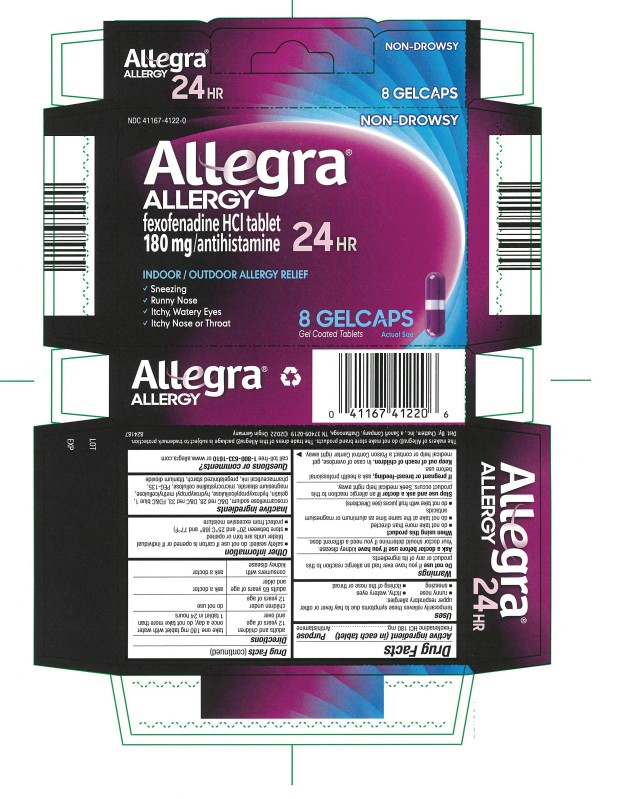
Allegra Allergy (Fexofenadine Hydrochloride) tablet, coated
Allegra Allergy® Gelcaps – 24 HO UR
Drug Facts
Active ingredient
(in each tablet)
Fexofenadine HCI 180 mg
Purpose
Antihistamine
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, water eyes
- itching of the nose or throat
Warnings
Do not use
if you have ever had an allergic reaction to this product or any of its ingredients.
Ask a doctor before use if you have
kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Stop use and ask a doctor if
an allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
Other information
- safety sealed: do not use if carton is opened or if individual blister units are torn or opened
- store between 20º and 25ºC (68º and 77ºF)
- protect from excessive moisture
Inactive ingredients
croscarmellose sodium, D&C red 28, D&C red 33, FD&C blue 1, gelatin, hydroxypropylcellulose, hydroxypropyl methylcellulose, magnesium stearate, microcrystalline cellulose, PEG-135, pharmaceutical ink, pregelatinized starch, titanium dioxide
Questions or comments?
call toll-free 1-800-633-1610 or www.allegra.com
The makers of Allegra® do not make store brand products. The trade dress of this Allegra® package is subject to trademark protection.
Dist. By: Chattem, Inc., a Sanofi Company, Chattanooga, TN 37409-0219 ©2014
PRINCIPAL DISPLAY PANEL
NDC 41167-4122-0
Allegra®
ALLERGY
fexofenadine HCI tablet
180 mg/antihistamine
24 HR
8 GELCAPS
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.