 years of age and who had no intention for transplant. The study excluded patients with total bilirubin > 1.5 × upper limit of normal (ULN), AST or ALT > 2.5 × ULN, or estimated creatinine clearance of ≤ 50 mL/min. Patients were randomized in a 1:1 ratio to receive CALQUENCE plus bendamustine and rituximab (CALQUENCE plus BR) or placebo plus BR. Dosing for both arms was administered in 28-day cycles as follows:
-
CALQUENCE plus BR was administered for a maximum of 6 treatment cycles. CALQUENCE 100 mg orally was administered twice daily starting on Cycle 1 Day 1. Bendamustine was administered at 90 mg/m2 intravenously over 30 minutes on Days 1 and 2 of each of 6 cycles. Rituximab was administered at 375 mg/m2 intravenously on Day 1 of each cycle for 6 cycles. -
For patients achieving a response (PR or CR), CALQUENCE 100 mg orally twice daily was administered continuously, in combination with rituximab given at 375 mg/m2 on Day 1 every other cycle for a maximum of 12 additional doses up to Cycle 30. After discontinuation of rituximab, patients continued CALQUENCE monotherapy at 100 mg orally twice daily until disease progression or unacceptable toxicity.
Patients on the control arm received the same regimen but placebo in lieu of CALQUENCE. Crossover to CALQUENCE monotherapy was permitted for patients in the placebo plus BR arm at disease progression.
Of all patients randomized, the median age was 71 years (range: 65-86); 71% were male; 78% were White, 16% Asian, 0.5% were Black or African American. In total, 80% had classic histology of MCL, 7.7% had blastoid MCL, and 5.5% had pleomorphic MCL. The simplified MIPI (Mantle Cell Lymphoma International Prognostic Index) score was low in 33%, intermediate in 43%, and high in 24% of patients. A total of 38% of patients had tumor bulk ≥ 5 cm and 86% had Ann Arbor stage IV disease.
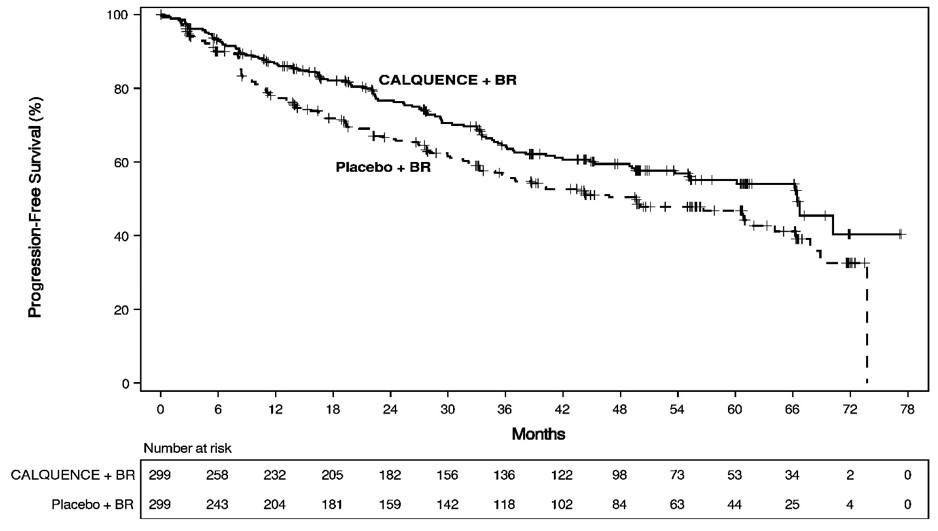
The major efficacy outcome was progression-free survival (PFS) as assessed by an Independent Review Committee (IRC) using the Lugano Classification. Efficacy results are presented in Table 15. The Kaplan-Meier curves for PFS are shown in Figure 1. At this prespecified interim analysis, the median follow-up for PFS was 49.8 months in both arms.
Figure 1. Kaplan-Meier Curve of IRC-Assessed PFS in Patients with Previously Untreated MCL in ECHO
At the time of the PFS analysis, the median overall survival had not been reached in either arm with a total of 203 deaths: 97 (32%) patients in the CALQUENCE plus BR arm and 106 (35%) patients in the placebo plus BR arm had died.
14.2 Previously Treated Mantle Cell Lymphoma
ACE-LY-004
The efficacy of CALQUENCE was based upon Trial LY-004 titled “An Open-label, Phase 2 Study of ACP-196 in Subjects with Mantle Cell Lymphoma” (NCT02213926). Trial LY-004 enrolled a total of 124 patients with MCL who had received at least one prior therapy.
The median age was 68 (range 42 to 90) years, 80% were male, and 74% were Caucasian. At baseline, 93% of patients had an ECOG performance status of 0 or 1. The median time since diagnosis was 46.3 months and the median number of prior treatments was 2 (range 1 to 5), including 18% with prior stem cell transplant. Patients who received prior treatment with BTK inhibitors were excluded. The most common prior regimens were CHOP-based (52%) and ARA-C (34%). At baseline, 37% of patients had at least one tumor with a longest diameter ≥ 5 cm, 73% had extra nodal involvement including 51% with bone marrow involvement. The simplified Mantle Cell Lymphoma International Prognostic Index (MIPI) score (which includes age, ECOG score, and baseline lactate dehydrogenase and white cell count) was intermediate in 44% and high in 17% of patients.
CALQUENCE was administered orally at 100 mg approximately every 12 hours until disease progression or unacceptable toxicity. The median dose intensity was 98.5%. The major efficacy outcome of Trial LY-004 was overall response rate and the median follow-up was 15.2 months.
The median time to best response was 1.9 months.
Lymphocytosis
Upon initiation of CALQUENCE, a temporary increase in lymphocyte counts (defined as absolute lymphocyte count (ALC) increased ≥ 50% from baseline and a post baseline assessment ≥ 5 x 109) in 31.5% of patients in Trial LY-004. The median time to onset of lymphocytosis was 1.1 weeks and the median duration of lymphocytosis was 6.7 weeks.
14.3 Chronic Lymphocytic Leukemia
The efficacy of CALQUENCE in patients with CLL was demonstrated in three randomized, controlled trials. The indication for CALQUENCE includes patients with SLL because it is the same disease.
ELEVATE-TN
The efficacy of CALQUENCE was evaluated in the ELEVATE-TN trial, a randomized, multicenter, open-label, actively controlled, 3 arm trial of CALQUENCE in combination with obinutuzumab, CALQUENCE monotherapy, and obinutuzumab in combination with chlorambucil in 535 patients with previously untreated chronic lymphocytic leukemia (NCT02475681). Patients 65 years of age or older or between 18 and 65 years of age with a total Cumulative Illness Rating Scale (CIRS) > 6 or creatinine clearance of 30 to 69 mL/min were enrolled. The trial also required hepatic transaminases ≤ 3 times upper limit of normal (ULN) and total bilirubin ≤ 1.5 times ULN, and excluded patients with Richter’s transformation.
Patients were randomized in a 1:1:1 ratio into 3 arms to receive:
-
CALQUENCE plus obinutuzumab (CALQUENCE+G): CALQUENCE 100 mg was administered approximately every 12 hours starting on Cycle 1 Day 1 until disease progression or unacceptable toxicity. Obinutuzumab was administered starting on Cycle 2 Day 1 for a maximum of 6 treatment cycles. Obinutuzumab 1000 mg was administered on Days 1 and 2 (100 mg on Day 1 and 900 mg on Day 2), 8 and 15 of Cycle 2 followed by 1000 mg on Day 1 of Cycles 3 up to 7. Each cycle was 28 days. -
CALQUENCE monotherapy: CALQUENCE 100 mg was administered approximately every 12 hours until disease progression or unacceptable toxicity. -
Obinutuzumab plus chlorambucil (GClb): Obinutuzumab and chlorambucil were administered for a maximum of 6 treatment cycles. Obinutuzumab 1000 mg was administered intravenously on Days 1 and 2 (100 mg on Day 1 and 900 mg on Day 2), 8 and 15 of Cycle 1 followed by 1000 mg on Day 1 of Cycles 2 to 6. Chlorambucil 0.5 mg/kg was administered orally on Days 1 and 15 of Cycles 1 to 6. Each cycle was 28 days.
Randomization was stratified by 17p deletion mutation status, ECOG performance status (0 or 1 versus 2), and geographic region. A total of 535 patients were randomized, 179 to CALQUENCE+G, 179 to CALQUENCE monotherapy, and 177 to GClb. The overall median age was 70 years (range: 41 to 91 years), 47% had Rai stage III or IV disease, 14% had 17p deletion or TP53 mutation, 63% of patients had an unmutated IGVH, and 18% had 11q deletion. Baseline demographic and disease characteristics were similar between treatment arms.
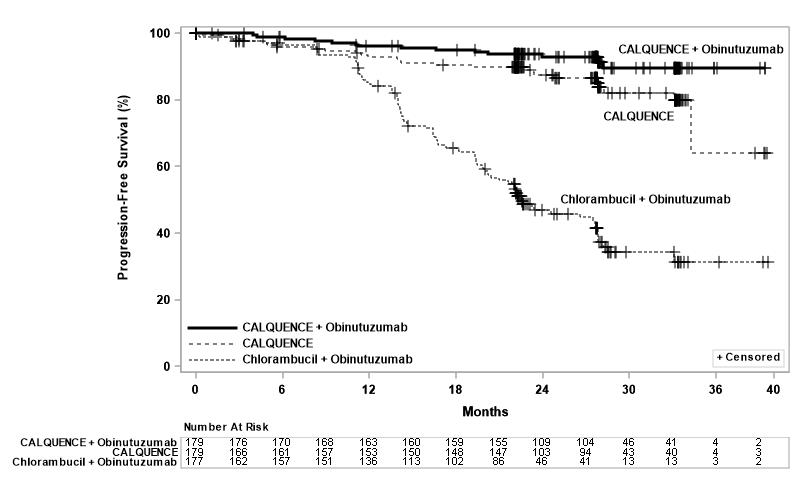
Efficacy was based on progression-free survival (PFS) as assessed by an Independent Review Committee (IRC). The median duration of follow-up was 28.3 months (range: 0.0 to 40.8 months). Efficacy results are presented in Table 17. The Kaplan-Meier curves for PFS are shown in Figure 2.
Figure 2: Kaplan-Meier Curve of IRC-Assessed PFS in Patients with CLL in ELEVATE-TN
With a median follow-up of 28.3 months, median overall survival was not reached in any arm, with fewer than 10% of patients experiencing an event.
ASCEND
The efficacy of CALQUENCE in patients with relapsed or refractory CLL was based upon a multicenter, randomized, open-label trial (ASCEND; NCT02970318). The trial enrolled 310 patients with relapsed or refractory CLL after at least 1 prior systemic therapy. The trial excluded patients with transformed disease, prolymphocytic leukemia, or previous treatment with venetoclax, a Bruton tyrosine kinase inhibitor, or a phosphoinositide-3 kinase inhibitor.
Patients were randomized in a 1:1 ratio to receive either:
-
CALQUENCE 100 mg approximately every 12 hours until disease progression or unacceptable toxicity, or -
Investigator’s choice: -
o Idelalisib plus a rituximab product (IR): Idelalisib 150 mg orally approximately every 12 hours until disease progression or unacceptable toxicity, in combination with 8 infusions of a rituximab product (375 mg/m2 intravenously on Day 1 of Cycle 1, followed by 500 mg/m2 every 2 weeks for 4 doses and then every 4 weeks for 3 doses), with a 28-day cycle length. -
o Bendamustine plus a rituximab product (BR): Bendamustine 70 mg/m2 intravenously (Day 1 and 2 of each 28-day cycle), in combination with a rituximab product (375 mg/m2 intravenously on Day 1 of Cycle 1, then 500 mg/m2 on Day 1 of subsequent cycles), for up to 6 cycles.
-
Randomization was stratified by 17p deletion mutation status, ECOG performance status (0 or 1 versus 2), and number of prior therapies (1 to 3 versus ≥ 4). Of 310 patients total, 155 were assigned to CALQUENCE monotherapy, 119 to IR, and 36 to BR. The median age overall was 67 years (range: 32 to 90 years), 42% had Rai stage III or IV disease, 28% had 17p deletion or TP53 mutation, 78% of patients had an unmutated IGVH, and 27% had a 11q deletion. The CALQUENCE arm had a median of 1 prior therapy (range 1-8), with 47% having at least 2 prior therapies. The investigator’s choice arm had a median of 2 prior therapies (range 1-10), with 57% having at least 2 prior therapies.
In the CALQUENCE arm, the median treatment duration was 15.7 months, with 94% of patients treated for at least 6 months and 86% of patients treated for at least 1 year. In the investigator’s choice arm, the median treatment duration was 8.4 months, with 59% of patients treated for at least 6 months and 37% treated for at least 1 year.
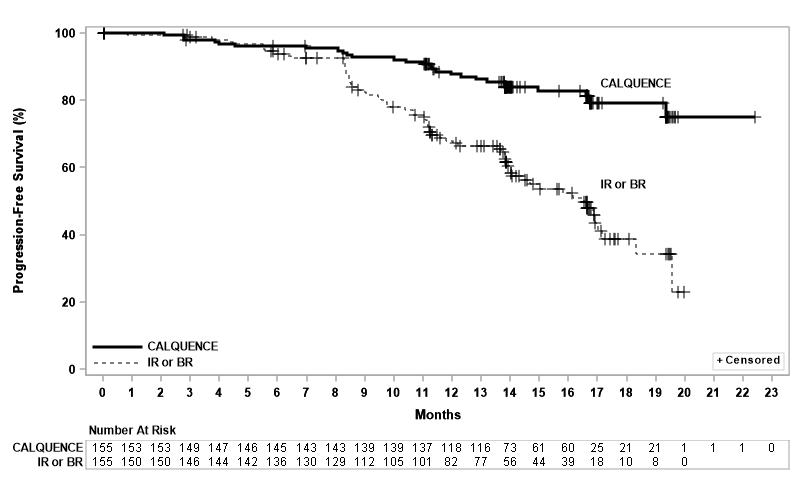
Efficacy was based on PFS as assessed by an IRC, with a median follow-up of 16.1 months (range 0.03 to 22.4 months). Efficacy results are presented in Table 18. The Kaplan-Meier curve for PFS is shown in Figure 3. There was no statistically significant difference in overall response rates between the two treatment arms.
Figure 3: Kaplan-Meier Curve of IRC-Assessed PFS in Patients with CLL in ASCEND
With a median follow up of 16.1 months, median overall survival was not reached in either arm, with fewer than 11% of patients experiencing an event.
Amplify
The efficacy of CALQUENCE in combination with venetoclax in previously untreated CLL patients was evaluated in AMPLIFY, a randomized, multi-center, open-label study (NCT03836261). The study included patients previously untreated for CLL without del(17p) or TP53 mutation that were 18 years of age and older. Patients were randomized to receive:
-
CALQUENCE plus venetoclax (AV): CALQUENCE 100 mg was administered twice daily starting on Cycle 1 Day 1 for a total of 14 cycles or until disease progression or unacceptable toxicity. On Cycle 3 Day 1 patients started the venetoclax 5-week dose-titration schedule, starting at 20 mg and increasing weekly to 50 mg, 100 mg, 200 mg and finally 400 mg once daily. Venetoclax was administered for a total of 12 cycles. Each cycle was 28 days. -
Investigator’s choice of chemoimmunotherapy (FCR/BR): -
o Fludarabine plus cyclophosphamide plus rituximab (FCR): Fludarabine (25 mg/m2 ) and cyclophosphamide (250 mg/m2) were administered on Days 1–3 up to a maximum of 6 cycles. Rituximab was administered at a dose of 375 mg/m2 on Day 1 Cycle 1 and 500 mg/m2 on Day 1 of Cycles 2 up to 6. Each cycle was 28 days. -
o Bendamustine plus rituximab (BR): Bendamustine 90 mg/m2 was administered on Days 1 and 2 up to maximum of 6 cycles. Rituximab was administered at a dose of 375 mg/m2 on Day 1 Cycle 1 and 500 mg/m2 on Day 1 of Cycles 2 up to 6. Each cycle was 28 days.
-
-
An additional investigational combination regimen [see Warnings and Precautions (5.1)].
Patients were stratified by age (> 65 years or ≤ 65), IGHV mutational status (mutated versus unmutated), Rai stage (high risk [≥ 3] versus non-high risk) and geographic region (North America and Europe versus other). In the efficacy population described in Table 19, overall median age was 61 years (range: 26 to 86 years) and 62% were males; 89% were White, 3.8% Asian, 1.7% were Black or African American, 0.3% American Indian or Alaska Native, 0.3% Native Hawaiian or Other Pacific Islander, and 4.8% not reported; 86% were not Hispanic or Latino, 7% Hispanic or Latino, and 7% not reported. The ECOG performance was 0-1 in 90%, bulky diseases with nodes ≥ 5 cm was seen in 41%, 45% had Rai stage III or IV disease, 17% had 11q deletion and 58% had unmutated IGHV.
The major efficacy outcome was IRC-assessed PFS of AV arm versus Investigator’s choice arm (FCR/BR). The median duration of PFS follow-up was 42.6 months. Efficacy results are presented in Table 19. The Kaplan-Meier curve for IRC-assessed PFS is shown in Figure 4.
With a median follow-up of 41.0 months, a total of 60 death events were reported; 18 (6%) in the AV arm and 42 (14%) in the FCR/BR arm.
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Storage
Store at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP Controlled Room Temperature].
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Serious and Opportunistic Infections
Inform patients of the possibility of serious infection and to immediately report signs or symptoms suggestive of infection [see Warnings and Precautions (5.1) ].
Hemorrhage
Inform patients to immediately report signs or symptoms of bleeding. Inform patients that CALQUENCE may need to be interrupted for major surgeries [see Warnings and Precautions (5.2) ].
Cytopenias
Inform patients that they will need periodic blood tests to check blood counts during treatment with CALQUENCE [see Warnings and Precautions (5.3) ].
Second Primary Malignancies
Inform patients that other malignancies have been reported in patients who have been treated with CALQUENCE, including skin cancer and other solid tumors. Advise patients to use sun protection [see Warnings and Precautions (5.4) ].
Cardiac Arrhythmias
Counsel patients to immediately report any signs of palpitations, dizziness, fainting, chest discomfort, and shortness of breath [see Warnings and Precautions (5.5) ].
Hepatotoxicity, Including Drug-Induced Liver Injury:
Inform patients that liver problems, including drug-induced liver injury and abnormalities in liver tests, may develop during CALQUENCE treatment. Advise patients to contact their healthcare provider immediately if they experience abdominal discomfort, dark urine, or jaundice [see Warnings and Precautions (5.6) ].
Pregnancy Complication
CALQUENCE may cause fetal harm and dystocia. Advise women to avoid becoming pregnant during treatment and for at least 1 week after the last dose of CALQUENCE [see Use in Specific Populations (8.3) ].
Lactation
Advise females not to breastfeed during treatment with CALQUENCE and for at least 2 weeks after the final dose [see Use in Specific Populations (8.2) ].
Dosing Instructions
Instruct patients to take CALQUENCE orally twice daily, about 12 hours apart. CALQUENCE may be taken with or without food. Advise patients that CALQUENCE capsules should be swallowed whole with a glass of water, without being opened, broken, or chewed [see Dosage and Administration (2.1) ].
Missed Dose
Advise patients that if they miss a dose of CALQUENCE, they may still take it up to 3 hours after the time they would normally take it. If more than 3 hours have elapsed, they should be instructed to skip that dose and take their next dose of CALQUENCE at the usual time. Warn patients they should not take extra capsules to make up for the dose that they missed [see Dosage and Administration (2.1) ].
Drug Interactions
Advise patients to inform their healthcare providers of all concomitant medications, including over-the-counter medications, vitamins and herbal products [see Drug Interactions (7) ].
Distributed by:
AstraZeneca Pharmaceuticals LP
Wilmington, DE 19850
CALQUENCE is a registered trademark of the AstraZeneca group of companies.
©AstraZeneca 2026
Patient Information
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 2/2026
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
NDC 0310-0512-60 60 capsules
CALQUENCE®
(acalabrutinib) capsules
100 mg
Rx only
Manufactured for:
AstraZeneca Pharmaceuticals LP
Wilmington, DE 19850
By: AstraZeneca AB
SE-151 85 Södertàlje, Sweden
Product of Switzerland
AstraZeneca
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.