Spikevax (COVID-19 Vaccine, mRNA) injection, suspension
1 INDICATIONS AND USAGE
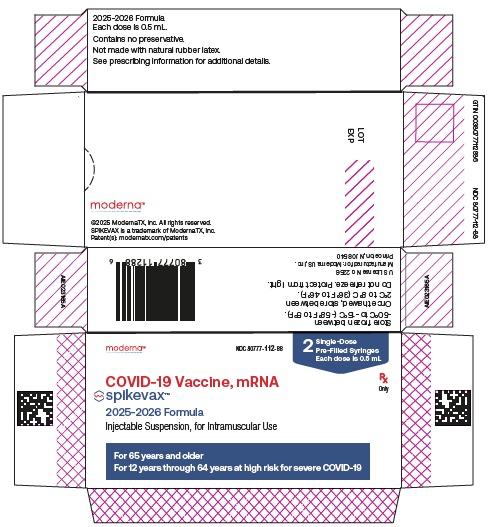
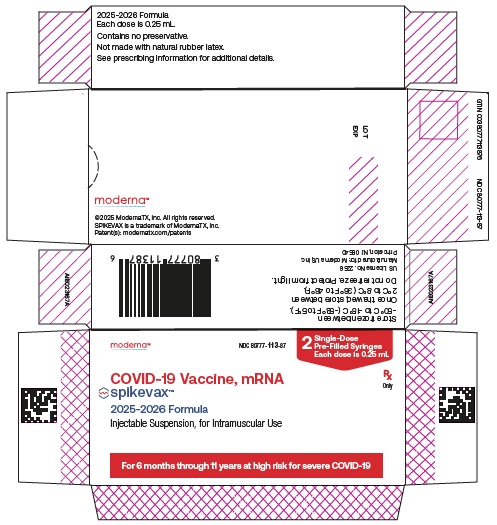
SPIKEVAX is a vaccine indicated for active immunization to prevent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
SPIKEVAX is approved for use in individuals who are:
-
65 years of age and older, or -
6 months through 64 years of age with at least one underlying condition that puts them at high risk for severe outcomes from COVID-19.
SPIKEVAX is a vaccine indicated for active immunization to prevent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
SPIKEVAX is approved for use in individuals who are:
-
65 years of age and older, or -
6 months through 64 years of age with at least one underlying condition that puts them at high risk for severe outcomes from COVID-19. (1)
2 DOSAGE AND ADMINISTRATION
For intramuscular use.
2.1 Preparation for Administration
-
Verify that the label on the pre-filled syringe states 2025-2026 Formula. -
If pre-filled syringes of SPIKEVAX are frozen, thaw before use following the instructions below.
-
After thawing, do not refreeze. -
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. -
SPIKEVAX is a white to off-white suspension. It may contain white or translucent product-related particulates. Do not administer if vaccine is discolored or contains other particulate matter. -
Do not shake. -
With tip cap upright, remove tip cap by twisting counterclockwise until tip cap releases. Remove tip cap in a slow, steady motion. Avoid pulling tip cap while twisting. -
Attach the needle by twisting in a clockwise direction until the needle fits securely on the syringe.
2.2 Administration
Administer SPIKEVAX intramuscularly. Discard after single use.
2.3 Dosing and Schedule
3 DOSAGE FORMS AND STRENGTHS
SPIKEVAX is an injectable suspension.
For individuals 6 months through 11 years of age, a single dose is 0.25 mL.
For individuals 12 years of age and older, a single dose is 0.5 mL.
4 CONTRAINDICATIONS
Do not administer SPIKEVAX to individuals with a known history of severe allergic reaction (e.g., anaphylaxis) to any component of SPIKEVAX [see Description (11)] or to individuals who had a severe allergic reaction (e.g., anaphylaxis) following a previous dose of a Moderna COVID-19 vaccine.
Do not administer SPIKEVAX to individuals with a known history of severe allergic reaction (e.g., anaphylaxis) to any component of SPIKEVAX or to individuals who had a severe allergic reaction (e.g., anaphylaxis) following a previous dose of a Moderna COVID-19 vaccine. (4)
5 WARNINGS AND PRECAUTIONS
Analyses of postmarketing data from use of authorized or approved mRNA COVID-19 vaccines, including SPIKEVAX, have demonstrated increased risks of myocarditis and pericarditis, with onset of symptoms typically in the first week following vaccination. The observed risk has been highest in males 12 years through 24 years of age. (5.2)
5.1 Management of Acute Allergic Reactions
Appropriate medical treatment must be immediately available to manage potential anaphylactic reactions following administration of SPIKEVAX.
5.2 Myocarditis and Pericarditis
Analyses of postmarketing data from use of authorized or approved mRNA COVID-19 vaccines, including SPIKEVAX, have demonstrated increased risks of myocarditis and pericarditis, with onset of symptoms typically in the first week following vaccination. The observed risk has been highest in males 12 years through 24 years of age.
Based on analyses of commercial health insurance claims data from inpatient and outpatient settings, the estimated unadjusted incidence of myocarditis and/or pericarditis during the period 1 through 7 days following administration of the 2023-2024 Formula of mRNA COVID-19 vaccines was approximately 8 cases per million doses in individuals 6 months through 64 years of age and approximately 27 cases per million doses in males 12 years through 24 years of age.
Although some individuals with myocarditis and/or pericarditis following administration of mRNA COVID-19 vaccines have required intensive care support, available data suggest that individuals typically have resolution of symptoms within a few days with conservative management.
Follow-up information on cardiovascular outcomes in hospitalized patients who had been diagnosed with COVID-19 vaccine-associated myocarditis is available from a longitudinal retrospective observational study. Most of these patients had received a two-dose primary series of an mRNA COVID-19 vaccine prior to their diagnosis. In this study, at a median follow-up of approximately 5 months post-vaccination, persistence of abnormal cardiac magnetic resonance imaging (CMR) findings that are a marker for myocardial injury was common. The clinical and prognostic significance of these CMR findings is not known1 [see Adverse Reactions (6.2)].
Information is not yet available about potential long-term sequelae of myocarditis or pericarditis following administration of mRNA COVID-19 vaccines.
The Centers for Disease Control and Prevention (CDC) has published considerations related to myocarditis and pericarditis after vaccination, including for vaccination of individuals with a history of myocarditis or pericarditis (https://www.cdc.gov/vaccines/covid-19/clinical-considerations/myocarditis.html).
5.3 Syncope
Syncope (fainting) may occur in association with administration of injectable vaccines. Procedures should be in place to avoid injury from fainting.
5.4 Altered Immunocompetence
Immunocompromised persons, including individuals receiving immunosuppressive therapy, may have a diminished immune response to SPIKEVAX [see Use in Specific Populations (8.6)].
5.5 Limitations of Vaccine Effectiveness
SPIKEVAX may not protect all vaccine recipients.
6 ADVERSE REACTIONS
In clinical studies contributing to the safety assessment of SPIKEVAX, participants received a single dose, a 2-dose series one month apart (referred to as primary series) and subsequent doses referred to as booster doses, as described in Table 3 below.
Most commonly (≥10%) reported adverse reactions following administration of SPIKEVAX, Moderna COVID-19 Vaccine, Bivalent (Original and Omicron BA.4/BA.5) (hereafter referred to as Moderna COVID-19 Vaccine, Bivalent and no longer authorized for use in the U.S.) or bivalent vaccine (Original and Omicron BA.1) (not authorized for use in the U.S.) containing the same amount of mRNA as the SPIKEVAX 2025-2026 Formula:
-
Participants 6 months through 36 months of age: irritability/crying (up to 82.8%), pain at the injection site (up to 77.2%), sleepiness (up to 52.2%), loss of appetite (up to 46.5%), fever (up to 26.8%), erythema (up to 19.2%), swelling at the injection site (up to 19.2%), and axillary (or groin) swelling/tenderness (up to 12.2%). -
Participants 37 months through 11 years of age: pain at the injection site (up to 98.4%), fatigue (up to 73.2%), headache (up to 62.2%), myalgia (up to 35.3%), chills (up to 34.6%), nausea/vomiting (up to 29.3%), axillary (or groin) swelling/tenderness (up to 27.0%), fever (up to 25.8%), erythema (up to 24.1%), swelling at the injection site (up to 22.3%), and arthralgia (up to 21.3%). -
Participants 12 years through 17 years of age: pain at the injection site (up to 90.6%), fatigue (up to 58.1%), headache (up to 56.3%), myalgia (up to 40.1%), chills (up to 30.2%), axillary swelling/tenderness (up to 27.8%), arthralgia (up to 23.9%), nausea/vomiting (up to 17.9%), and swelling at the injection site (up to 13.3%). -
Participants 18 years through 64 years of age: pain at injection site (up to 86.3%), fatigue (up to 62.0%), headache (up to 58.9%), myalgia (up to 49.6%), arthralgia (up to 41.9%), chills (up to 40.3%), axillary swelling/tenderness (up to 24.8%), and nausea/vomiting (up to 16.7%). -
Participants 65 years of age and older: pain at injection site (up to 76.3%), fatigue (up to 58.1%), myalgia (up to 47.4%), headache (up to 42.1%), arthralgia (up to 39.5%), chills (up to 18.4%), and axillary swelling/tenderness (up to 14.3%).
Most commonly reported adverse reactions following administration of SPIKEVAX or Moderna COVID-19 Vaccine, Bivalent containing the same amount of mRNA as the SPIKEVAX 2025-2026 Formula (≥10%):
-
Participants 6 months through 36 months of age: irritability/crying (up to 82.8%), pain at the injection site (up to 77.2%), sleepiness (up to 52.2%), loss of appetite (up to 46.5%), fever (up to 26.8%), erythema (up to 19.2%), swelling at the injection site (up to 19.2%), and axillary (or groin) swelling/tenderness (up to 12.2%). (6) -
Participants 37 months through 11 years of age: pain at the injection site (up to 98.4%), fatigue (up to 73.2%), headache (up to 62.2%), myalgia (up to 35.3%), chills (up to 34.6%), nausea/vomiting (up to 29.3%), axillary (or groin) swelling/tenderness (up to 27.0%), fever (up to 25.8%), erythema (up to 24.1%), swelling at the injection site (up to 22.3%), and arthralgia (up to 21.3%). (6) -
Participants 12 years through 17 years of age: pain at the injection site (up to 90.6%), fatigue (up to 58.1%), headache (up to 56.3%), myalgia (up to 40.1%), chills (up to 30.2%), axillary swelling/tenderness (up to 27.8%), arthralgia (up to 23.9%), nausea/vomiting (up to 17.9%), and swelling at the injection site (up to 13.3%). (6) -
Participants 18 years through 64 years of age: pain at injection site (up to 86.3%), fatigue (up to 62.0%), headache (up to 58.9%), myalgia (up to 49.6%), arthralgia (up to 41.9%), chills (up to 40.3%), axillary swelling/tenderness (up to 24.8%), and nausea/vomiting (up to 16.7%). (6) -
Participants 65 years of age and older: pain at injection site (up to 76.3%), fatigue (up to 58.1%), myalgia (up to 47.4%), headache (up to 42.1%), arthralgia (up to 39.5%), chills (up to 18.4%), and axillary swelling/tenderness (up to 14.3%). (6)
To report SUSPECTED ADVERSE REACTIONS, contact ModernaTX, Inc. at 1-866-663-3762 or VAERS at 1-800-822-7967 or https://vaers.hhs.gov.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a vaccine cannot be directly compared with rates in the clinical trials of another vaccine and may not reflect the rates observed in practice.
Adults 18 Years of Age and Older
2-Dose Series (Original Monovalent) in Vaccine-Naïve Adults (Study 1)
Study 1 was a Phase 3 clinical trial with multiple parts to evaluate the safety, reactogenicity, and immunogenicity of SPIKEVAX in participants 18 years of age and older (NCT04470427). The randomized, placebo-controlled, observer-blind phase of the trial was conducted in the United States involving 30,346 participants 18 years of age and older who received at least one dose of SPIKEVAX (100 mcg mRNA; n=15,184) or placebo (n=15,162). Upon issuance of the Emergency Use Authorization (December 18, 2020) for Moderna COVID-19 Vaccine (SPIKEVAX), participants were unblinded in a phased manner over a period of months to offer placebo participants SPIKEVAX. The median duration of follow-up for safety after the second injection during the blinded phase was 4 months. The median duration of follow-up for safety after the second injection including both the blinded phase and the open-label phase was 6 months.
In Study 1, the median age of the population was 52 years (range 18-95); 75.2% of participants were 18 years through 64 years of age and 24.8% were 65 years of age and older. Overall, 52.6% of the participants were male, 47.4% were female, 20.5% were Hispanic or Latino, 79.2% were White, 10.2% were African American, 4.6% were Asian, 0.8% were American Indian or Alaska Native, 0.2% were Native Hawaiian or Pacific Islander, 2.0% were other races, and 2.1% were Multiracial. Demographic characteristics were similar between participants who received SPIKEVAX and those who received placebo.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following each injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX (n=15,179) and participants receiving placebo (n=15,159) with at least 1 documented dose. Events that persisted for more than 7 days were followed until resolution. Solicited adverse reactions were reported more frequently among vaccine participants than placebo participants.
The reported number and percentage of the solicited local and systemic adverse reactions by age group and dose are presented in Table 4 and Table 5, respectively.
Solicited local and systemic adverse reactions reported following administration of SPIKEVAX had a median duration of 1 to 3 days.
Grade 3 solicited local adverse reactions were more frequently reported after Dose 2 than after Dose 1. Solicited systemic adverse reactions were more frequently reported by vaccine recipients after Dose 2 than after Dose 1.
In Study 1, 2.3% of participants (vaccine=347, placebo=337) had evidence of prior SARS-CoV-2 infection at baseline (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]). Overall, among the 347 vaccine participants, there were no notable differences in reactogenicity compared to the 14,750 vaccine participants who had no evidence of prior SARS-CoV-2 infection at baseline (negative RT-PCR test and negative Elecsys immunoassay result at Day 1).
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration (2 years). Among the 30,346 participants who had received at least 1 dose of vaccine (N=15,184) or placebo (N=15,162), unsolicited adverse events that occurred within 28 days following any vaccination were reported by 31.3% of participants (n=4,752) who received SPIKEVAX and 28.6% of participants (n=4,338) who received placebo.
During the 28-day follow-up period following any dose, lymphadenopathy-related events were reported by 1.7% of vaccine recipients and 0.8% of placebo recipients. These events included lymphadenopathy, lymphadenitis, lymph node pain, vaccination-site lymphadenopathy, injection-site lymphadenopathy, and axillary mass. This imbalance is consistent with the imbalance observed for solicited axillary swelling/tenderness at the injected arm.
During the 7-day follow-up period of any vaccination, hypersensitivity events of injection site rash or injection site urticaria, likely related to vaccination, were reported by 6 participants in the SPIKEVAX group and none in the placebo group. Delayed injection site reactions that began >7 days after vaccination were reported in 1.4% of vaccine recipients and 0.7% of placebo recipients. Delayed injection site reactions included pain, erythema, and swelling and are likely related to vaccination.
In the blinded portion of the study, there were 8 reports of facial paralysis (including Bell’s palsy) in the SPIKEVAX group, and 3 in the placebo group. In the 28-day follow-up period there were two cases of facial paralysis in the SPIKEVAX group, which occurred on 8 and 22 days, respectively, after vaccination, and one in the placebo group, which occurred 17 days after vaccination. Currently available information on facial paralysis is insufficient to determine a causal relationship with the vaccine.
In the blinded portion of the study, there were 50 reports of herpes zoster in the SPIKEVAX group and 23 in the placebo group. In the 28-day period after any vaccination, there were 22 cases of herpes zoster in the SPIKEVAX group and 15 in the placebo group. Currently available information on herpes zoster infection is insufficient to determine a causal relationship with the vaccine.
There were no other notable patterns or numerical imbalances between treatment groups for specific categories of adverse events (including other neurologic, neuro-inflammatory, and thrombotic events) that would suggest a causal relationship to SPIKEVAX.
Serious Adverse Events
During the blinded phase of the study, serious adverse events were reported by 1.8% (n=268) of participants who received SPIKEVAX and 1.9% (n=292) of participants who received placebo.
There were three serious adverse events of angioedema/facial swelling in the vaccine group in recipients with a history of injection of dermatological fillers. The onset of swelling was reported 1-2 days after the second dose and was likely related to vaccination.
There were no other notable patterns or imbalances between treatment groups for specific categories of serious adverse events (including neurologic, neuro-inflammatory, and thrombotic events) that would suggest a causal relationship to SPIKEVAX.
Single Dose (Original Monovalent) in Vaccine-Experienced Adults (Study 2)
Study 2 was a Phase 2, randomized, observer-blind, placebo-controlled, dose-confirmation study to evaluate the safety, reactogenicity, and immunogenicity of SPIKEVAX in participants 18 years of age and older (NCT04405076). In this study, 198 participants received two doses 1 month apart of SPIKEVAX primary series (100 mcg mRNA per dose). In an open-label phase of the study, 171 of those participants received a single booster dose (50 mcg mRNA) at least 6 months (range 5.8-8.5) after receiving the second dose of the primary series.
Among the 171 booster dose recipients, the median age was 55 years (range 18-87); 77.8% of participants were 18 years through 64 years of age, 22.2% were 65 years of age and older, 39.2% were male, 60.8% were female, 5.8% were Hispanic or Latino, 95.9% were White, 2.9% were African American, 0.6% were Asian, and 0.6% were American Indian or Alaska Native.
Solicited Adverse Reactions
Solicited local and systemic adverse reactions in participants 18 years through 64 years of age starting within 7 days after administration of a booster dose included pain at the injection site (86.0%), fatigue (62.0%), headache (58.9%), myalgia (49.6%), arthralgia (41.9%), chills (40.3%), axillary swelling/tenderness (24.8%), nausea/vomiting (12.4%), fever (7.0%), swelling at the injection site (6.2%), erythema at the injection site (5.4%), and rash (2.3%).
Solicited local and systemic adverse reactions in participants 65 years of age and older starting within 7 days after administration of a booster dose included pain at the injection site (76.3%), fatigue (47.4%), myalgia (47.4%), headache (42.1%), arthralgia (39.5%), chills (18.4%), nausea/vomiting (7.9%), fever (5.4%), axillary swelling/tenderness (5.3%), erythema at the injection site (2.6%), and swelling at the injection site (2.6%).
No Grade 4 adverse reactions were reported. The median duration of solicited local and systemic adverse reactions was 2 to 3 days.
Unsolicited Adverse Events
Overall, the 171 participants who received a booster dose had a median follow-up time of 176 days after the booster dose to the database lock date (November 23, 2021). Through 28 days after the booster dose, unsolicited adverse events were reported by 14.6% of participants (n=25) after the booster dose. There were no unsolicited adverse events not already captured by solicited local and systemic reactions that were considered causally related to SPIKEVAX.
Serious Adverse Events
There were no serious adverse events reported from the booster dose through 28 days after the booster dose. Through the database lock date (November 23, 2021), there were no serious adverse events following the booster dose considered causally related to SPIKEVAX.
Single Dose (Original Monovalent) in Vaccine-Experienced Adults (Study 1)
Study 1 was a Phase 3 clinical trial with multiple parts to evaluate the safety, reactogenicity, and immunogenicity of SPIKEVAX in participants 18 years of age and older (NCT04470427). In the open-label booster dose phase of this study, 19,609 participants received a single booster dose of SPIKEVAX (50 mcg mRNA). Of these participants, 19,599 had previously received a primary series of SPIKEVAX and received a single booster dose at least 6 months (range 5-19) after receiving the second dose of the primary series.
Among the 19,609 booster dose recipients, the median age was 55 years (range 19-96); 69.6% of participants were 18 years through 64 years of age, 30.4% were 65 years of age and older, 52.4% were male, 47.6% were female, 20.2% were Hispanic or Latino, 78.9% were White, 10.6% were African American, 4.1% were Asian, 0.8% were American Indian or Alaska Native, 0.2% were Native Hawaiian or Pacific Islander, 2.0% were other races, and 2.2% were Multiracial. After October 1, 2021, cases of potential myocarditis and/or pericarditis that were identified by the investigator or Applicant were adjudicated by an independent CEAC to determine if they met the CDC definition of confirmed or probable myocarditis and/or pericarditis. In these analyses, the median follow-up time after the booster dose through the cutoff date (April 5, 2022) was 161 days and 3,361 study participants who received the booster dose (17.1%) had at least 6 months of follow-up.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following the booster dose. As of the cutoff date (April 5, 2022), among the 19,609 participants who had received a booster dose, unsolicited adverse events that occurred within 28 days after the booster were reported by 31.7% of participants (n=6,209). The safety profile for the booster dose of SPIKEVAX was similar to the safety profile of the SPIKEVAX primary series in this population.
Serious Adverse Events
Serious adverse events through 28 days following the booster dose of SPIKEVAX were reported by 0.5% of participants (n=94). Through the cutoff date (April 5, 2022), 2.3% of participants (n=442) reported serious adverse events following the booster dose.
A 42-year-old male experienced probable myocarditis (per CEAC adjudication) with onset of symptoms the same day following receipt of the booster dose of SPIKEVAX. Myocarditis was reported as resolved without sequelae on Day 72. This event was considered related to SPIKEVAX.
There was one serious adverse event of erythema nodosum 8 days after the booster dose in a 73-year-old female. This event was considered causally related to SPIKEVAX and was reported as resolved without treatment on Day 30.
Single Dose (Bivalent Original and Omicron BA.4/BA.5) in Vaccine-Experienced Adults (Study 5)
Study 5 (NCT04927065), a Phase 2/3 open-label study with multiple parts conducted in the United States, evaluated the immunogenicity, safety, and reactogenicity of a second booster dose of Moderna COVID-19 Vaccine, Bivalent (50 mcg mRNA) compared to a second booster dose of SPIKEVAX (50 mcg mRNA) when administered to participants 18 years of age and older who had previously received a primary series and a first booster dose with SPIKEVAX at least 3 months prior. The safety analysis set included 511 participants in the Moderna COVID-19 Vaccine, Bivalent booster dose group and 376 participants in the SPIKEVAX booster dose group.
For the Moderna COVID-19 Vaccine, Bivalent group, the median age of the population was 50 years (range 19-89); 79.5% of participants were 18 years through 64 years of age and 20.5% were 65 years of age and older. Overall, 38.2% were male, 61.8% were female, 11.4% were Hispanic or Latino, 83.4% were White, 11.0% were African American, 2.2% were Asian, 0.2% were American Indian or Alaska Native, 1.2% were other races, and 1.6% were Multiracial. For the SPIKEVAX group, the median age of the population was 61 years (range 20-96); 60.1% of participants were 18 years through 64 years of age and 39.9% were 65 years of age and older. Overall, 49.5% were male, 50.5% were female, 9.8% were Hispanic or Latino, 85.6% were White, 7.4% were African American, 4.3% were Asian, 0.3% were American Indian or Alaska Native, 0.3% were Native Hawaiian or Pacific Islander, 0.5% were other races, and 0.5% were Multiracial.
In these analyses, the median follow-up time after the booster dose through the cutoff date (September 23, 2022) for the Moderna COVID-19 Vaccine, Bivalent recipients was 37 days. The median follow-up time after the booster dose through the cutoff date (July 6, 2022) for the SPIKEVAX recipients was 127 days.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following the injection (i.e., day of vaccination and the next 6 days) among participants receiving Moderna COVID-19 Vaccine, Bivalent and participants receiving SPIKEVAX. Events that persisted for more than 7 days were followed until resolution.
Table 6 and Table 7 present the frequency and severity of reported solicited local and systemic adverse reactions within 7 days following a second booster dose with Moderna COVID-19 Vaccine, Bivalent compared to SPIKEVAX in participants 18 years through 64 years of age and 65 years of age and older.
The median duration of solicited local and systemic adverse reactions was 3 days in participants who received either vaccine booster dose.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following the booster dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. As of September 23, 2022, among participants who had received a booster dose of Moderna COVID-19 Vaccine, Bivalent (n=511), unsolicited adverse events that occurred within 28 days following vaccination were reported by 22.7% of participants (n=116). As of July 6, 2022, among participants who had received a booster dose of SPIKEVAX (n=376), unsolicited adverse events that occurred within 28 days following vaccination were reported by 21.3% of participants (n=80). In these analyses, 99.6% of study participants in the Moderna COVID-19 Vaccine, Bivalent group had at least 28 days of follow-up after the booster dose to the cutoff date (September 23, 2022) and 100.0% of participants in the SPIKEVAX group had at least 28 days of follow-up after the booster dose to the cutoff date (July 6, 2022). There were no notable differences in specific categories of adverse events that were reported between the vaccine groups.
Serious Adverse Events
As of the cutoff date (September 23, 2022), the median duration of follow-up was 37 days among Moderna COVID-19 Vaccine, Bivalent booster dose recipients. Serious adverse events were reported by 0.6% of participants (n=3) who received Moderna COVID-19 Vaccine, Bivalent. As of the cutoff date (July 6, 2022), the median duration of follow-up was 127 days among the SPIKEVAX booster dose recipients. Serious adverse events were reported by 2.7% of participants (n=10) who received SPIKEVAX. None of the events in either vaccine group were considered to be related to vaccine.
Adolescents 12 Years Through 17 Years of Age
2-Dose Series (Original Monovalent) in Vaccine-Naïve Adolescents (Study 3)
The safety of SPIKEVAX was evaluated in a Phase 3 clinical trial with multiple parts. The randomized, placebo-controlled, observer-blind clinical trial was conducted in the United States involving 3,726 participants 12 years through 17 years of age who received at least one dose of SPIKEVAX (100 mcg mRNA; n=2,486) or placebo (n=1,240) (Study 3, NCT04649151). Participants started to enter an open-label, observational phase after May 10, 2021. After October 1, 2021, cases of potential myocarditis and/or pericarditis that were identified by the investigator or Applicant were adjudicated by an independent Cardiac Event Adjudication Committee (CEAC) to determine if they met the CDC definition of confirmed or probable myocarditis and/or pericarditis. A safety analysis was conducted in participants who received SPIKEVAX (n=2,486) with a cutoff date of January 31, 2022. In these analyses, the median duration of follow-up including both the blinded and open-label phases was 312 days after Dose 2 and 95.7% of study participants had at least 6 months of follow-up after Dose 2.
Overall, 51.4% were male, 48.6% were female, 11.6% were Hispanic or Latino, 83.8% were White, 3.4% were African American, 6.0% were Asian, 0.5% were American Indian or Alaska Native, <0.1% were Native Hawaiian or Pacific Islander, 1.0% were other races, and 4.5% were Multiracial. Demographic characteristics were similar among participants who received SPIKEVAX and those who received placebo.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following each injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX (n=2,485) and participants receiving placebo (n=1,240) with at least 1 documented dose. Events that persisted for more than 7 days were followed until resolution. Solicited adverse reactions were reported more frequently among vaccine participants than placebo participants.
The reported number and percentage of the solicited local and systemic adverse reactions in participants 12 years through 17 years of age by dose are presented in Table 8.
Solicited local and systemic adverse reactions reported following administration of SPIKEVAX had a median duration of 2 to 3 days.
In ages 12 years through 17 years, 5.8% of participants (vaccine=147, placebo=70) had evidence of prior SARS-CoV-2 infection at baseline (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]). An assessment of reactogenicity among participants with evidence of prior SARS-CoV-2 infection compared to those with no evidence of infection at baseline (negative RT-PCR test and negative Elecsys immunoassay result at Day 1) was conducted. Table 9 presents the number and percentage of the solicited local and systemic adverse reactions in SPIKEVAX participants starting within 7 days after each dose by SARS-CoV-2 status.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among the 3,726 participants who had received at least 1 dose of vaccine (n=2,486) or placebo (n=1,240), unsolicited adverse events that occurred within 28 days following any vaccination were reported by 23.4% of participants (n=582) who received SPIKEVAX and 19.1% of participants (n=237) who received placebo.
In the open-label portion of the study, a 14-year-old male experienced probable myocarditis with onset of symptoms 1 day after Dose 2 of SPIKEVAX. Symptoms resolved after 8 days and no sequelae were observed at 5 months. This event was considered related to SPIKEVAX and was subsequently adjudicated by the CEAC as probable myocarditis. There were no cases of myocarditis among placebo recipients.
During the 28-day follow-up period following any dose, lymphadenopathy-related events were reported by 6.0% of vaccine recipients and 0.6% of placebo recipients. These events included lymphadenopathy, vaccination-site lymphadenopathy, and injection-site lymphadenopathy which were plausibly related to vaccination. This imbalance is consistent with the imbalance observed for solicited axillary swelling/tenderness in the injected arm.
During the 28-day follow-up period following any dose, hypersensitivity events of injection site rash or injection site urticaria, likely related to vaccination, were reported by 0.3% of participants in the SPIKEVAX group and <0.1% in the placebo group. Delayed injection site reactions that began >7 days after vaccination were reported in 1.5% of vaccine recipients and in <0.1% of placebo recipients. Delayed injection site reactions included pain, erythema, and swelling and are likely related to vaccination.
There were no other notable patterns or numerical imbalances between treatment groups for specific categories of adverse events (including other neurologic, neuro-inflammatory, and thrombotic events) that would suggest a causal relationship to SPIKEVAX.
Serious Adverse Events
During the blinded portion of the study, serious adverse events were reported by 0.4% (n=9) of participants who received SPIKEVAX and 0.2% (n=3) of participants who received placebo. In the open-label phase, an additional 12 SPIKEVAX recipients reported serious adverse events. There were no serious adverse events considered causally related to the vaccine.
There were no notable patterns or imbalances between treatment groups for specific categories of serious adverse events (including neurologic, neuro-inflammatory, and thrombotic events) that would suggest a causal relationship to SPIKEVAX.
Single Dose (Original Monovalent) in Vaccine-Experienced Adolescents (Study 3)
Safety data for a booster dose of SPIKEVAX in adolescents were collected in a Phase 3 clinical trial with multiple parts. The open-label booster portion of the study included 1,405 participants who were 12 years through 17 years of age at the time of first dose of the primary series and who received a booster dose of SPIKEVAX (50 mcg mRNA) at least 5 months (range 2.1-16.9) after the second dose of the primary series (Study 3, NCT04649151). Overall, 51.5% were male, 48.5% were female, 13.4% were Hispanic or Latino, 84.9% were White, 3.1% were African American, 4.9% were Asian, 0.5% were American Indian or Alaska Native, <0.1% were Native Hawaiian or Pacific Islander, 0.7% were other races, and 5.2% were Multiracial. The median duration of follow-up for safety after the booster dose was 204 days.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following the injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX as a booster dose. Events that persisted for more than 7 days were followed until resolution.
Table 10 presents the frequency and severity of reported solicited local and systemic adverse reactions among SPIKEVAX booster dose recipients 12 years through 17 years of age within 7 days of a booster vaccination.
In participants who received a booster dose, the median duration of solicited local and systemic adverse reactions was 2 to 3 days.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following the booster dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. As of August 15, 2022, among the 1,405 participants who had received a booster dose of SPIKEVAX, unsolicited adverse events that occurred within 28 days following vaccination were reported by 14.9% of participants (n=209). In these analyses, 85.7% of study participants had at least 6 months of follow-up after the booster dose. Overall, there were no notable differences in the safety profiles observed between participants who had received a booster dose of SPIKEVAX and those who had received a primary series.
Serious Adverse Events
Through the cutoff date of August 15, 2022, with a median follow-up duration of 204 days after the booster dose, there were no serious adverse events considered causally related to the vaccine.
Single Dose (Bivalent Original and Omicron BA.4/BA.5) in Vaccine-Naïve Adolescents with Evidence of Prior SARS-CoV-2 Infection (Study 3)
Safety data for a single dose of Moderna COVID-19 Vaccine, Bivalent in adolescents were collected in a Phase 3 clinical trial with multiple parts. The open-label single dose portion of the study conducted in the United States and the Dominican Republic included 379 participants 12 years through 17 years of age who were COVID-19 vaccine-naïve and received a single 50 mcg dose of Moderna COVID-19 Vaccine, Bivalent (Study 3, NCT04649151). Overall, 52.8% were male, 47.2% were female, 94.5% were Hispanic or Latino, 10.3% were White, 32.2% were African American or Black, 56.7% were other races, and 0.8% were Multiracial. Of the 379 participants, 99.7% had evidence of prior SARS-CoV-2 infection at baseline (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]). The median duration of follow-up for safety after vaccination was 35 days.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following the injection (i.e., day of vaccination and the next 6 days) among participants receiving a single dose of Moderna COVID-19 Vaccine, Bivalent. Events that persisted for more than 7 days were followed until resolution.
Table 11 presents the frequency and severity of reported solicited local and systemic adverse reactions among Moderna COVID-19 Vaccine, Bivalent single dose recipients 12 years through 17 years of age within 7 days of vaccination.
The median duration of solicited local and systemic adverse reactions was 2 days.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following vaccination. Serious adverse events and medically attended adverse events were recorded for the entire study duration. As of June 5, 2023, among the 379 participants who had received a single dose of Moderna COVID-19 Vaccine, Bivalent, unsolicited adverse events that occurred within 28 days following vaccination were reported by 12.9% of participants (n=49). In these analyses, 68.9% of study participants had at least 28 days of follow-up after vaccination. There were no unsolicited adverse events not already captured by solicited local and systemic reactions that were considered causally related to SPIKEVAX.
Serious Adverse Events
Through the cutoff date of June 5, 2023, with a median follow-up duration of 35 days after vaccination, serious adverse events were reported by 0.8% of participants (n=3). None of these events were considered to be related to vaccine.
Children 2 Years Through 11 Years of Age
2-Dose Series (Original Monovalent) in Vaccine-Naïve Children 6 Years Through 11 Years of Age (Study 4)
The safety of SPIKEVAX was evaluated in a Phase 2/3 clinical trial with multiple parts. The study included a randomized, placebo-controlled, observer-blind phase conducted in the United States and Canada involving 4,002 participants 6 years through 11 years of age who received at least one dose of SPIKEVAX (50 mcg mRNA; n=3,007) or placebo (n=995) (Study 4, NCT04796896). Participants started to enter an open-label, observational phase after November 1, 2021, in which placebo recipients were offered SPIKEVAX. A safety analysis was conducted in participants who received SPIKEVAX (n=3,708). In these analyses, the median duration of follow-up including both the blinded and open-label phases was 260 days after Dose 2 and 97.4% of study participants had at least 6 months of follow-up after Dose 2.
Overall, 50.8% were male, 49.2% were female, 18.5% were Hispanic or Latino, 65.6% were White, 10.1% were Black or African American, 9.9% were Asian, 0.4% were American Indian or Alaska Native, <0.1% were Native Hawaiian or Pacific Islander, 2.1% were other races, and 10.7% were Multiracial. Demographic characteristics were similar among participants who received SPIKEVAX and those who received placebo.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following each injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX (n=3,005) and participants receiving placebo (n=994) with at least 1 documented dose. Events that persisted for more than 7 days were followed until resolution. Solicited adverse reactions were reported more frequently among vaccine participants than placebo participants.
The reported number and percentage of the solicited local and systemic adverse reactions in participants 6 years through 11 years of age by dose are presented in Table 12.
Solicited local and systemic adverse reactions reported following administration of SPIKEVAX had a median duration of 2 to 3 days.
An assessment of reactogenicity among participants with evidence of prior SARS-CoV-2 infection (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]) compared to those with no evidence of infection at baseline (negative RT-PCR test and negative Elecsys immunoassay result at Day 1) was conducted. In ages 6 years through 11 years, 8.6% of participants (vaccine=257, placebo=87) had evidence of prior SARS-CoV-2 infection at baseline. Fever (temperature ≥38°C) and headache were reported in a greater proportion of baseline SARS-CoV-2 positive vaccine participants compared to baseline SARS-CoV-2 negative vaccine participants. There were no notable differences in other solicited adverse reactions.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among the 4,002 participants who had received at least 1 dose of vaccine (n=3,007) or placebo (n=995), unsolicited adverse events that occurred within 28 days following any vaccination were reported by 26.1% of participants (n=785) who received SPIKEVAX and 20.8% of participants (n=207) who received placebo. A 7-year-old female experienced a moderate, nonserious event of alopecia areata 27 days after receiving Dose 1. The participant received Dose 2. The event was considered related to the vaccine and was ongoing when the participant discontinued from the study at Day 255 due to withdrawal of consent.
There were no notable patterns or numerical imbalances between treatment groups for specific categories of adverse events that would suggest a causal relationship to SPIKEVAX.
Serious Adverse Events
During the blinded portion of the study, serious adverse events were reported by 0.3% of participants (n=8) who received SPIKEVAX and 0.1% of participants (n=1) who received placebo. In the open-label phase, an additional 17 SPIKEVAX recipients reported serious adverse events. There were no serious adverse events considered causally related to the vaccine.
2-Dose Series (Original Monovalent) in Vaccine-Naïve Infants and Children 2 Years Through 5 Years of Age (Study 4)
The safety of SPIKEVAX was evaluated in a Phase 2/3 clinical trial with multiple parts. The study included a randomized, placebo-controlled, observer-blind phase conducted in the United States and Canada involving 6,698 participants 6 months through 5 years of age who received at least one dose of SPIKEVAX (25 mcg mRNA; n=5,025) or placebo (n=1,673) (Study 4, NCT04796896). Participants started to enter an open-label, observational phase after June 17, 2022, in which placebo recipients were unblinded and offered SPIKEVAX. A safety analysis was conducted in 3,671 participants 2 years through 5 years of age who received SPIKEVAX. In these analyses, the median duration of follow-up including both the blinded and open-label phases for participants 2 years through 5 years of age was 330 days after Dose 2 and 84.9% of study participants had at least 6 months of follow-up after Dose 2.
For participants 2 years through 5 years of age, 50.8% were male, 49.2% were female, 14.1% were Hispanic or Latino, 76.5% were White, 4.5% were Black or African American, 6.0% were Asian, 0.3% were American Indian or Alaska Native, 0.2% were Native Hawaiian or Pacific Islander, 1.5% were other races, and 10.5% were Multiracial. Demographic characteristics were similar among participants who received SPIKEVAX and those who received placebo.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following each injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX and participants receiving placebo with at least 1 documented dose (for participants 24 months to 36 months, vaccine=983, placebo=340; for participants 37 months to 5 years, vaccine=2,031, placebo=658). Events that persisted for more than 7 days were followed until resolution. Solicited adverse reactions were reported more frequently among vaccine participants than placebo participants.
The reported number and percentage of the solicited local and systemic adverse reactions by dose in participants 24 months through 36 months of age are presented in Table 13 and participants 37 months to 5 years of age are presented in Table 14.
Solicited local and systemic adverse reactions reported following administration of SPIKEVAX had a median duration of 2 days for participants 2 years through 5 years of age.
Solicited Adverse Reactions Among Participants with Evidence of Prior SARS-CoV-2 Infection
An assessment of reactogenicity among participants with evidence of prior SARS-CoV-2 infection (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]) compared to those with no evidence of infection at baseline (negative RT-PCR test and negative Elecsys immunoassay result at Day 1) was conducted. In the 2 years through 5 years of age cohort, 8.6% of participants (vaccine=267, placebo=82) had evidence of prior SARS-CoV-2 infection at baseline. Fever (temperature ≥38°C) was reported in a greater proportion of baseline SARS-CoV-2 positive vaccine participants compared to baseline SARS-CoV-2 negative vaccine participants. There were no notable differences in other solicited adverse reactions.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. In the blinded phase, among participants 2 years through 5 years of age who had received at least 1 dose of vaccine or placebo (vaccine=3,031, placebo=1,007), unsolicited adverse events that occurred within 28 days following each vaccination were reported by 35.9% of participants (n=1,087) who received SPIKEVAX and 32.3% of participants (n=325) who received placebo. In these analyses, 95.2% of study participants had at least 28 days of follow-up after Dose 2. A 3-year-old male participant experienced mild erythema multiforme (bilateral arms) 3 days after receiving Dose 2 which resolved the next day without treatment. The event was considered related to the vaccine.
Serious Adverse Events
During the blinded part of the study, serious adverse events were reported by 0.6% of participants (n=19) who received vaccine and 0.3% of participants (n=3) who received placebo who were 2 years through 5 years of age. In the open-label phase, an additional 21 SPIKEVAX recipients reported serious adverse events. None of the events were considered related to vaccine.
There were no notable patterns or imbalances between treatment groups for specific categories of serious adverse events that would suggest a causal relationship to SPIKEVAX.
Single Dose (Monovalent Omicron XBB.1.5) in Vaccine-Naïve Children 2 Years Through 4 Years of Age (Study 6)
Safety data for a single dose of SPIKEVAX (2023-2024 Formula) in individuals 2 years through 4 years of age were collected in an ongoing Phase 3 open-label clinical trial with multiple parts (Study 6, NCT05436834). The single dose phase of the study conducted in the United States, Panama, and the Dominican Republic involved 199 participants 2 years through 4 years of age who were COVID-19 vaccine-naïve and received a single 25 mcg dose of SPIKEVAX (2023-2024 Formula). Overall, 50.3% were male, 49.7% were female, 69.3% were Hispanic or Latino, 24.1% were White, 33.2% were Black or African American, 0.5% were American Indian or Alaska Native, 36.7% were other races, and 5.5% were Multiracial. In these analyses, the median duration of follow-up after vaccination was 92 days and 99.5% of study participants had at least 3 months of follow-up.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following the injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX (2023-2024 Formula). Events that persisted for more than 7 days were followed until resolution.
Table 15 presents the frequency and severity of reported solicited local and systemic adverse reactions among SPIKEVAX (2023-2024 Formula) single dose recipients 2 years through 4 years of age within 7 days of vaccination.
The median duration of solicited local and systemic adverse reactions was 1 day in participants 2 years through 4 years of age.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following vaccination. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among participants 2 years through 4 years of age who had received a single dose of SPIKEVAX (2023-2024 Formula), unsolicited adverse events that occurred within 28 days following vaccination were reported by 19.6% of participants (n=39). In these analyses, all study participants had at least 28 days of follow-up after vaccination. There were no unsolicited adverse events assessed as related to SPIKEVAX (2023-2024 Formula).
Serious Adverse Events
Through the end of this part of the study for participants 2 years through 4 years of age, with a median follow-up duration of 92 days after vaccination, no serious adverse events were reported.
2-Dose Series (Bivalent Original and Omicron BA.1) in Vaccine-Naive Children 2 Years Through 5 Years of Age (Study 6)
Safety data for bivalent vaccine (Original and Omicron BA.1) in individuals 6 months through 5 years of age were collected in an ongoing Phase 3 open-label clinical trial with multiple parts. The study included a phase conducted in the United States involving 391 participants 6 months through 5 years of age who received at least one dose of bivalent vaccine (Original and Omicron BA.1) (25 mcg mRNA) (Study 6, NCT05436834). A safety analysis was conducted in 261 participants 2 years through 5 years of age who received bivalent vaccine (Original and Omicron BA.1). In this analysis, 52.5% were male, 47.5% were female, 18.4% were Hispanic or Latino, 54.4% were White, 37.5% were Black or African American, 0.8% were Asian, 1.5% were American Indian or Alaska Native, 0.4% were other races, and 5.4% were Multiracial. The median duration of follow-up was 358 days after Dose 2 and 92.7% of study participants had at least 6 months of follow-up after Dose 2.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among participants 2 years through 5 years of age who had received at least 1 dose of vaccine (n=261), unsolicited adverse events that occurred within 28 days following each vaccination were reported by 30.7% of participants (n=80). In these analyses, 96.6% of study participants had at least 28 days of follow-up after Dose 2.
Serious Adverse Events
Through the end of the open-label phase, serious adverse events were reported by 0.8% of participants 2 years through 5 years of age who received vaccine (n=2). None of the events were considered related to vaccine.
Single Dose (Original Monovalent) in Vaccine-Experienced Children 6 Years Through 11 Years of Age (Study 4)
Safety data for a booster dose of SPIKEVAX in individuals 6 years through 11 years of age were collected in a Phase 2/3 clinical trial with multiple parts. The open-label booster phase of the study involved 2,290 participants 6 years through 11 years of age who received a booster dose of SPIKEVAX (25 mcg mRNA) at least 6 months after the second dose of the primary series (Study 4, NCT04796896). Overall, 52.6% were male, 47.4% were female, 16.9% were Hispanic or Latino, 65.6% were White, 11.1% were Black or African American, 8.2% were Asian, 0.5% were American Indian or Alaska Native, 0.1% were Native Hawaiian or Pacific Islander, 2.1% were other races, and 11.5% were Multiracial. The median duration of follow-up for safety after the booster dose was 368 days and 96.9% had at least 6 months of follow-up.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following the injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX. Events that persisted for more than 7 days were followed until resolution.
Table 16 presents the frequency and severity of reported solicited local and systemic adverse reactions among SPIKEVAX booster dose recipients 6 years through 11 years of age within 7 days of a booster vaccination.
In participants who received a booster dose, the median duration of solicited local and systemic adverse reactions was 2 days.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following the booster dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among the 2,290 participants who had received a booster dose of SPIKEVAX, unsolicited adverse events that occurred within 28 days following vaccination were reported by 10.0% of participants (n=229). In these analyses, 96.9% of study participants had at least 6 months of follow-up after the booster dose. A nonserious event of serum sickness-like reaction with onset 10 days following administration of a booster dose was reported in an 8-year-old male participant. This event was mild and assessed as related to vaccination. After initiation of treatment with antihistamines and steroids, symptoms resolved and the event was considered resolved 47 days after onset.
Serious Adverse Events
Through the end of the open-label booster phase of this study, serious adverse events were reported by 0.4% of participants (n=10) who received a booster dose. None of the events were considered causally related to the vaccine.
Single Dose (Bivalent Original and Omicron BA.1) in Vaccine-Experienced Children 2 Years Through 5 Years of Age (Study 6)
Safety data for a booster dose of bivalent vaccine (Original and Omicron BA.1) in individuals 6 months through 5 years of age were collected in an ongoing Phase 3 open-label clinical trial with multiple parts (Study 6, NCT05436834). The booster phase of the study conducted in the United States involved 539 participants 6 months through 5 years of age who received a booster dose of bivalent vaccine (Original and Omicron BA.1) (10 mcg mRNA) at least 4 months (range 4-12; median 8 months) after the second dose of the SPIKEVAX primary series. A safety analysis was conducted in 425 participants 2 years through 5 years of age. In this analysis, 52.7% were male, 47.3% were female, 12.2% were Hispanic or Latino, 81.4% were White, 3.8% were Black or African American, 4.7% were Asian, 0.2% were Native Hawaiian or Pacific Islander, and 8.9% were Multiracial. The median duration of follow-up after the booster dose was 184 days and 97.2% of study participants had at least 6 months of follow-up.
Solicited Adverse Reactions
Solicited local and systemic adverse reactions in participants 24 months through 36 months of age (n=145) starting within 7 days after administration of a booster dose included irritability/crying (51.3%), pain at the injection site (36.6%), sleepiness (23.5%), loss of appetite (18.3%), swelling at the injection site (7.6%), erythema at the injection site (6.9%), fever (6.9%), and axillary (or groin) swelling/tenderness (3.4%).
Solicited local and systemic adverse reactions in participants 37 months through 5 years of age (n=281) starting within 7 days after administration of a booster dose included pain at the injection site (54.8%), fatigue (32.1%), headache (14.2%), myalgia (12.4%), arthralgia (9.1%), axillary (or groin) swelling/tenderness (8.2%), nausea/vomiting (8.0%), fever (6.0%), chills (5.8%), erythema at the injection site (4.3%), and swelling at the injection site (3.6%).
No Grade 4 adverse reactions were reported. In participants 2 years through 5 years of age who received a booster dose, the median duration of solicited local and systemic adverse reactions was 1 day.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following the booster dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among participants 2 years through 5 years of age who received a booster dose of bivalent vaccine (Original and Omicron BA.1), unsolicited adverse events that occurred within 28 days following vaccination were reported by 18.4% of participants (n=78). In these analyses, 99.8% of study participants had at least 28 days of follow-up after the booster dose.
Serious Adverse Events
Through the end of this part of the study, serious adverse events were reported by 0.9% of participants 2 years through 5 years of age who received a booster dose (n=4). None of the events were considered causally related to the vaccine.
Single Dose (Bivalent Original and Omicron BA.1) in Vaccine-Experienced Children 2 Years Through 5 Years of Age (Study 4)
Safety data for a booster dose of bivalent vaccine (Original and Omicron BA.1) were collected in participants 6 months through 5 years of age in a Phase 2/3 clinical trial with multiple parts. The open-label booster portion of the study involved 1,514 participants 2 years through 5 years of age who received a booster dose of bivalent vaccine (Original and Omicron BA.1) (10 mcg mRNA) at least 6 months (range 3-18 months; median 12 months) after the second dose of the SPIKEVAX primary series (Study 4, NCT04796896). Safety data included medically attended adverse events and serious adverse events collected for the duration of the study. In this analysis, 51.2% were male, 48.8% were female, 12.7% were Hispanic or Latino, 75.8% were White, 5.2% were African American, 5.1% were Asian, 0.5% were American Indian or Alaska Native, 0.1% were Native Hawaiian or Pacific Islander, 1.0% were other races, and 11.9% were Multiracial. The median duration of follow-up for safety after the booster dose was 184 days and 95.6% had at least 6 months of follow-up.
Through the end of this part of the study, serious adverse events were reported by 0.9% of participants 2 years through 5 years of age who received a booster dose (n=13). None of the events were considered causally related to the vaccine.
Infants and Children 6 Months Through 23 Months of Age
2-Dose Series (Original Monovalent) in Vaccine-Naïve Infants and Children 6 Months Through 23 Months of Age (Study 4)
The safety of SPIKEVAX was evaluated in a Phase 2/3 clinical trial with multiple parts. The study included a randomized, placebo-controlled, observer-blind phase conducted in the United States and Canada involving 6,698 participants 6 months through 5 years of age who received at least one dose of SPIKEVAX (25 mcg mRNA; n=5,025) or placebo (n=1,673) (Study 4, NCT04796896). Participants started to enter an open-label, observational phase after June 17, 2022, in which placebo recipients were unblinded and offered SPIKEVAX. A safety analysis was conducted in 2,438 participants 6 months through 23 months of age who received SPIKEVAX. In these analyses, the median duration of follow-up including both the blinded and open-label phases for participants 6 months through 23 months of age was 314 days after Dose 2 and 80.7% of study participants had at least 6 months of follow-up after Dose 2.
For participants 6 months through 23 months of age, 50.3% were male, 49.7% were female, 13.2% were Hispanic or Latino, 78.6% were White, 3.0% were Black or African American, 5.0% were Asian, 0.3% were American Indian or Alaska Native, 1.5% were other races, and 10.9% were Multiracial. Demographic characteristics were similar among participants who received SPIKEVAX and those who received placebo.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following each injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX and participants receiving placebo with at least 1 documented dose (vaccine=1,991, placebo=664). Events that persisted for more than 7 days were followed until resolution. Solicited adverse reactions were reported more frequently among vaccine participants than placebo participants.
The reported number and percentage of the solicited local and systemic adverse reactions by dose in participants 6 months through 23 months of age are presented in Table 17.
Solicited local and systemic adverse reactions reported following administration of SPIKEVAX had a median duration of 2 to 4 days for participants 6 months through 23 months of age.
Solicited Adverse Reactions Among Participants with Evidence of Prior SARS-CoV-2 Infection
An assessment of reactogenicity among participants with evidence of prior SARS-CoV-2 infection (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]) compared to those with no evidence of infection at baseline (negative RT-PCR test and negative Elecsys immunoassay result at Day 1) was conducted. In the 6 months through 23 months of age cohort, 6.8% of participants (vaccine=133, placebo=47) had evidence of prior SARS-CoV-2 infection at baseline. Fever (temperature ≥38°C) was reported in a greater proportion of baseline SARS-CoV-2 positive vaccine participants compared to baseline SARS-CoV-2 negative vaccine participants. There were no notable differences in other solicited adverse reactions.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. In the blinded phase, among participants 6 months through 23 months of age who had received at least 1 dose of vaccine or placebo (vaccine=1,994, placebo=666), unsolicited adverse events that occurred within 28 days following each vaccination were reported by 44.3% of participants (n=883) who received SPIKEVAX and 42.5% of participants (n=283) who received placebo. In these analyses, 96.2% of study participants had at least 28 days of follow-up after Dose 2.
There were no notable patterns or numerical imbalances between treatment groups for specific categories of adverse events that would suggest a causal relationship to SPIKEVAX.
Serious Adverse Events
During the blinded part of the study, serious adverse events were reported by 1.6% of participants (n=31) who received vaccine and 0.9% of participants (n=6) who received placebo who were 6 months through 23 months of age. In the open-label phase, an additional 18 SPIKEVAX recipients reported serious adverse events. Two serious adverse events were considered related to vaccination: a 1-year-old female experienced serious adverse events of a Grade 3 fever 6 hours after Dose 1; and a febrile convulsion 1 day after Dose 1.
2-Dose Series (Monovalent Omicron XBB.1.5) in Vaccine-Naïve Infants and Children 6 Months Through 23 Months of Age (Study 6)
Safety data for a 2-dose series of SPIKEVAX (2023-2024 Formula) in individuals 6 months through 23 months of age were collected in an ongoing Phase 3 open-label clinical trial with multiple parts (Study 6, NCT05436834). The 2-dose series phase of the study conducted in the United States, Panama, and the Dominican Republic involved 399 participants 6 months through 23 months of age who were COVID-19 vaccine-naïve and received 2 doses of SPIKEVAX (2023-2024 Formula) (25 mcg mRNA). Overall, 52.1% were male, 47.9% were female, 84.7 % were Hispanic or Latino, 10.3% were White, 25.1% were Black or African American, 0.3% were American Indian or Alaska Native, 56.4% were other races, and 7.3% were Multiracial. In these analyses, the median duration of follow-up after Dose 2 was 59 days and 95.7% of study participants had at least 28 days of follow-up after Dose 2.
Solicited Adverse Reactions
Local and systemic adverse reactions and use of antipyretic medication were solicited in an electronic diary for 7 days following each injection (i.e., day of vaccination and the next 6 days) among participants receiving SPIKEVAX (2023-2024 Formula) with at least 1 documented dose. Events that persisted for more than 7 days were followed until resolution.
The reported number and percentage of the solicited local and systemic adverse reactions by dose in participants 6 months through 23 months of age are presented in Table 18.
Solicited local and systemic adverse reactions reported following administration of SPIKEVAX (2023-2024 Formula) had a median duration of 1 day for participants 6 months through 23 months of age.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among participants 6 months through 23 months of age who had received at least 1 dose of vaccine (n=399), unsolicited adverse events that occurred within 28 days following each vaccination were reported by 51.9% of participants (n=207). In these analyses, 95.7% of study participants had at least 28 days of follow-up after Dose 2.
Serious Adverse Events
Through the end of this part of the study, serious adverse events were reported by 3.3% of participants 6 months through 23 months of age who received vaccine (n=13). None of the events were considered related to vaccine.
2-Dose Series (Bivalent Original and Omicron BA.1) in Vaccine-Naive Infants and Children 6 Months Through 23 Months of Age (Study 6)
Safety data for bivalent vaccine (Original and Omicron BA.1) in individuals 6 months through 5 years of age were collected in an ongoing Phase 3 open-label clinical trial with multiple parts. The study included a phase conducted in the United States involving 391 participants 6 months through 5 years of age who received at least one dose of bivalent vaccine (Original and Omicron BA.1) (25 mcg mRNA) (Study 6, NCT05436834). A safety analysis was conducted in 130 participants 6 months through 23 months of age who received SPIKEVAX. In this analysis, 50.0% were male, 50.0% were female, 10.0% were Hispanic or Latino, 63.1% were White, 26.9% were Black or African American, 3.1% were Asian, 0.8% were American Indian or Alaska Native, 0.8% were Native Hawaiian or Pacific Islander, 0.8% were other races, and 4.6% were Multiracial. The median duration of follow-up was 358 days after Dose 2 and 90.8% of study participants had at least 6 months of follow-up after Dose 2.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following each dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration. Among participants 6 months through 23 months of age who had received at least 1 dose of vaccine (n=130), unsolicited adverse events that occurred within 28 days following each vaccination were reported by 44.6% of participants (n=58). In these analyses, 95.4% of study participants had at least 28 days of follow-up after Dose 2. An 11-month-old female participant experienced a nonserious event of Henoch-Schönlein purpura 3 days after receiving Dose 2. There were no symptoms other than rash. The event was considered related to vaccine, and it resolved without treatment 8 days later.
Serious Adverse Events
Through the end of the open-label phase, serious adverse events were reported by 6.2% of participants 6 months through 23 months of age who received vaccine (n=8). None of the events were considered related to vaccine.
Single Dose (Bivalent Original and Omicron BA.1) in Vaccine-Experienced Infants and Children 6 Months Through 23 Months of Age (Study 6)
Safety data for a booster dose of bivalent vaccine (Original and Omicron BA.1) in individuals 6 months through 5 years of age were collected in an ongoing Phase 3 open-label clinical trial with multiple parts (Study 6, NCT05436834). The booster phase of the study conducted in the United States involved 539 participants 6 months through 5 years of age who received a booster dose of bivalent vaccine (Original and Omicron BA.1) (10 mcg mRNA) at least 4 months (range 4-10; median 7 months) after the second dose of the SPIKEVAX primary series. A safety analysis was conducted in 114 participants 6 months through 23 months of age. In this analysis, 45.6% were male, 54.4% were female, 6.1% were Hispanic or Latino, 79.8% were White, 0.9% were Black or African American, 5.3% were Asian, and 13.2% were Multiracial. The median duration of follow-up after the booster dose was 183 days and 97.4% of study participants had at least 6 months of follow-up.
Solicited Adverse Reactions
Solicited local and systemic adverse reactions in participants 6 months through 23 months of age (n=114) starting within 7 days after administration of a booster dose included irritability/crying (54.9%), pain at the injection site (32.5%), loss of appetite (27.4%), sleepiness (17.7%), erythema at the injection site (11.4%), fever (11.4%), swelling at the injection site (8.8%), and axillary (or groin) swelling/tenderness (5.3%).
No Grade 4 adverse reactions were reported. In participants 6 months through 23 months of age who received a booster dose, the median duration of solicited local and systemic adverse reactions was 1 to 3 days.
Unsolicited Adverse Events
Participants were monitored for unsolicited adverse events for up to 28 days following the booster dose. Serious adverse events and medically attended adverse events were recorded for the entire study duration.
Among participants 6 months through 23 months of age who had a booster dose of bivalent vaccine (Original and Omicron BA.1), unsolicited adverse events that occurred within 28 days following vaccination were reported by 29.8% of participants (n=34). In these analyses, 99.1% of study participants had at least 28 days of follow-up after the booster dose.
Serious Adverse Events
Through the end of the booster phase of the study, serious adverse events were reported by 4.4% of participants 6 months through 23 months of age who received a booster dose (n=5). None of the events were considered causally related to the vaccine.
Single Dose (Bivalent Original and Omicron BA.1) in Vaccine-Experienced Infants and Children 6 Months Through 23 Months of Age (Study 4)
Safety data for a booster dose of bivalent vaccine (Original and Omicron BA.1) were collected in participants 6 months through 5 years of age in a Phase 2/3 clinical trial with multiple parts. The open-label booster portion of the study involved 1,252 participants 6 months through 23 months of age who received a booster dose of bivalent vaccine (Original and Omicron BA.1) (10 mcg mRNA) at least 6 months (range 2-20 months; median 11 months) after the second dose of the SPIKEVAX primary series (Study 4, NCT04796896). Safety data included medically attended adverse events and serious adverse events collected for the duration of the study. In this analysis, 50.3% were male, 49.7% were female, 12.2% were Hispanic or Latino, 78.6% were White, 3.7% were African American, 3.8% were Asian, 0.3% were American Indian or Alaska Native, 1.2% were other races, and 11.9% were Multiracial. The median duration of follow-up for safety after the booster dose was 184 days and 96.7% had at least 6 months of follow-up.
Through the end of this part of the study, serious adverse events were reported by 0.8% of participants 6 months through 23 months of age who received a booster dose (n=10). None of the events were considered causally related to the vaccine.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postmarketing use of SPIKEVAX, Moderna COVID-19 Vaccine, or Moderna COVID-19 Vaccine, Bivalent. Because these reactions are reported voluntarily, it is not always possible to reliably estimate their frequency or establish a causal relationship to vaccine exposure.
Cardiac Disorders: myocarditis, pericarditis
Immune System Disorders: anaphylaxis, urticaria
Nervous System Disorders: syncope, febrile seizures
Cardiovascular Outcomes in Patients Diagnosed with mRNA COVID-19 Vaccine-associated Myocarditis
In a longitudinal retrospective observational cohort study across 38 hospitals in the U.S., information on cardiovascular outcomes was collected on 333 patients 5 years through 29 years of age who had been diagnosed with COVID-19 vaccine-associated myocarditis. Among these patients, 322 were confirmed to have received an mRNA COVID-19 vaccine encoding the S glycoprotein of the Original SARS-CoV-2. Of 331 patients, 278 had onset of symptoms following the second dose of a primary series, 33 following the first dose of a primary series, and 20 following a first booster dose.1
Among 307 patients who had been diagnosed with COVID-19 vaccine-associated myocarditis for whom follow-up information was available, 89 reported cardiac symptoms at a median follow-up of 91 days (interquartile range 25-186 days) post-vaccination.1
Initial gadolinium-enhanced cardiac magnetic resonance imaging (CMR) was performed on 216 patients, of whom 177 had late gadolinium enhancement (LGE), a marker of myocardial injury. Among 161 patients who had LGE on initial CMR and who had a follow-up gadolinium-enhanced CMR at a median follow-up of 159 days (interquartile range 78-253 days), 98 had persistence of LGE. Overall, the severity of LGE decreased during follow-up. The clinical and prognostic significance of these CMR findings is not known.1
Limitations of this study include potential selection bias towards patients with more severe myocarditis who are more likely to be hospitalized and have CMR, variability in diagnostic testing, and variability in follow-up.1
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively. Available data on SPIKEVAX administered to pregnant women are insufficient to inform vaccine-associated risks in pregnancy.
A developmental toxicity study was performed in female rats administered the equivalent of a single primary series dose of SPIKEVAX twice prior to mating and twice during gestation. The study revealed no evidence of harm to the fetus due to the vaccine (see Animal Data ).
Data
Animal Data
In a developmental toxicity study, 0.2 mL of a vaccine formulation containing the same quantity of nucleoside-modified messenger ribonucleic acid (mRNA) (100 mcg) and other ingredients included in a single primary series dose of SPIKEVAX for individuals 12 years of age and older was administered to female rats by the intramuscular route on four occasions: 28 and 14 days prior to mating, and on gestation days 1 and 13. No vaccine-related fetal malformations or variations and no adverse effect on postnatal development were observed in the study.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Pregnant individuals infected with SARS-CoV-2 are at increased risk of severe COVID-19 compared with non-pregnant individuals.
8.2 Lactation
Risk Summary
It is not known whether SPIKEVAX is excreted in human milk. Data are not available to assess the effects of SPIKEVAX on the breastfed infant or on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for SPIKEVAX and any potential adverse effects on the breastfed infant from SPIKEVAX or from the underlying maternal condition. For preventive vaccines, the underlying maternal condition is susceptibility to disease prevented by the vaccine.
8.4 Pediatric Use
Safety and effectiveness of SPIKEVAX in individuals 6 months through 17 years of age with at least one high risk factor for severe COVID-19 outcomes is based on safety and effectiveness data in this age group and in adults [see Adverse Reactions (6) and Clinical Studies (14)]. Safety and effectiveness of SPIKEVAX have not been established in individuals less than 6 months of age.
In addition to the Centers for Disease Control and Prevention published list of medical conditions which place individuals at higher risk of experiencing severe outcomes of COVID-19 (https://www.cdc.gov/covid/hcp/clinical-care/underlying-conditions.html), prematurity has been associated with COVID-19-related hospitalizations in children 6 months through 23 months of age.2
8.5 Geriatric Use
Clinical studies of SPIKEVAX and Moderna COVID-19 Vaccine, Bivalent included approximately 7,800 participants 65 years of age and older and 1,400 participants 75 years of age and older [see Adverse Reactions (6.1) and Clinical Studies ( 14.1 )].
Some local and systemic adverse reactions were reported in a lower proportion of participants 65 years of age and older compared to participants 18 years through 64 years of age [see Adverse Reactions (6.1)].
Vaccine effectiveness was similar between participants 65 years of age and older and participants 18 years through 64 years [see Clinical Studies (14.1)].
8.6 Immunocompromised Individuals
The Centers for Disease Control and Prevention has published considerations related to COVID-19 vaccination for individuals who are moderately to severely immunocompromised (https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html).
11 DESCRIPTION
SPIKEVAX (COVID-19 Vaccine, mRNA) is a sterile white to off-white injectable suspension for intramuscular use.
Each 0.5 mL dose of SPIKEVAX (2025-2026 Formula) contains 50 mcg nucleoside-modified messenger RNA (mRNA) encoding the pre-fusion stabilized Spike glycoprotein (S) of the SARS-CoV-2 Omicron variant sublineage LP.8.1. Each dose also contains the following ingredients: a total lipid content of 1.01 mg (SM-102, polyethylene glycol [PEG] 2000 dimyristoyl glycerol [DMG], cholesterol, and 1,2-distearoyl-sn-glycero-3-phosphocholine [DSPC]), 0.25 mg tromethamine, 1.2 mg tromethamine hydrochloride, 0.021 mg acetic acid, 0.10 mg sodium acetate trihydrate, and 43.5 mg sucrose.
Each 0.25 mL dose of SPIKEVAX (2025-2026 Formula) contains 25 mcg nucleoside-modified messenger RNA (mRNA) encoding the pre-fusion stabilized Spike glycoprotein (S) of the SARS-CoV-2 Omicron variant sublineage LP.8.1. Each dose also contains the following ingredients: a total lipid content of 0.50 mg (SM-102, polyethylene glycol [PEG] 2000 dimyristoyl glycerol [DMG], cholesterol, and 1,2-distearoyl-sn-glycero-3-phosphocholine [DSPC]), 0.13 mg tromethamine, 0.62 mg tromethamine hydrochloride, 0.011 mg acetic acid, 0.049 mg sodium acetate trihydrate, and 21.8 mg sucrose.
SPIKEVAX does not contain a preservative.
The rubber tip cap and plunger used for the single-dose syringes are not made with natural rubber latex.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The nucleoside-modified mRNA in SPIKEVAX is formulated in lipid particles, which enable delivery of the nucleoside-modified mRNA into host cells to allow expression of the SARS-CoV-2 S antigen. The vaccine elicits an immune response to the S antigen, which protects against COVID-19.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
SPIKEVAX has not been evaluated for carcinogenic, mutagenic potential, or impairment of male fertility in animals. A developmental toxicity study was conducted in female rats that received a vaccine formulation containing nucleoside-modified messenger ribonucleic acid (mRNA) (100 mcg) and other ingredients included in a single human dose of SPIKEVAX. No impact on female fertility was reported (see Use in Specific Populations [8.1]).
14 CLINICAL STUDIES
14.1 Adults 18 Years of Age and Older
Efficacy of 2-Dose Series (Original Monovalent) in Vaccine-Naïve Adults
Study 1 was a Phase 3 clinical trial with multiple parts. The randomized, placebo-controlled, observer-blind phase evaluated the efficacy, safety, and immunogenicity of SPIKEVAX in participants 18 years of age and older in the United States. Randomization was stratified by age and health risk: 18 to <65 years of age without comorbidities (not at risk for progression to severe COVID-19), 18 to <65 years of age with comorbidities (at risk for progression to severe COVID-19), and 65 years of age and older with or without comorbidities. Participants who were immunocompromised and those with a known history of SARS-CoV-2 infection were excluded from the study. Participants with no known history of SARS-CoV-2 infection but with positive laboratory results indicative of infection at study entry were included. The study allowed for the inclusion of participants with stable pre-existing medical conditions, defined as disease not requiring significant change in therapy or hospitalization for worsening disease during the 3 months before enrollment, as well as participants with stable human immunodeficiency virus (HIV) infection. A total of 30,415 participants were randomized equally to receive 2 doses of SPIKEVAX or saline placebo 1 month apart. Participants were followed for efficacy and safety until 2 years after the second dose.
The primary efficacy analysis population (referred to as the Per-Protocol Set) included 28,451 participants who received two doses (at 0 and 1 month) of either SPIKEVAX (100 mcg mRNA per dose; n=14,287) or placebo (n=14,164), and had a negative baseline SARS-CoV-2 status. In the Per-Protocol Set, 47.5% of participants were female, 19.7% were Hispanic or Latino; 79.7% were White, 9.7% were African American, 4.7% were Asian, and 2.0% were other races. The median age of participants was 53 years (range 18-95) and 25.4% of participants were 65 years of age and older. Of the study participants in the Per-Protocol Set, 22.8% were at increased risk of severe COVID-19 due to at least one pre-existing medical condition (chronic lung disease, significant cardiac disease, severe obesity, diabetes, liver disease, or HIV infection) regardless of age. There were no notable differences in demographics or pre-existing medical conditions between participants who received SPIKEVAX and those who received placebo.
The population for the vaccine efficacy analysis included participants 18 years of age and older who were enrolled from July 27, 2020, and followed for the development of COVID-19 through the data cutoff of March 26, 2021, or the Participant Decision Visit for treatment unblinding, whichever was earlier. The median length of follow-up for participants in the blinded placebo-controlled phase of the study was 4 months following Dose 2.
COVID-19 was defined based on the following criteria: The participant must have experienced at least two of the following systemic symptoms: fever (≥38ºC / ≥100.4ºF), chills, myalgia, headache, sore throat, new olfactory and taste disorder(s); or the participant must have experienced at least one of the following respiratory signs/symptoms: cough, shortness of breath or difficulty breathing, or clinical or radiographical evidence of pneumonia; and the participant must have at least one NP swab, nasal swab, or saliva sample (or respiratory sample, if hospitalized) positive for SARS-CoV-2 by RT-PCR. COVID-19 cases were adjudicated by a Clinical Adjudication Committee.
There were 55 COVID-19 cases in the SPIKEVAX group and 744 cases in the placebo group, with a vaccine efficacy of 93.2% (95% confidence interval of 91.0% to 94.8%) (Table 19).
SARS-CoV-2 identified in the majority of COVID-19 cases in this study were sequenced to be the B.1.2 variant. Additional SARS-CoV-2 variants identified in this study included B.1.427/B.1.429 (Epsilon), P.1 (Gamma), and P.2 (Zeta). Representation of identified variants among cases in the vaccine versus placebo recipients did not suggest decreased vaccine effectiveness against these variants.
Severe COVID-19 was defined based on confirmed COVID-19 as per the primary efficacy endpoint case definition, plus any of the following: Clinical signs indicative of severe systemic illness, respiratory rate ≥30 per minute, heart rate ≥125 beats per minute, SpO2 ≤93% on room air at sea level or PaO2/FIO2 <300 mm Hg; or respiratory failure or ARDS (defined as needing high-flow oxygen, non-invasive or mechanical ventilation, or ECMO), evidence of shock (systolic blood pressure <90 mmHg, diastolic BP <60 mmHg or requiring vasopressors); or significant acute renal, hepatic, or neurologic dysfunction; or admission to an intensive care unit or death.
Among all participants in the Per-Protocol Set analysis, which included COVID-19 cases confirmed by an adjudication committee, 2 cases of severe COVID-19 were reported in the SPIKEVAX group compared with 106 cases reported in the placebo group, with a vaccine efficacy of 98.2% (95% confidence interval of 92.8% to 99.6%) (Table 20).
In an exploratory analysis, occurrence of asymptomatic SARS-CoV-2 infection was assessed among participants in the Per-Protocol Set (enrolled from July 27, 2020, and followed maximally through March 26, 2021). Asymptomatic SARS-CoV-2 infection was defined as having a positive scheduled serology test based on binding antibody against SARS-CoV-2 nucleocapsid protein as measured by the Roche Elecsys immunoassay (N-serology) and/or a positive RT-PCR test for SARS-CoV-2, in the absence of any reported COVID-19 symptoms included as part of the primary efficacy endpoint case definition (described above) or symptoms included in the secondary COVID-19 endpoint case definition (fever >38°C / ≥100.4°F, chills, cough, shortness of breath or difficulty breathing, fatigue, muscle aches, body aches, headache, new loss of taste or smell, sore throat, congestion or runny nose, nausea, vomiting, or diarrhea) at any time during the study. To assess for asymptomatic infection starting 14 days after Dose 2, all participants had scheduled blood draws for N-serology collected at the 1-month post-Dose 2 visit and the 6-month post-Dose 2 visit (if still blinded to treatment arm), and scheduled N-serology and nasopharyngeal swab for RT-PCR collection at the Participant Decision Visit for treatment unblinding.
In the Per-Protocol Set, 14,287 participants in the SPIKEVAX group and 14,164 participants in the placebo group had N-serology and/or RT-PCR results available from one or more of the pre-specified timepoints listed above. Among these participants, there were 180 cases of asymptomatic SARS-CoV-2 infection in the SPIKEVAX group compared with 399 cases in the placebo group. Limitations of this analysis include the infrequently scheduled assessments for serology and PCR testing, which may not have captured all cases of asymptomatic infections which occurred during the study.
Immunogenicity of a Single Dose (Original Monovalent) in Vaccine-Experienced Adults
Effectiveness of a booster dose of SPIKEVAX was based on assessment of neutralizing antibody geometric mean concentration (GMC) against a pseudovirus expressing the original SARS-CoV-2 Spike protein (D614G). Immunogenicity analyses compared the GMC following the booster dose to the GMC following the primary series.
In the open-label booster dose phase of Study 1, the primary immunogenicity analysis population included 682 participants 18 years of age and older who received a single booster dose of SPIKEVAX (50 mcg mRNA) at least 6 months after completion of the primary series of SPIKEVAX (two doses of 100 mcg mRNA 1 month apart). Participants had no serologic or virologic evidence of SARS-CoV-2 infection prior to the first primary series dose and prior to the booster dose. Among participants assessed for immunogenicity, the median age at the time of booster dose was 59 years (range 19-87); 61.3% of participants were 18 years through 64 years of age, 38.7% were 65 years of age and older, 52.9% were male, 47.1% were female, 30.4% were Hispanic or Latino; 70.8% were White, 19.6% were African American, 2.5% were Asian, 1.3% were American Indian or Alaska Native, 0.4% were Native Hawaiian or Pacific Islander, 3.1% were other races, and 1.3% were Multiracial.
The primary immunogenicity analyses of the GMC ratio and difference in seroresponse rates following the booster dose compared to after the primary series met the pre-defined success criteria. Seroresponse for a participant was defined as achieving a ≥4-fold rise of neutralizing antibody concentration from baseline (before the first dose of the primary series). These analyses are summarized in Table 21.
A descriptive analysis evaluated seroresponse rates using pre-booster neutralizing antibody concentration. The booster dose seroresponse rate, with seroresponse defined as at least a 4-fold rise relative to the pre-booster concentration, was 98.4%. The difference in seroresponse rates in this post-hoc analysis was -0.8% (95% CI: -2.1%, 0.5%).
Immunogenicity of a Single Dose (Bivalent Original and Omicron BA.4/BA.5) in Vaccine-Experienced Adults
Study 5 was a Phase 2/3 open-label study with multiple parts which evaluated the immunogenicity of a second booster dose of Moderna COVID-19 Vaccine, Bivalent (50 mcg mRNA) compared to a second booster dose of SPIKEVAX (50 mcg mRNA) when administered to participants 18 years of age and older who had previously received a primary series and a first booster dose with SPIKEVAX at least 3 months prior. The primary immunogenicity analysis population included 209 participants who received a booster dose of Moderna COVID-19 Vaccine, Bivalent and 259 participants who received a booster dose of SPIKEVAX. Participants included in the analysis population had no serologic or virologic evidence of SARS-CoV-2 infection prior to the second booster dose.
Among participants assessed for immunogenicity for the Moderna COVID-19 Vaccine, Bivalent group, the median age was 51 years (range 21-84); 74.6% of participants were 18 years through 64 years of age, 25.4% were 65 years of age and older, 35.4% were male, 64.6% were female, 9.6% were Hispanic or Latino, 89.0% were White, 5.3% were African American, 3.3% were Asian, 0.5% were American Indian or Alaska Native, 0.5% were other races, and 1.0% were Multiracial. Among participants assessed for immunogenicity for the SPIKEVAX group, the median age was 63 years (range 21-96); 53.7% of participants were 18 years through 64 years of age, 46.3% were 65 years of age and older, 48.3% of participants were male, 51.7% were female, 8.5% were Hispanic or Latino, 90.0% were White, 4.2% were African American, 4.2% were Asian, and 0.4% were other races. The median time between the first booster dose and the second booster dose was 288 days in the Moderna COVID-19 Vaccine, Bivalent group and 133 days in the SPIKEVAX group.
In Study 5, the neutralizing antibody titers (50% inhibitory dose [ID50]) against a pseudovirus expressing the original SARS-CoV-2 Spike protein (D614G) and a pseudovirus expressing the Omicron BA.4/BA.5 Spike protein were evaluated. Primary immunogenicity analyses compared the ID50 GMTs and seroresponse rates 28 days following a second booster dose with Moderna COVID-19 Vaccine, Bivalent to those following a second booster dose with SPIKEVAX. Analyses of GMTs met predefined success criteria for superiority against Omicron BA.4/BA.5 and noninferiority against the Original strain. The analysis of seroresponse against Omicron BA.4/BA.5 met the criterion for noninferiority: Lower limit of the 2-sided 95% CI for the percentage difference in seroresponse rate (Moderna COVID-19 Vaccine, Bivalent minus SPIKEVAX) >-10%. Table 22 presents the analyses of ID50 GMTs. Table 23 presents the analyses of differences in seroresponse rates (the proportion achieving a ≥4-fold rise in ID50 from pre-second booster) against both the Original strain and Omicron BA.4/BA.5.
In subgroup analyses, neutralizing antibodies against a pseudovirus expressing the original SARS-CoV-2 Spike protein (D614G) and a pseudovirus expressing the Omicron BA.4/BA.5 Spike protein were similar in participants 65 years of age and older compared to participants 18 years through 64 years of age. The seroresponse rate observed at Day 29 was similar between age groups.
14.2 Adolescents 12 Years Through 17 Years of Age
Effectiveness of 2-Dose Series (Original Monovalent) in Vaccine-Naïve Adolescents
Study 3 was a Phase 3 clinical trial with multiple parts. The randomized, placebo-controlled, observer-blind phase evaluated the safety, reactogenicity, and effectiveness of SPIKEVAX in participants ages 12 years through 17 years in the United States. Participants with a known history of SARS-CoV-2 infection were excluded from the study. A total of 3,733 participants were randomized 2:1 to receive 2 doses of SPIKEVAX (100 mcg mRNA per dose) or saline placebo 1 month apart. Among participants assessed for immunogenicity, 52.4% of participants were male, 47.6% were female, 7.6% were Hispanic or Latino; 83.5% were White, 1.2% were African American, 4.4% were Asian, 0.3% were Native Hawaiian or Pacific Islander, 2.1% were other races, and 5.6% were Multiracial.
Effectiveness in individuals 12 years through 17 years of age is based on a comparison of immune responses in this age group to adults 18 years through 25 years of age.
In Study 3, an analysis was conducted of SARS-CoV-2 50% neutralizing titers and seroresponse rates 28 days after Dose 2 in a subset of participants 12 years through 17 years of age in Study 3 and participants 18 years through 25 years of age in Study 1 who had no immunologic or virologic evidence of prior SARS-CoV-2 at baseline. Noninferior immune responses as assessed by geometric mean 50% neutralizing titers and seroresponse rates were demonstrated in a comparison of participants 12 years through 17 years of age to participants 18 years through 25 years of age (Table 24).
A descriptive efficacy analysis evaluating confirmed COVID-19 cases accrued up to the blinded data cutoff date of May 31, 2021, was performed in 3,186 participants who received two doses (at 0 and 1 month) of either SPIKEVAX (n=2,142) or placebo (n=1,044) and had a negative baseline SARS-CoV-2 status (referred to as the Per-Protocol Set for Efficacy). In the Per-Protocol Set for Efficacy, 51.5% were male, 48.5% were female, 11.0% were Hispanic or Latino; 84.0% were White, 2.7% were African American, 6.2% were Asian, 0.5% were American Indian or Alaska Native, <0.1% were Native Hawaiian or Pacific Islander, 0.9% were other races, and 4.8% were Multiracial. There were no notable differences in demographics between participants who received SPIKEVAX and those who received placebo.
The population for the vaccine efficacy analysis included participants 12 years through 17 years of age who were enrolled from December 9, 2020, and followed for the development of COVID-19 through the data cutoff of May 31, 2021. The median length of follow-up for participants in the blinded, placebo-controlled phase of the study was 112 days following Dose 2.
The efficacy information in participants 12 years through 17 years of age is presented in Table 25.
Immunogenicity of a Single Dose (Original Monovalent) in Vaccine-Experienced Adolescents
Effectiveness of a booster dose of SPIKEVAX in participants 12 years through 17 years of age was based on a comparison of immune responses, as assessed by neutralizing antibody concentration against a pseudovirus expressing the original SARS-CoV-2 Spike protein (D614G), following the booster dose in this age group to that following the primary series in adults 18 years through 25 years of age.
In an open-label booster phase of Study 3, participants who were 12 years through 17 years of age at the time of first dose of a primary series received a single booster dose of SPIKEVAX (50 mcg mRNA) at least 5 months (range 2.1-16.9) after completion of the primary series (two doses 1 month apart). The primary immunogenicity analysis population included 264 booster dose participants in Study 3 and a random subset of 295 participants 18 years through 25 years of age from Study 1 who received two doses of SPIKEVAX 1 month apart. Study 1 and Study 3 participants included in the analysis population had no serologic or virologic evidence of SARS-CoV-2 infection prior to the first primary series dose and prior to the booster dose, respectively. Among participants 12 years through 17 years of age assessed for immunogenicity, 50.8% were male, 49.2% were female, 12.5% were Hispanic or Latino; 87.9% were White, 1.5% were African American, 3.4% were Asian, 1.1% were other races, and 5.7% were Multiracial.
The primary immunogenicity analyses of the GMC ratio and difference in seroresponse rates following the booster dose in Study 3 compared to after the primary series in Study 1 met the pre-defined success criteria. Seroresponse for a participant was defined as achieving a ≥4-fold rise of neutralizing antibody concentration from baseline (before the first dose of the primary series in Study 1 and Study 3). These analyses are summarized in Table 26.
A descriptive analysis evaluated seroresponse rates using pre-booster neutralizing antibody concentration. The booster dose seroresponse rate, with seroresponse defined as at least a 4-fold rise relative to the pre-booster concentration, was 96.6%. The difference in seroresponse rates in this post-hoc analysis was -2.7% (95% CI: -5.8%, -0.5%).
Immunogenicity of Single Dose (Bivalent Original and Omicron BA.4/BA.5) in Vaccine-Naïve Adolescents with Evidence of Prior SARS-CoV-2 Infection
In an open-label phase of Study 3 conducted in the United States and the Dominican Republic, participants 12 years through 17 years of age who were COVID-19 vaccine-naïve received a single dose (50 mcg mRNA) of Moderna COVID-19 Vaccine, Bivalent. Of the 246 participants in the immunogenicity subset, 99.6% had evidence of prior SARS-CoV-2 infection at baseline (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]). The primary immunogenicity analysis population included 245 participants who were baseline SARS-COV-2 positive in Study 3 and a subset of 296 participants 18 years through 25 years of age from Study 1 who had no evidence of prior SARS-CoV-2 infection at baseline and received two doses (100 mcg mRNA per dose) of SPIKEVAX 1 month apart. Among participants 12 years through 17 years of age assessed for immunogenicity, 53.5% were male, 46.5% were female, 94.3% were Hispanic or Latino, 14.3% were White, 33.9% were African American or Black, 51.0% were other races, and 0.8% were Multiracial.
A comparison of neutralizing antibody concentrations against a pseudovirus expressing Omicron BA.4/BA.5 and the original SARS-CoV-2 Spike protein (D614G) was conducted. The primary immunogenicity analyses of the GMC ratio following the single dose in Study 3 compared to after the primary series in Study 1 met the pre-defined success criteria for superiority against Omicron BA.4/BA.5 and noninferiority against the Original strain. Secondary analyses included the difference in seroresponse rates, where seroresponse for a participant was defined as achieving a ≥4-fold rise of neutralizing antibody concentration from baseline (before the first dose of the primary series in Study 1 and the single dose in Study 3). These analyses are summarized in Table 27.
14.3 Infants and Children 6 Months Through 11 Years of Age
Effectiveness of 2-Dose Series (Original Monovalent) in Vaccine-Naïve Children 6 Years Through 11 Years of Age (Study 4)
Study 4 included a randomized, placebo-controlled, observer-blind phase to evaluate the safety, reactogenicity, and effectiveness of SPIKEVAX in individuals 6 years through 11 years of age in the United States and Canada. Participants with a known history of SARS-CoV-2 infection within 2 weeks of study vaccination were excluded from the study. A total of 4,015 participants were randomized 3:1 to receive 2 doses of SPIKEVAX (50 mcg mRNA per dose) or saline placebo 1 month apart.
Effectiveness in individuals 6 years through 11 years of age is based on comparison of SARS-CoV-2 50% neutralizing titers and seroresponse rates 28 days after Dose 2 in a subset of individuals 6 years through 11 years of age in Study 4 and participants 18 years through 25 years of age in Study 1 who had no immunologic or virologic evidence of prior SARS-CoV-2 at baseline (Table 28). Demographics in the subset of individuals 6 years through 11 years of age in this analysis were consistent with the overall demographics in this phase of Study 4.
A descriptive efficacy analysis evaluating confirmed COVID-19 cases accrued between September 1, 2021 through the date of individual participant unblinding, was performed in 3,578 participants 6 years through 11 years of age who received two doses (at 0 and 1 month) of either SPIKEVAX or placebo, had a negative baseline SARS-CoV-2 status, and received the correct treatment (referred to as the modified Intent-to-Treat-1 [mITT-1] Set) (vaccine=2,694, placebo=884). Between participants who received SPIKEVAX and those who received placebo, there were no notable differences in demographics.
Vaccine efficacy among individuals 6 years through 11 years of age in Study 4 was evaluated during the period when Delta was the predominant strain in circulation.
The efficacy information in individuals 6 years through 11 years of age is presented in Table 29.
Effectiveness of 2-Dose Series (Original Monovalent) in Vaccine-Naïve Infants and Children 6 Months Through 5 Years of Age (Study 4)
Study 4 included a randomized, placebo-controlled, observer-blind phase to evaluate the safety, reactogenicity, and effectiveness of SPIKEVAX in individuals 6 months through 5 years of age in the United States and Canada. Participants with a known history of SARS-CoV-2 infection within 2 weeks of study vaccination were excluded from the study. A total of 6,712 participants were randomized 3:1 to receive 2 doses of SPIKEVAX (25 mcg mRNA per dose) or saline placebo 1 month apart.
Effectiveness in individuals 6 months through 5 years of age is based on a comparison of SARS-CoV-2 neutralizing antibody concentrations and seroresponse rates 28 days after Dose 2 in a subset of individuals 6 months through 5 years of age in Study 4 and participants 18 years through 25 years of age in Study 1 who had no immunologic or virologic evidence of prior SARS-CoV-2 at baseline (Table 30 and Table 31). Demographics in the subset of individuals 6 months through 5 years of age in this analysis were consistent with the overall demographics in this phase of Study 4.
A descriptive efficacy analysis evaluating confirmed COVID-19 cases accrued between November 1, 2021, through the blinded cutoff date of June 30, 2022, was performed in 5,693 participants 6 months through 5 years of age who received two doses (at 0 and 1 month) of either SPIKEVAX or placebo and had a negative baseline SARS-CoV-2 status (referred to as the Per-Protocol Set for Efficacy) (for participants 6 months through 23 months, vaccine=1,686, placebo=563; for participants 2 years through 5 years, vaccine=2,592, placebo=854). Between participants who received SPIKEVAX and those who received placebo, there were no notable differences in demographics.
The median length of follow-up for efficacy post-Dose 2 was 182 days for participants 6 months through 23 months of age and 186 days for participants 2 years through 5 years of age. Vaccine efficacy among individuals 6 months through 5 years of age in Study 4 was evaluated during the period when the B.1.1.529 (Omicron) variant was the predominant variant in circulation. The efficacy information in individuals 6 months through 23 months of age and 2 years through 5 years of age are presented in Table 32 and Table 33, respectively.
Immunogenicity of Single Dose (Monovalent Omicron XBB.1.5) in Vaccine-Naïve Children 2 Years Through 4 Years of Age and 2-Dose Series (Monovalent Omicron XBB.1.5) in Vaccine-Naïve Infants and Children 6 Months Through 23 Months of Age (Study 6)
Study 6 is an ongoing open-label clinical trial that included a phase conducted in the United States, Panama, and the Dominican Republic in which participants 2 years through 4 years of age who were COVID-19 vaccine-naïve received a single dose (25 mcg mRNA) of SPIKEVAX (2023-2024 Formula) and individuals 6 months through 23 months of age received two doses (25 mcg mRNA) of SPIKEVAX (2023-2024 Formula). Of the 148 participants 2 years through 4 years of age in the immunogenicity subset, 96.6% had evidence of prior SARS-CoV-2 infection at baseline (immunologic or virologic evidence of prior SARS-CoV-2 infection [defined as positive RT-PCR test and/or positive Elecsys immunoassay result at Day 1]). The primary immunogenicity analysis population included 143 participants 2 years through 4 years of age who were baseline SARS-CoV-2 positive and received a single dose of SPIKEVAX (2023-2024 Formula) and a subset of 76 participants 6 months through 23 months of age who had no evidence of prior SARS-CoV-2 infection at baseline and received two doses (25 mcg mRNA per dose) of SPIKEVAX (2023-2024 Formula) 1 month apart. Demographics in the subsets of individuals 6 months through 23 months of age and 2 years through 4 years of age in this analysis were consistent with the overall demographics in this phase of Study 4.
A comparison of neutralizing antibody concentrations against a pseudovirus expressing Omicron XBB.1.5 was conducted. The primary immunogenicity analyses of the GMC ratio following the single dose compared to after the primary series met the pre-defined success criteria for noninferiority against Omicron XBB.1.5 (Table 34).
Secondary analyses included seroresponse rates, where seroresponse for a participant was defined as achieving a ≥4-fold rise of neutralizing antibody concentration from baseline (before the first dose of the primary series or the single dose) if baseline values were ≥ LLOQ, otherwise, 4 times LLOQ. In the 2 years through 4 years single dose group who had evidence of prior SARS-CoV-2 infection at baseline, 73.4% (95% CI: 65.2%, 80.5%) of participants achieved seroresponse against Omicron XBB.1.5. In the 6 months through 23 months primary series group who had no evidence of prior SARS-CoV-2 infection at baseline, 96.1% (95% CI: 88.9%, 99.2%) of participants achieved seroresponse. The seroresponse rate difference between the two age groups was -22.7% (95% CI: -31.3%, -13.5%). The noninferiority criterion for seroresponse rate difference (lower bound of the 95% CI >-10%) was not met.
Immunogenicity of Single Dose (Original Monovalent) in Vaccine-Experienced Children 6 Years Through 11 Years of Age (Study 4)
Effectiveness of a booster dose of SPIKEVAX in participants 6 years through 11 years of age was based on a comparison of immune responses, as assessed by neutralizing antibody concentration against a pseudovirus expressing the original SARS-CoV-2 Spike protein (D614G), following the booster dose in this age group to that following the primary series in young adults 18 years through 25 years.
In an open-label phase of Study 4, participants 6 years through 11 years of age received a single booster dose of SPIKEVAX (25 mcg mRNA) at least 6 months after completion of the primary series (two doses 1 month apart). The primary immunogenicity analysis population included 137 booster dose participants in Study 4 and a random subset of 296 participants 18 years through 25 years of age from Study 1 who received two doses of SPIKEVAX 1 month apart. Study 1 and Study 4 participants included in the analysis population had no serologic or virologic evidence of SARS-CoV-2 infection prior to the first primary series dose and prior to the booster dose, respectively. Demographics in the subset of individuals 6 years through 11 years of age in this analysis were consistent with the overall demographics in this phase of Study 4.
The primary immunogenicity analyses of the GMC ratio and difference in seroresponse rates following the booster dose in Study 4 compared to following the primary series in Study 1 met the pre-defined immunobridging success criteria. Seroresponse for a participant was defined as achieving a ≥4-fold rise of neutralizing antibody concentration from baseline (before the first dose of the primary series in Study 4 and Study 1). These analyses are summarized in Table 35.
A descriptive analysis evaluated seroresponse rates using pre-booster neutralizing antibody concentration. The booster dose seroresponse rate, with seroresponse defined as at least a 4-fold rise relative to the pre-booster concentration for Study 4, was 93.4%. The difference in seroresponse rates in this post-hoc analysis was -5.9% (95% CI: -11.5%, -2.6%).
Immunogenicity of Single Dose (Bivalent Original and Omicron BA.1) in Vaccine-Experienced Infants and Children 6 Months Through 5 Years of Age (Study 6)
Study 6 is an ongoing open-label clinical trial that included a phase conducted in the United States in which participants 6 months through 5 years of age received a single booster dose of bivalent vaccine (Original and Omicron BA.1) (10 mcg mRNA per dose) at least 4 months after completion of a SPIKEVAX primary series (two doses 1 month apart) (NCT05436834). The primary immunogenicity analysis population included 319 booster dose participants in Study 6 and a subset of 590 participants 6 months through 5 years of age from Study 4 who had completed primary vaccination with two doses of SPIKEVAX (25 mcg mRNA per dose) 1 month apart. Study 4 and Study 6 participants included in the analysis population had no serologic or virologic evidence of SARS-CoV-2 infection prior to the first primary series dose and prior to the booster dose, respectively. Demographics in the subset of individuals 6 months through 5 years of age in this analysis were consistent with the overall demographics in this phase of Study 6. Among the bivalent vaccine (Original and Omicron BA.1) participants in the primary immunogenicity analysis population, the median age for receipt of the booster dose was 3 years (range 0.9-5).
The primary immunogenicity analyses of the GMC ratio and difference in seroresponse rates following the booster dose in Study 6 compared to following the primary series in Study 4 met the pre-defined immunobridging success criteria. Seroresponse for a participant was defined as achieving a ≥4-fold rise of neutralizing antibody concentration from baseline (before the first dose of the primary series in Study 6 and Study 4). These analyses are summarized in Table 36 and Table 37.
A descriptive analysis evaluated seroresponse rates using pre-booster neutralizing antibody concentration as baseline. Seroresponse rate was defined as at least a 4-fold rise relative to the pre-booster concentration for Study 6. For the Omicron BA.1 assay, the seroresponse rate using pre-booster baseline was 96.5 % (95% CI: 93.8%, 98.2%) and the difference from Study 4 SPIKEVAX primary series seroresponse rate was 11.6 % (95% CI: 7.9%, 15.2%). For the Original SARS-CoV-2 (D614G) assay, the seroresponse using pre-booster baseline was 93.7% (95% CI: 90.4%, 96.1%) and the difference from Study 4 SPIKEVAX primary series seroresponse rate was -5.8% (95% CI: -9.1%, -3.5%).
15 REFERENCES
-
1. Jain SS, Anderson SA, Steele JM, et al. Cardiac manifestations and outcomes of COVID-19 vaccine-associated myocarditis in the young in the USA: longitudinal results from the Myocarditis After COVID Vaccination (MACiV) multicenter study. Lancet. 2024;76:1-13. https://doi.org/10.1016/j.eclinm.2024.102809 -
2. Free RJ, Patel K, Taylor CA, et al. Hospitalization for COVID-19 and risk factors for severe disease among children: 2022-2024. Pediatrics. Published online July 3, 2025. doi:10.1542/peds.2025-072788
16 HOW SUPPLIED/STORAGE AND HANDLING
SPIKEVAX is supplied as follows:
For 6 months through 11 years of age
NDC 80777-113-87 Carton of 2 single-dose pre-filled syringes, each syringe containing 1 dose of 0.25 mL (NDC 80777-113-09)
NDC 80777-113-80 Carton of 10 single-dose pre-filled syringes, each syringe containing 1 dose of 0.25 mL (NDC 80777-113-09)
For 12 years of age and older
NDC 80777-112-88 Carton of 2 single-dose pre-filled syringes, each syringe containing 1 dose of 0.5 mL (NDC 80777-112-01)
NDC 80777-112-96 Carton of 10 single-dose pre-filled syringes, each syringe containing 1 dose of 0.5 mL (NDC 80777-112-01)
NDC 80777-112-93 Carton of 10 single-dose pre-filled syringes, each syringe containing 1 dose of 0.5 mL (NDC 80777-112-01). Each carton contains 5 blisters, and each blister contains two syringes. Use one syringe per dose.
During storage, minimize exposure to room light, and avoid exposure to direct sunlight and ultraviolet light.
Storage
Store frozen between -50°C to -15°C (-58°F to 5°F).
After thawing, SPIKEVAX may be stored refrigerated between 2°C to 8°C (36°F to 46°F) for up to 60 days or up to the expiration date printed on the carton, whichever comes first.
After thawing, SPIKEVAX may be stored between 8°C to 25°C (46°F to 77°F) for up to 12 hours.
Do not refreeze once thawed.
Thawed syringes can be handled in room light conditions.
Transportation of Thawed Syringes at 2°C to 8°C (36°F to 46°F)
Thawed pre-filled syringes can be transported at 2°C to 8°C (36°F to 46°F) in shipping containers qualified to maintain 2°C to 8°C (36°F to 46°F). Once thawed and transported at 2°C to 8°C (36°F to 46°F), pre-filled syringes should not be refrozen and should be stored at 2°C to 8°C (36°F to 46°F) until use.
17 PATIENT COUNSELING INFORMATION
Advise the vaccine recipient or caregiver to read the FDA-approved patient labeling.
Inform the vaccine recipient or caregiver of the potential benefits and risks of vaccination with SPIKEVAX.
Instruct the vaccine recipient or caregiver to report any adverse events to their healthcare provider or to the Vaccine Adverse Event Reporting System at 1-800-822-7967 and https://vaers.hhs.gov.
Manufactured for:
Moderna US, Inc.
Princeton, NJ 08540
©2025 ModernaTX, Inc. All rights reserved.
SPIKEVAX is a trademark of ModernaTX, Inc.
Patent(s): www.modernatx.com/patents
US Govt. License No. 2256
Revised: 8/2025
Information for Recipients and Caregivers
SPIKEVAX (pronounced SPĪK-văx)
(COVID-19 Vaccine, mRNA)
(2025-2026 Formula)
Please read this information sheet before getting SPIKEVAX. This summary is not intended to take the place of talking with your or your child’s healthcare provider. If you have questions or would like more information, please talk with your or your child’s healthcare provider.
What is SPIKEVAX?
SPIKEVAX is a vaccine to protect against COVID-19. SPIKEVAX is for people who are:
-
65 years of age and older, or -
6 months through 64 years of age at high risk for severe COVID-19.
Vaccination with SPIKEVAX may not protect all people who receive the vaccine.
SPIKEVAX does not contain SARS-CoV-2, the virus that causes COVID-19. SPIKEVAX cannot give you or your child COVID-19.
Who should not get SPIKEVAX?
You or your child should not get SPIKEVAX if you or your child had:
-
a severe allergic reaction after a previous dose of SPIKEVAX or a Moderna COVID-19 vaccine -
a severe allergic reaction to any ingredient of this vaccine (see What are the ingredients in SPIKEVAX?)
What should I tell the healthcare provider?
Tell the healthcare provider about all of your or your child’s medical conditions, including if you or your child:
-
have any allergies -
had a severe allergic reaction after receiving a previous dose of any COVID-19 vaccine -
have had myocarditis (inflammation of the heart muscle) or pericarditis (inflammation of the lining outside the heart) -
have a fever -
have a bleeding disorder or are on a blood thinner -
are immunocompromised or are on a medicine that affects your immune system -
are pregnant or plan to become pregnant -
are breastfeeding -
have received any other COVID-19 vaccine -
have ever fainted in association with an injection
How is SPIKEVAX given?
SPIKEVAX is given as an injection into the muscle.
What are the risks of SPIKEVAX?
Severe allergic reactions have occurred in some people who have received SPIKEVAX and Moderna COVID-19 vaccines. There is a very small chance that SPIKEVAX could cause a severe allergic reaction. A severe allergic reaction would usually occur within a few minutes to 1 hour after getting a dose of SPIKEVAX. For this reason, the healthcare provider may ask you or your child to stay for a short time at the place where you or your child received the vaccine. Signs of a severe allergic reaction can include:
-
Trouble breathing -
Swelling of your face and throat -
A fast heartbeat -
A rash all over your body -
Dizziness and weakness
Myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the lining outside the heart) have occurred in some people who have received mRNA COVID-19 vaccines, including SPIKEVAX and Moderna COVID-19 vaccines. Myocarditis and pericarditis following administration of mRNA COVID-19 vaccines have occurred most commonly in males 12 years through 24 years of age. In most of these people, symptoms began within a week following vaccination. Based on available data, estimated rates of myocarditis and/or pericarditis from 1 through 7 days after getting a dose of the 2023-2024 Formula of mRNA COVID-19 vaccines were approximately 8 cases per million doses in people 6 months through 64 years of age and approximately 27 cases per million doses in males 12 years through 24 years of age.
In most people who have had myocarditis or pericarditis after receiving an mRNA COVID-19 vaccine, symptoms have gone away a few days after receiving treatment with medicines used to reduce inflammation.
In a study, follow-up information was collected on people who developed myocarditis after receiving the original formula of a COVID-19 vaccine; most people had received an mRNA COVID-19 vaccine. Some people in the study reported having heart symptoms approximately 3 months after developing myocarditis. Some people in the study had heart MRIs (scans that show detailed images of the heart muscle) initially after developing myocarditis and again approximately 5 months later. The initial and follow-up heart MRIs commonly showed signs of injury to the heart muscle, with improvement over time in most people. It is not known if these heart MRI findings might predict long-term heart effects of myocarditis. Studies are underway to find out if there are long-term heart effects in people who have had myocarditis after receiving an mRNA COVID-19 vaccine.
You should seek medical attention right away if you or your child has any of the following symptoms after receiving SPIKEVAX, particularly during the 2 weeks after receiving a dose of the vaccine:
-
Chest pain -
Shortness of breath -
Feelings of having a fast-beating, fluttering, or pounding heart
These could be symptoms of myocarditis or pericarditis.
Additional symptoms, particularly in children, may include:
-
Fainting -
Unusual and persistent irritability -
Unusual and persistent poor feeding -
Unusual and persistent fatigue or lack of energy -
Persistent vomiting -
Persistent pain in the abdomen -
Unusual and persistent cool, pale skin
Side effects that have been reported in clinical trials with SPIKEVAX and Moderna COVID-19 vaccines include:
-
Injection site reactions: pain, tenderness and swelling of the lymph nodes in the same arm of the injection or in the groin, swelling (hardness), and redness -
General side effects: fatigue, headache, muscle pain, joint pain, chills, nausea and vomiting, fever, rash, irritability/crying, sleepiness, and loss of appetite
Other side effects that have been reported include:
-
Severe allergic reactions -
Urticaria (itchy rash/hives) -
Myocarditis (inflammation of the heart muscle) -
Pericarditis (inflammation of the lining outside the heart) -
Fainting in association with injection of the vaccine -
Febrile seizures (convulsions during a fever)
These may not be all of the possible side effects of SPIKEVAX. Ask your healthcare provider about any side effects that concern you. You may report side effects to the Vaccine Adverse Event Reporting System (VAERS) at 1-800-822-7967 or https://vaers.hhs.gov.
What if I am or my child is pregnant or breastfeeding?
If you are or your child is pregnant or breastfeeding, discuss your options with your or your child’s healthcare provider.
What are the ingredients in SPIKEVAX?
SPIKEVAX contains the following ingredients:
-
messenger ribonucleic acid (mRNA) -
lipids (SM-102, polyethylene glycol [PEG] 2000 dimyristoyl glycerol [DMG], cholesterol, and 1,2-distearoyl-sn-glycero-3-phosphocholine [DSPC]) -
tromethamine -
tromethamine hydrochloride -
acetic acid -
sodium acetate trihydrate -
sucrose
SPIKEVAX does not contain preservatives.
If you would like more information, talk to your healthcare provider or visit www.spikevax.com or call 1-866-MODERNA (1-866-663-3762).
Manufactured for:
Moderna US, Inc.
Princeton, NJ 08540
©2025 ModernaTX, Inc. All rights reserved.
SPIKEVAX is a trademark of ModernaTX, Inc.
Patent(s): www.modernatx.com/patents
Revised: August 2025
Package/Label Principal Display Panel
COVID-19 Vaccine, mRNA
Spikevax
2025-2026 Formula
0.5 mL Single Dose
NDC 80777-112-01
Rx Only
For IM Use
Mfd. for: Moderna US, Inc.
Package/Label Principal Display Panel
NDC 80777-112-96
Moderna
Rx Only
COVID-19 Vaccine, mRNA
Spikevax
2025-2026 Formula
Injectable Suspension, for Intramuscular Use
10 Single-Dose Pre-Filled Syringes
Each dose is 0.5 mL
For 65 years and older
For 12 years through 64 years at high risk for severe COVID-19
Package/Label Principal Display Panel
NDC 80777-112-93
Moderna
Rx Only
COVID-19 Vaccine, mRNA
Spikevax
2025-2026 Formula
Injectable Suspension, for Intramuscular Use
10 Single-Dose Pre-Filled Syringes
Each dose is 0.5 mL
For 65 years and older
For 12 years through 64 years at high risk for severe COVID-19
Package/Label Principal Display Panel
NDC 80777-112-88
Moderna
Rx Only
COVID-19 Vaccine, mRNA
Spikevax
2025-2026 Formula
Injectable Suspension, for Intramuscular Use
2 Single-Dose Pre-Filled Syringes
Each dose is 0.5 mL
For 65 years and older
For 12 years through 64 years at high risk for severe COVID-19
Package/Label Principal Display Panel
COVID-19 Vaccine, mRNA
Spikevax
2025-2026 Formula
0.25 mL Single Dose
NDC 80777-113-09
Rx Only
For IM Use
Mfd. for: Moderna US, Inc.
Package/Label Principal Display Panel
NDC 80777-113-80
Moderna
Rx Only
COVID-19 Vaccine, mRNA
Spikevax
2025-2026 Formula
Injectable Suspension, for Intramuscular Use
10 Single-Dose Pre-Filled Syringes
Each dose is 0.25 mL
For 6 months through 11 years at high risk for severe COVID-19
Package/Label Principal Display Panel
NDC 80777-113-87
Moderna
Rx Only
COVID-19 Vaccine, mRNA
Spikevax
2025-2026 Formula
Injectable Suspension, for Intramuscular Use
2 Single-Dose Pre-Filled Syringes
Each dose is 0.25 mL
For 6 months through 11 years at high risk for severe COVID-19
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.