OMNIPAQUE (Iohexol) injection, solution
WARNING: RISKS WITH INADVERTENT INTRATHECAL ADMINISTRATION
See full prescribing information for complete boxed warning
Inadvertent intrathecal administration may cause death, convulsions/seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema [see Contraindications ( 4 ) and Warnings and Precautions ( 5.1 )].
WARNING: RISKS WITH INADVERTENT INTRATHECAL ADMINISTRATION OF OMNIPAQUE injection 140 and 350 mg iodine/mL
See full prescribing information for complete boxed warning.
Inadvertent intrathecal administration may cause death, convulsions/seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema ( 4 , 5.1 ).
1 INDICATIONS AND USAGE
OMNIPAQUE (iohexol) injection is a radiographic contrast agent indicated for intrathecal, intravascular, oral, rectal, intraarticular and body cavity use. OMNIPAQUE oral solution is indicated for oral use only in conjunction with OMNIPAQUE injection administered intravenously for computed tomography (CT) of the abdomen ( 1).
1.1 Intrathecal Administration
Adults
OMNIPAQUE 180, 240, and 300
- Myelography (lumbar, thoracic, cervical, total columnar)
- Computerized Tomography (CT) (myelography, cisternography, ventriculography)
Pediatrics
OMNIPAQUE 180
- Myelography (lumbar, thoracic, cervical, total columnar)
- CT (myelography, cisternography)
1.2 Intravascular Administration
Adults
OMNIPAQUE 140
- Intra-arterial digital subtraction angiography of the head, neck, abdominal, renal and peripheral vessels
OMNIPAQUE 240
- CT head imaging
- Peripheral venography (phlebography)
OMNIPAQUE 300
- Aortography including studies of the aortic arch, abdominal aorta and its branches
- CT head and body imaging
- Cerebral arteriography
- Peripheral venography (phlebography)
- Peripheral arteriography
- Excretory urography
OMNIPAQUE 350
- Angiocardiography (ventriculography, selective coronary arteriography)
- Aortography including studies of the aortic root, aortic arch, ascending aorta, abdominal aorta and its branches
- CT head and body imaging
- Intravenous digital subtraction angiography of the head, neck, abdominal, renal and peripheral vessels
- Peripheral arteriography
- Excretory urography
Pediatrics
OMNIPAQUE 240
- CT head and body imaging
OMNIPAQUE 300
- Angiocardiography (ventriculography)
- Excretory urography
- CT head and body imaging
OMNIPAQUE 350
- Angiocardiography (ventriculography, pulmonary arteriography, venography, and studies of the collateral arteries)
- Aortography including the aortic root, aortic arch, ascending and descending aorta
1.3 Oral or Rectal Administration
Adults
OMNIPAQUE 350
- Oral radiographic examination of the gastrointestinal tract
Pediatrics
OMNIPAQUE 180, 240 and 300
- Oral and rectal radiographic examination of the gastrointestinal tract
1.4 Oral Administration in Conjunction with Intravenous Administration
Diluted OMNIPAQUE Injection
Adults
OMNIPAQUE 240, 300 and 350 diluted and administered orally in conjunction with OMNIPAQUE 300 administered intravenously
- CT of the abdomen
Pediatrics
OMNIPAQUE 240, 300 and 350 diluted and administered orally in conjunction with OMNIPAQUE 240 or OMNIPAQUE 300 administered intravenously
- CT of the abdomen
OMNIPAQUE Oral Solution
Adults
OMNIPAQUE oral solution 9 and 12 administered orally in conjunction with OMNIPAQUE 300 administered intravenously
- CT of the abdomen
Pediatrics
OMNIPAQUE oral solution 9 and 12 administered orally in conjunction with OMNIPAQUE 240 or OMNIPAQUE 300 administered intravenously
- CT of the abdomen
1.5 Intraarticular Administration
Adults
OMNIPAQUE 240, 300, and 350
- Arthrography
1.6 Body Cavity Administration
Adults
OMNIPAQUE 240
- Endoscopic retrograde pancreatography (ERP) and cholangiopancreatography (ERCP)
- Herniography
- Hysterosalpingography
OMNIPAQUE 300
- Hysterosalpingography
Pediatrics
OMNIPAQUE 240, 300 and 350 diluted
- Voiding cystourethrography (VCU)
2 DOSAGE AND ADMINISTRATION
The concentration and volume required will depend on the indication, size and condition of the patient, and the equipment and imaging technique used. For CT of the head and body, OMNIPAQUE may be used with an automated contrast injection system or contrast media management system cleared for use with OMNIPAQUE. See full prescribing information for complete dosing information ( 2).
2.1 Important Dosage and Administration Instructions
- OMNIPAQUE 140, 180, 240, 300 and 350 are indicated for intravascular, oral, rectal, intraarticular, and body cavity administration. OMNIPAQUE 180, 240, and 300 are indicated for intrathecal administration [ see Boxed Warning , Contraindications (4), and Warnings and Precautions (5.1)].
- Use sterile technique for all handling and administration of OMNIPAQUE for intravascular, intrathecal, intraarticular, and body cavity administration.
- OMNIPAQUE oral solution 9 and 12 are indicated for oral use only [ see Contraindications (4)and Warnings and Precautions (5.2)].
- Do not use if tamper-evident ring is broken or missing.
- OMNIPAQUE injection may be administered at either body (37°C, 98.6°F) or room temperature (20° to 25°C, 68° to 77°F).
- Inspect OMNIPAQUE injection for particulate matter or discoloration before administration, whenever solution and container permit. Do not administer if OMNIPAQUE injection contains particulate matter or is discolored.
- Do not mix OMNIPAQUE injection with, or inject in intravenous lines containing, other drugs or total nutritional admixtures.
- Use the lowest dose necessary to obtain adequate visualization.
- Individualize the volume, strength, and rate of administration of OMNIPAQUE injection. Consider factors such as age, body weight, vessel size, blood flow rate within the vessel, anticipated pathology, degree and extent of opacification required, structures or area to be examined, disease processes affecting the patient, and equipment and technique to be employed.
- Avoid extravasation when administering OMNIPAQUE injection intravascularly, especially in patients with severe arterial or venous disease [ see Warnings and Precautions (5.6) ] .
- Hydrate patients before and after intravascular administration of OMNIPAQUE injection [ see Warnings and Precautions (5.4) ] .
- Each bottle of OMNIPAQUE injection and oral solution is intended for one procedure only. Discard any unused portion.
2.2 Intrathecal Dosage and Administration
- Rate of injection: Injection should be made slowly over 1 to 2 minutes
- Repeat procedures: If sequential or repeat examinations are required, a suitable interval of time between administrations should be observed to allow for normal clearance of the drug from the body; at least 48 hours should be allowed before repeat examination; however, whenever possible, 5 days to 7 days is recommended.
- If computerized tomographic (CT) myelography follows myelography, delay imaging several hours to allow the degree of contrast to decrease.
2.3 Intravascular Dosage and Administration
Intra-arterial Procedures
Intravenous Procedures
2.4 Oral or Rectal Dosage and Administration
Oral and Rectal Administration – Undiluted OMNIPAQUE Injection for Radiographic Examination of the Gastrointestinal (GI) Tract
2.5 Oral Dosage and Administration in Conjunction with Intravenous Administration
See Table 16for concurrent intravenous dosing.
Oral Administration of Diluted OMNIPAQUE Injection in Conjunction with Intravenous Administration of OMNIPAQUE Injection for CT of the Abdomen
Oral Administration of OMNIPAQUE Oral Solution in Conjunction with Intravenous Administration of OMNIPAQUE Injection for CT of the Abdomen
2.6 Intraarticular Dosage and Administration
2.7 Body Cavity Dosage and Administration
Body Cavity Administration - Undiluted OMNIPAQUE Injection
Body Cavity Administration - Diluted OMNIPAQUE Injection
2.8 Instructions for Use with an Automated Contrast Injection System or Contrast Management System for CT of the Head and Body
- OMNIPAQUE may be used with an automated contrast injection system cleared for use with contrast media.
- See above Important Dosage and Administration Instructions for OMNIPAQUE (2.1).
- See device labeling for information on device indications, instructions for use, and techniques to help assure safe use.
- OMNIPAQUE 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles may be used with a contrast media management system cleared for use with OMNIPAQUE 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles.
- See device labeling for information on device indications, instructions for use, and techniques to help assure safe use.
- Use sterile technique for penetrating the container closure of OMNIPAQUE 300 and 350 and transferring OMNIPAQUE solution. The container closure may be penetrated only one time with a suitable sterile component of the contrast media management system cleared for use with OMNIPAQUE 300 and 350 in 150 mL bottles.
- Once the OMNIPAQUE 300 and 350 Injection is punctured, do not remove the bottle from the work area during the entire period of use.
- Maximum use time is 4 hours after initial puncture.
- Each bottle is for one procedure only. Discard unused portion.
3 DOSAGE FORMS AND STRENGTHS
OMNIPAQUE Injection ( 3 )
- 140 mg of iodine per mL (302 mg of iohexol/mL) in +PlusPak™ polymer bottles
- 180 mg of iodine per mL (388 mg of iohexol/mL) in glass vials
- 240 mg of iodine per mL (518 mg of iohexol/mL), 300 mg of iodine per mL (647 mg of iohexol/mL) and 350 mg of iodine per mL (755 mg of iohexol/mL) in glass vials and bottles and +PlusPak™ polymer bottles
OMNIPAQUE Oral Solution ( 3 )
- 9 mg of iodine per mL (19 mg of iohexol/mL) and 12 mg of iodine per mL (26 mg of iohexol/mL) in +PlusPak™ polymer bottles
OMNIPAQUE (iohexol) Injection and Oral Solution
Sterile, pyrogen-free, gluten-free, colorless to pale yellow solution containing the nonionic, water-soluble x-ray contrast medium iohexol, and available in the following strengths and formats:
OMNIPAQUE (iohexol) Injection
- 140 mg of organically bound iodine per mL (302 mg iohexol/mL)
- Available in + PLUSPAK™ (polymer bottle)
- 180 mg of organically bound iodine per mL (388 mg iohexol/mL)
- Available in glass vials
- 240 mg of organically bound iodine per mL (518 mg iohexol/mL)
- 300 mg of organically bound iodine per mL (647 mg iohexol/mL)
- 350 mg of organically bound iodine per mL (755 mg iohexol/mL)
- Available in glass vials and bottles and + PLUSPAK™ polymer bottles.
OMNIPAQUE Oral Solution
- 9 mg of organically bound iodine per mL (19 mg iohexol/mL)
- 12 mg of organically bound iodine per mL (26 mg iohexol/mL)
- Available in + PLUSPAK™ polymer bottles.
4 CONTRAINDICATIONS
- OMNIPAQUE 140 and OMNIPAQUE 350 are contraindicated for intrathecal use [see Warnings and Precautions (5.1)] :
- OMNIPAQUE oral solution 9 and 12 are contraindicated for parenteral administration [see Warnings and Precautions (5.2)] :
- OMNIPAQUE body cavity 240 and 300 for hysterosalpingography is contraindicated during pregnancy or suspected pregnancy, menstruation or when menstruation is imminent, within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract including the external genitalia, and when reproductive tract neoplasia is known or suspected because of the risk of peritoneal spread of neoplasm.
- OMNIPAQUE injection 140 and 350 are contraindicated for intrathecal use ( 4 )
- OMNIPAQUE oral solution 9 and 12 are contraindicated for parenteral use ( 4)
- OMNIPAQUE body cavity 240 and 300 for hysterosalpingography is contraindicated during pregnancy (or suspected pregnancy), menstruation (or when menstruation is imminent), within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract, including the external genitalia, and when reproductive tract neoplasia is known or suspected. ( 4)
5 WARNINGS AND PRECAUTIONS
- Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency equipment and trained personnel available. ( 5.3)
- Contrast-Induced Acute Kidney Injury: Acute injury including renal failure can occur. Minimize dose and maintain adequate hydration to minimize risk. ( 5.4)
- Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after administration. ( 5.5)
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. ( 5.9)
5.1 Risks Associated with Inadvertent Intrathecal Administration
OMNIPAQUE injection 140 and 350 are contraindicated for intrathecal use [ see Contraindications (4) and Dosage and Administration (2.1) ] .Inadvertent intrathecal administration can cause death, convulsions/seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema.
5.2 Risks Associated with Inadvertent Parenteral Administration
OMNIPAQUE oral solution 9 and 12 are contraindicated for parenteral administration [ see Contraindications (4)and Dosage and Administration (2.1)]. Adverse reactions such as hemolysis may occur if administered intravascularly. Do not administer OMNIPAQUE oral solution 9 and 12 parenterally.
5.3 Hypersensitivity Reactions
OMNIPAQUE can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock. Most severe reactions develop shortly after the start of the injection (within 3 minutes), but reactions can occur up to hours later. There is an increased risk in patients with a history of a previous reaction to contrast agent, and known allergies (i.e., bronchial asthma, drug, or food allergies) or other hypersensitivities. Premedication with antihistamines or corticosteroids does not prevent serious life-threatening reactions, but may reduce both their incidence and severity.
Obtain a history of allergy, hypersensitivity, or hypersensitivity reactions to iodinated contrast agents and always have emergency resuscitation equipment and trained personnel available prior to OMNIPAQUE administration. Monitor all patients for hypersensitivity reactions.
5.4 Contrast-Induced Acute Kidney Injury
Acute kidney injury, including renal failure, may occur after parenteral administration of OMNIPAQUE. Risk factors include: pre-existing renal impairment, dehydration, diabetes mellitus, congestive heart failure, advanced vascular disease, elderly age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma/paraproteinaceous diseases, repetitive and/or large doses of an iodinated contrast agent.
Use the lowest necessary dose of OMNIPAQUE in patients with renal impairment. Adequately hydrate patients prior to and following parenteral administration of OMNIPAQUE. Do not use laxatives, diuretics, or preparatory dehydration prior to OMNIPAQUE administration.
5.5 Cardiovascular Adverse Reactions
Life-threatening or fatal cardiovascular reactions including hypotension, shock, cardiac arrest have occurred with the parenteral administration of OMNIPAQUE. Most deaths occur during injection or five to ten minutes later, with cardiovascular disease as the main aggravating factor. Cardiac decompensation, serious arrhythmias, and myocardial ischemia or infarction can occur during coronary arteriography and ventriculography.
Based upon clinical literature reported deaths from the administration of iodinated contrast agents range from 6.6 per million (0.00066%) to 1 in 10,000 (0.01%). Use the lowest necessary dose of OMNIPAQUE in patients with congestive heart failure and always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
5.6 Thromboembolic Events
Angiocardiography
Serious, rarely fatal, thromboembolic events causing myocardial infarction and stroke can occur during angiocardiography procedures with both ionic and nonionic contrast media. During these procedures, increased thrombosis and activation of the complement system occurs. Risk factors for thromboembolic events include: length of procedure, catheter and syringe material, underlying disease state, and concomitant medications.
To minimize thromboembolic events, use meticulous angiographic techniques, and minimize the length of the procedure. Avoid blood remaining in contact with syringes containing iodinated contrast agents, which increases the risk of clotting. Avoid angiocardiography in patients with homocystinuria because of the risk of inducing thrombosis and embolism.
5.7 Extravasation and Injection Site Reactions
Extravasation of OMNIPAQUE during intravascular injection may cause tissue necrosis and/or compartment syndrome, particularly in patients with severe arterial or venous disease. Ensure intravascular placement of catheters prior to injection. Monitor patients for extravasation and advise patients to seek medical care for progression of symptoms.
5.8 Thyroid Storm in Patients with Hyperthyroidism
Thyroid storm has occurred after the intravascular use of iodinated contrast agents in patients with hyperthyroidism, or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of OMNIPAQUE.
5.9 Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age
Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast media (ICM) in pediatric patients 0 to 3 years of age.
Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after ICM exposure. Pediatric patients with congenital cardiac conditions may be at the greatest risk given that they often require high doses of contrast during invasive cardiac procedures.
An underactive thyroid during early life may be harmful for cognitive and neurological development and may require thyroid hormone replacement therapy. After exposure to ICM, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates.
5.10 Hypertensive Crisis in Patients with Pheochromocytoma
Hypertensive crisis has occurred after the use of iodinated contrast agents in patient with pheochromocytoma. Monitor patients when administering OMNIPAQUE intravascularly if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of contrast necessary, assess the blood pressure throughout the procedure, and have measures for treatment of a hypertensive crisis readily available.
5.11 Sickle Cell Crisis in Patients with Sickle Cell Disease
Iodinated contrast agents when administered intravascularly may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following OMNIPAQUE administration and use OMNIPAQUE only if the necessary imaging information cannot be obtained with alternative imaging modalities.
5.12 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravascular contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agents; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering OMNIPAQUE to patients with a history of a severe cutaneous adverse reaction to OMNIPAQUE.
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks Associated with Inadvertent Intrathecal Administration [see Warnings and Precautions (5.1)]
- Risks Associated with Inadvertent Parenteral Administration [see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Contrast-Induced Kidney Injury [see Warnings and Precautions (5.4)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.5)]
- Thromboembolic Events [see Warnings and Precautions (5.6)]
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions (5.9)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.12)]
Most common adverse reactions (incidence ≥ 1.0%) in adult patients after OMNIPAQUE administration.( 6.1)
- Intrathecal: Headaches, Pain including backache, neckache, stiffness and neuralgia, nausea, vomiting and dizziness
- Intravascular: Pain, vision abnormalities (including blurred vision and photomas), headache, taste perversion, arrhythmias including premature ventricular contractions (PVCs) and premature atrial contractions (PACs), angina/chest pain, nausea
- Oral: Diarrhea, nausea, vomiting, abdominal pain, flatulence, headache
- Body Cavity: Pain, swelling and heat sensation
Post-marketing adverse reactions ( 6.2 ):Hypersensitivity and manifestations like rash, pruritus, urticaria, and dyspnea, in addition chest pain, and swelling.
To report SUSPECTED ADVERSE REACTIONS, contact GE Healthcare at 1-800-654-0118 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Intrathecal Administration
Adults
Pediatric Patients
Intravascular Administration
Immediately following intravascular injection of contrast medium, a transient sensation of mild warmth is not unusual. Warmth is less frequent with OMNIPAQUE than with ionic contrast media.
Adults
In controlled clinical studies involving 1485 patients, the following adverse reactions occurred (Table 25).
Pediatric Patients
In controlled clinical studies involving 391 patients for pediatric angiocardiography, urography, and CT head imaging, adverse reactions following the use of OMNIPAQUE 240, 300, and 350 were generally similar in quality and frequency to those reported in adults (Table 26).
Oral Administration for Examination of the Gastrointestinal Tract
Adults
Nausea, vomiting, and diarrhea have been most frequently reported following orally administered undiluted OMNIPAQUE for radiographic examination of the gastrointestinal tract. In controlled clinical studies involving 54 adult patients for oral radiographic examination of the gastrointestinal tract using undiluted OMNIPAQUE 350 the following adverse reactions were reported (Table 27).
Pediatrics Patients (Oral and Rectal Administration)
In clinical studies involving 58 pediatric patients, the adverse reactions were found to mostly affect the gastrointestinal system with diarrhea (36%), vomiting (9%), nausea (5%) and abdominal pain (2%). However, fever (5%), hypotension (2%) and urticaria (2%) were also reported.
Oral Administration for CT of the Abdomen in Conjunction with Intravenous Administration
Adults
In a controlled clinical study involving 44 adult patients receiving oral administration of diluted OMNIPAQUE (4-9 mg iodine/mL) in conjunction with intravenously injected OMNIPAQUE 300 for CT examination of the abdomen, adverse reactions were limited to a single report of vomiting.
Pediatric Patients
In clinical studies involving 69 pediatric patients receiving oral administration of diluted OMNIPAQUE (9-29 mg iodine/mL) in conjunction with intravenously administered OMNIPAQUE 240 and OMNIPAQUE 300 for CT examination of the abdomen, adverse reactions were limited to a single report of vomiting (1.4%).
Body Cavity Use
Adults
Arthrography : In controlled clinical studies involving 285 adult patients for various body cavity examinations using OMNIPAQUE 240, 300 and 350, the most frequent adverse reactions were administration site reactions: pain 26% and swelling 22%, were exclusively reported for arthrography and were generally related to the procedure rather than the contrast medium. Patients also experienced heat (7%). All other adverse reaction occurred at a rate less than or equal to 1%.
Pediatric Patients
No adverse reactions associated with the use of OMNIPAQUE for VCU procedures were reported in 51 pediatric patients studied.
6.2 Post-marketing Experience
The following additional reactions listed by indication have been identified during post-approval use of OMNIPAQUE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
General
Immune System Disorders:Hypersensitivity reactions, anaphylactic or anaphylactoid reactions, anaphylactic or anaphylactoid shock including life-threatening or fatal anaphylaxis
General Disorders and Administration Site Conditions:Pyrexia, chills, pain and discomfort, asthenia, administration site conditions including extravasation
Intrathecal Administration
Nervous System Disorders:Meningism, aseptic meningitis, seizures or status epilepticus, disorientation, coma, depressed or loss of consciousness, transient contrast-induced toxic encephalopathy (including amnesia, hallucination, paralysis, paresis, speech disorder, aphasia, dysarthria), restlessness, tremors, hypoesthesia
Musculoskeletal and Connective Tissue Disorders:Pain, muscle spasms or spasticity
Psychiatric Disorders:Confusional state, agitation, anxiety
Eye Disorders:Transient visual impairment including cortical blindness
Renal Reactions:Acute kidney injury
Intravascular Administration
Cardiovascular Disorders:Severe cardiac complications (including cardiac arrest, cardiopulmonary arrest), shock, peripheral vasodilatation, palpitations, vasospasm including spasm of coronary arteries, myocardial infarction, syncope, cyanosis, pallor, flushing, chest pain
Hemodynamic Reactions:Vasospasm and thrombophlebitis following intravenous injection
Blood and Lymphatic System Disorders:Neutropenia
Nervous System Disorders:Disorientation, coma, depressed or loss of consciousness, transient contrast-induced toxic encephalopathy (including amnesia, hallucination, paralysis, paresis, speech disorder, aphasia, dysarthria), restlessness, tremors, hypoesthesia
Psychiatric Disorders:Confusional state, agitation
Eye Disorders:Eye irritation or itchiness, periorbital edema, ocular or conjunctival hyperemia, lacrimation
Renal Reactions:Acute kidney injury, toxic nephropathy (CIN), transient proteinuria, oliguria or anuria, increased serum creatinine
Gastrointestinal Disorders:Abdominal pain, pancreatitis aggravated, salivary gland enlargement
Endocrin e Reactions:Hyperthyroidism, hypothyroidism
Respiratory; Thoracic, and Mediastinal Disorders:Respiratory distress, respiratory failure, pulmonary edema, bronchospasm, laryngospasm, throat irritation, throat tightness, laryngeal edema, wheezing, chest discomfort, asthmatic attack
Skin and Subcutaneous Tissue Disorders:Contrast media reactions range from mild (e.g., pleomorphic rashes, drug eruption, erythema and skin discoloration, blisters, hyperhidrosis, angioedema, localized areas of edema) to severe: [e.g., Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), bullous or exfoliative dermatitis, acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS)]
Oral Administration
Gastrointestinal Disorders:Dysphagia, abdominal pain
Body Cavity Administration
Gastrointestinal Disorders:Pancreatitis
Musculoskeletal and Connective Tissue Disorders:Arthritis (arthrography)
Hysterosalpingography: Injection of OMNIPAQUE for hysterosalpingography is associated with immediate, transient pain. Monitor injection pressure and volume instilled to minimize pain and to avoid disruptive distention of the uterus and fallopian tubes Fluoroscopic monitoring is recommended.
Nervous system: Pain (49%), somnolence and fever each with an individual incidence of 3%
Gastrointestinal system: Nausea (3%)
7 DRUG INTERACTIONS
7.1 Drug-Drug Interactions
Metformin
In patients with renal impairment, metformin can cause lactic acidosis. Iodinated contrast agents appear to increase the risk of metformin-induced lactic acidosis, possibly as a result of worsening renal function. Stop metformin at the time of, or prior to, OMNIPAQUE administration in patients with an eGFR between 30 and 60 mL/min/1.73 m 2; in patients with a history of hepatic impairment, alcoholism or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and reinstitute metformin only after renal function is stable.
Radioactive Iodine
Administration of iodinated contrast agents may interfere with thyroid uptake of radioactive iodine (I-131 and I-123) and decrease therapeutic and diagnostic efficacy in patients with carcinoma of the thyroid. The decrease in efficacy lasts for 6 to 8 weeks.
Beta-adrenergic Blocking Agents
The use of beta-adrenergic blocking agents lowers the threshold for and increases the severity of contrast reactions and reduces the responsiveness of treatment of hypersensitivity reactions with epinephrine. Because of the risk of hypersensitivity reactions, use caution when administering OMNIPAQUE to patients taking beta-blockers.
Drugs that Lower Seizure Threshold
Drugs that lower seizure threshold, especially phenothiazine derivatives including those used for their antihistaminic or antinauseant properties, are not recommended for use with intrathecal administration of OMNIPAQUE.
CNS Active Drugs
Drugs such as monoamine oxidase (MAO) inhibitors, tricyclic antidepressants, CNS stimulants, psychoactive drugs described as analeptics, major tranquilizers, or antipsychotic drugs. Such medications should be discontinued at least 48 hours before myelography, should not be used for the control of nausea or vomiting during or after myelography, and should not be resumed for at least 24 hours post procedure. In non-elective procedures in patients on these drugs, consider prophylactic use of anticonvulsants.
7.2 Drug-Laboratory Test Interactions
Effect on Thyroid Tests
If iodine-containing isotopes are to be administered for the diagnosis of thyroid disease, the iodine-binding capacity of thyroid tissue may be reduced for up to 2 weeks after contrast medium administration. Thyroid function tests that do not depend on iodine estimation, e.g., T 3resin uptake or direct thyroxine assays, are not affected.
8 USE IN SPECIFIC POPULATIONS
- Lactation: A lactating woman may pump and discard breast milk for 10 hours after OMNIPAQUE administration. ( 8.2)
8.1 Pregnancy
Risk Summary
Hysterosalpingography is contraindicated in pregnant women due to the potential risk to the fetus from an intrauterine procedure [see Contraindications (4)]. There are no data with iohexol use in pregnant women to inform any drug-associated risks. Iohexol crosses the placenta and reaches fetal tissues in small amounts (see Data ). In animal reproduction studies, no developmental toxicity occurred with intravenous iohexol administration to rats and rabbits at doses up to 0.4 (rat) and 0.5 (rabbit) times the maximum recommended human intravenous dose ( see Data ).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Literature reports show that intravenously administered iohexol crosses the placenta and is visualized in the digestive tract of exposed infants after birth.
Animal Data
Iohexol was neither embryotoxic nor teratogenic in either rats or rabbits at the following dose levels tested: 1.0, 2.0, 4.0 g iodine/kg in rats, administered intravenously to 3 groups of 25 dams once daily during days 6 through 15 of pregnancy; 0.3, 1.0, 2.5 g iodine/kg in rabbits, administered intravenously to 3 groups of 18 rabbits dosed once a day during days 6 through 18 of pregnancy.
8.2 Lactation
Risk Summary
Published literature reports that breast feeding after intravenous iohexol administration to the mother would result in the infant receiving an oral dose of approximately 0.7% of the maternal intravenous dose; however, lactation studies have not been conducted with oral, intrathecal, or intracavity administration of iohexol. There is no information on the effects of the drug on the breastfed infant or on milk production. Iodinated contrast agents are excreted unchanged in human milk in very low amounts with poor absorption from the gastrointestinal tract of a breastfed infant. Exposure to iohexol to a breastfed infant can be minimized by temporary discontinuation of breastfeeding (see Clinical Considerations). The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for OMNIPAQUE and any potential adverse effects on the breastfed infant from OMNIPAQUE or from the underlying maternal condition.
Clinical Considerations
Interruption of breastfeeding after exposure to iodinated contrast agents is not necessary because the potential exposure of the breastfed infant to iodine is small. However, a lactating woman may consider interrupting breastfeeding and pumping and discarding breast milk for 10 hours (approximately 5 elimination half-lives) after OMNIPAQUE administration to minimize drug exposure to a breastfed infant.
8.4 Pediatric Use
Intrathecal Use
The safety and effectiveness of OMNIPAQUE 180 have been established in pediatric patients 2 weeks to 17 years of age for myelography (lumbar, thoracic, cervical, total columnar) and for CT (myelography, cisternography). Use of OMNIPAQUE 180 is supported by controlled clinical studies in adults for myelography, in addition to clinical studies in pediatric patients undergoing myelography. The safety and effectiveness of OMNIPAQUE 180 have not been established for intrathecal use in patient pediatric patients less than 2 weeks of age. The safety and effectiveness of OMNIPAQUE 240 and 300 have not been established in pediatric patients for myelography (lumbar, thoracic, cervical, total columnar) and for CT (myelography, cisternography, or ventriculography).
Intravascular Use
Angiocardiography( Ventriculography, Pulmonary Arteriography, Venography, and Studies of the Collateral Arteries) and Aortography
The safety and effectiveness of OMNIPAQUE 300 have been established in pediatric patients from birth to 17 years of age for angiocardiography (ventriculography) and of OMNIPAQUE 350 in pediatric patients from birth to 17 years of age for angiocardiography (ventriculography, pulmonary arteriography, venography, and studies of the collateral arteries) and aortography. Use of OMNIPAQUE 300 and 350 is supported by controlled clinical studies in adults for angiocardiography and aortography, in addition to controlled clinical studies in pediatric patients undergoing angiocardiography, including aortography. The safety and effectiveness of OMNIPAQUE 300 have not been established in pediatric patients for aortography.
Intra-arterial Digital Subtraction Angiography, Intravenous Digital Subtraction Angiography, Cerebral Arteriography, or Peripheral Arteriography and Venography
The safety and effectiveness of OMNIPAQUE have not been established in pediatric patients for intra-arterial digital subtraction angiography, intravenous digital subtraction angiography, cerebral arteriography, or peripheral arteriography and venography.
CT of the Head and Body
The safety and effectiveness of OMNIPAQUE 240 and 300 have been established in pediatric patients from birth to 17 years of age for CT imaging of the head and body. Use of OMNIPAQUE 240 and 300 is supported by controlled clinical studies in adults for head and body CT, in addition to clinical studies in pediatric patients undergoing head CT and in 69 pediatric patients undergoing CT of the abdomen after oral administration of diluted OMNIPAQUE plus intravenous administration of OMNIPAQUE. The safety and effectiveness of OMNIPAQUE 350 have not been established in pediatric patients for CT imaging of the head and body.
Urography
The safety and effectiveness of OMNIPQUE 300 have been established in pediatric patients from birth to 17 years of age for urography. Use of OMNIPAQUE 300 is supported by controlled clinical studies in adults for urography, in addition to controlled clinical studies in pediatric patients undergoing urography and clinical safety data in pediatric patients down to birth.
Oral or Rectal Use
Undiluted OMNIPAQUE Injection
The safety and effectiveness of OMNIPAQUE 180, 240, and 300 administered orally and rectally have been established in pediatric patients, from birth to 17 years of age for examination of the GI tract. Use of OMNIPAQUE 180, 240, and 300 administered orally and rectally is supported by controlled studies in adults for examination of the GI tract, in addition to clinical studies in pediatric patients undergoing examination of the GI tract.
Oral Use in Conjunction with Intravenous Use
Diluted OMNIPAQUE Injection
The safety and effectiveness of OMNIPAQUE injection diluted to concentrations from 9 to 21 mg iodine/mL administered orally in conjunction with OMNIPAQUE injection administered intravenously for CT of the abdomen have been established in pediatric patients from birth to 17 years of age. Use is supported by clinical trials in adults, in addition to clinical studies in 69 pediatric patients undergoing CT of the abdomen after oral administration of diluted OMNIPAQUE plus intravenous administration of OMNIPAQUE.
OMNIPAQUE Oral Solution
The safety and effectiveness of OMNIPAQUE oral solution 9 and 12 administered orally in conjunction with OMNIPAQUE injection administered intravenously for CT of the abdomen in pediatric patients have been established in pediatric patients from birth to 17 years of age. Use is supported by the data establishing safety and effectiveness for OMNIPAQUE injection diluted and administered orally in conjunction with OMNIPAQUE injection administered intravenously for CT of the abdomen in pediatric patients.
Intraarticular Use
The safety and effectiveness of OMNIPAQUE have not been established in pediatric patients for arthrography.
Body Cavity Use
OMNIPAQUE 240, 300, 350 diluted to concentrations from 50 mg iodine/mL to 100 mg iodine/mL is indicated for use in pediatric patients from birth to 17 years of age for voiding cystourethrography (VCU). The use for voiding cystourethrography is supported by clinical studies in 51 pediatric patients undergoing VCU. The safety and effectiveness of OMNIPAQUE have not been established in pediatric patients for ERCP, herniography, or hysterosalpingography.
In general, the frequency of adverse reactions in pediatric patients was similar to that seen in adults [ see Adverse Reactions (6.1) ]. Pediatric patients at higher risk of experiencing adverse events during contrast-medium administration may include those having asthma, a sensitivity to medication and/or allergens, congestive heart failure, a serum creatinine greater than 1.5 mg/dL or those less than 12 months of age.
Thyroid function tests indicative of thyroid dysfunction, characterized by hypothyroidism or transient thyroid suppression have been reported following iodinated contrast media administration in pediatric patients, including term and preterm neonates. Some patients were treated for hypothyroidism. After exposure to iodinated contrast media, individualize thyroid function monitoring in pediatric patients 0 to 3 years of age based on underlying risk factors, especially in term and preterm neonates [see Warnings and Precautions (5.9)and Adverse Reactions (6.2)].
8.5 Geriatric Use
In clinical studies of OMNIPAQUE for CT, 52/299 (17%) of patients were 70 and over. No overall differences in safety were observed between these patients and younger patients. Other reported clinical experience has not identified differences in response between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. In general, dose selection for an elderly patient should be cautious usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
10 OVERDOSAGE
10.1 Intravascular Administration
The adverse effects of overdosage are life-threatening and affect mainly the pulmonary and cardiovascular systems. The symptoms included: cyanosis, bradycardia, acidosis, pulmonary hemorrhage, convulsions, coma, and cardiac arrest. Treatment of an overdosage is directed toward the support of all vital functions, and prompt institution of symptomatic therapy. Iohexol displays a low affinity for serum or plasma proteins and is poorly bound to serum albumin and can be dialyzed.
11 DESCRIPTION
11.1 Chemical Characteristics
OMNIPAQUE (iohexol) injection is a nonionic, x-ray or radiographic contrast medium for intrathecal, intravenous, oral, rectal and body cavity use. OMNIPAQUE oral solution is for oral use only.
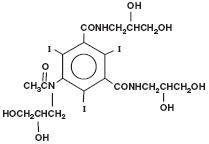
OMNIPAQUE injection and OMNIPAQUE oral solution are both provided as sterile, pyrogen-free and gluten-free solutions. OMNIPAQUE injection and OMNIPAQUE oral solution are colorless to pale yellow solutions. The chemical name of iohexol is Bis(2,3-dihydroxypropyl)-5-[ N-(2,3-dihydroxypropyl)-acetamido]-2,4,6- triiodoisophthalamide with a molecular weight of 821.14 (iodine content 46.36%). Iohexol has the following structural formula:
OMNIPAQUE injection is available in five strengths:
- OMNIPAQUE 140 mg iodine/mL (302 mg of iohexol/mL): Each mL contains 140 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 180 mg iodine/mL (388 mg of iohexol/mL): Each mL contains 180 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 240 mg iodine/mL (518 mg of iohexol/mL): Each mL contains 240 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 300 mg iodine/mL (647 mg of iohexol/mL): Each mL contains 300 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 350 mg iodine/mL (755 mg of iohexol/mL): Each mL contains 350 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
OMNIPAQUE oral solution is available in two strengths:
- OMNIPAQUE oral solution 9 mg iodine/mL (19 mg of iohexol/mL): Each mL contains 9 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE oral solution 12 mg iodine/mL (26 mg of iohexol/mL): Each mL contains 12 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium hydroxide. OMNIPAQUE injection and OMNIPAQUE oral solution are sterilized by autoclaving and contain no preservatives.
11.2 Physical Characteristics
OMNIPAQUE injection and OMNIPAQUE oral solution have the following physical properties:
OMNIPAQUE 140, OMNIPAQUE 180, OMNIPAQUE 240, OMNIPAQUE 300, and OMNIPAQUE 350 have osmolalities from approximately 1.1 to 3.0 times that of plasma (285 mOsmol/kg water) or cerebrospinal fluid (301 mOsmol/kg water) as shown in the above table and are hypertonic under conditions of use.
OMNIPAQUE oral solution 9 and OMNIPAQUE oral solution 12 are hypotonic under conditions of use (see tableabove).
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The iodine atoms in iohexol provide attenuation of X-rays in direct proportion to the concentration of iohexol. Since concentration changes over time, iohexol provides time-dependent image contrast which may assist in visualizing body structures.
12.2 Pharmacodynamics
Intrathecal Administration
The initial concentration and volume of the contrast medium, in conjunction with patient manipulation and the volume of cerebrospinal fluid (CSF) into which the contrast medium is placed, will determine the extent of the contrast that can be achieved. Following intrathecal injection in conventional radiography, OMNIPAQUE 180, 240, and 300 will continue to provide contrast for at least 30 minutes. Slow diffusion of iohexol takes place throughout the CSF with subsequent absorption into the bloodstream. At approximately 1 hour following injection, contrast will no longer be sufficient for conventional myelography.
After administration into the lumbar subarachnoid space, computerized tomography shows the presence of contrast medium in the thoracic region in about 1 hour, in the cervical region in about 2 hours, and in the basal cisterns in 3 to 4 hours.
Intravascular Administration
Following intravascular administration of OMNIPAQUE, the degree of contrast enhancement is directly related to the iodine concentration of an administered dose; peak iodine blood concentrations occur immediately (15 seconds to 120 seconds) following rapid intravenous injection. The time to maximum contrast enhancement can vary, depending on the organ, from the time that peak blood iodine concentrations are reached to one hour after intravenous bolus administration. When a delay between peak blood iodine concentrations and peak contrast is present, it suggests that radiographic contrast enhancement is at least in part dependent on the accumulation of iodine containing agent within the lesion and outside the blood pool.
Oral Administration
Orally administered OMNIPAQUE produces visualization of the gastrointestinal tract. Less than 1% of orally administered iohexol is recovered in the urine, suggesting minimal amounts are absorbed from the normal gastrointestinal tract. This amount may increase in the presence of bowel perforation or bowel obstruction.
Intraarticular Administration
Visualization of the joint spaces can be accomplished by direct injection of contrast medium. For intraarticular cavities, the injected iohexol is absorbed into the surrounding tissue and subsequently absorbed into systemic circulation.
Body Cavity Administration
For most body cavities, the injected iohexol is absorbed into the surrounding tissue and subsequently absorbed into systemic circulation. Examinations of the uterus (hysterosalpingography) and bladder (voiding cystourethrography) involve the almost immediate drainage of contrast medium from the cavity upon conclusion of the radiographic procedure.
12.3 Pharmacokinetics
Following the intravenous administration of iohexol (between 500 mg iodine/kg to 1500 mg iodine/kg) to 16 adult human subjects, apparent first-order terminal elimination half-life was 12.6 hrs and total body clearance was 131 (98 to 165) mL/min. Clearance was not dose dependent.
Absorption
As evidenced by the amount recovered in urine, <1% of orally administered iohexol is absorbed from the normal gastrointestinal tract. This amount may increase in the presence of bowel perforation or bowel obstruction.
Distribution
In 16 adult subjects (receiving between 500 mg iodine/kg to 1500 mg iodine/kg intravenous iohexol) the plasma volume of distribution was165 (108 to 219) mL/kg.
In five adult patients receiving 16 mL to 18 mL of OMNIPAQUE (180 mg iodine/mL) by lumbar intrathecal injection the plasma volume of distribution was 559 (350 to 849) mL/kg.
Elimination
Metabolism
No significant metabolism, deiodination or biotransformation occurs.
Excretion
Following intravascular or intrathecal administration, iohexol is excreted unchanged by glomerular filtration. Approximately 90% of the intravenously injected iohexol dose is excreted within the first 24 hours. Following intravascular administration, peak urine concentration occurs in the first hour after injection.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed with iohexol to evaluate carcinogenic potential. Iohexol was not genotoxic in a series of studies, including the Ames test, the mouse lymphoma TK locus forward mutation assay, and a mouse micronucleus assay. Iohexol did not impair the fertility of male or female rats when repeatedly administered at intravenous dosages up to 4 g iodine/kg.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
The container closure system components (bottle, vial, stopper and cap) of OMNIPAQUE injection and OMNIPAQUE oral solution are not made with natural rubber latex.
16.2 Storage and Handling
Protect OMNIPAQUE glass vials and bottles and + PLUSPAK™ polymer bottles from light. Do not freeze. Discard any product that is inadvertently frozen, as freezing may compromise the closure integrity of the immediate container.
OMNIPAQUE Injection 140, 180, 240, 300 and 350
Store at controlled room temperature, 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. May be stored in a contrast media warmer for up to one month at 36° to 38°C (96.8° to 100.4°F).
OMNIPAQUE Oral Solution 9 and 12
Store between 0° and 30°C (32° to 86°F).
17 PATIENT COUNSELING INFORMATION
Hypersensitivity Reactions
Advise the patient concerning the risk of hypersensitivity reactions that can occur both during and after OMNIPAQUE administration. Advise the patient to report any signs or symptoms of hypersensitivity reactions during the procedure and to seek immediate medical attention for any signs or symptoms experienced after discharge [see Warnings and Precautions (5.3)]
Advise patients to inform their physician if they develop a rash after receiving OMNIPAQUE [see Warnings and Precautions (5.12)].
Contrast Induced Acute Kidney Injury
Advise the patient concerning appropriate hydration to decrease the risk of contrast induced acute kidney injury [ see Warnings and Precautions (5.4) ].
Extravasation
If extravasation occurs during injection, advise patients to seek medical care for progression of symptoms [ see Warnings and Precautions (5.7) ] .
Lactation
Advise a lactating woman that interruption of breastfeeding is not necessary. However, to avoid any exposure, a lactating woman may consider pumping and discarding breast milk for 10 hours after OMNIPAQUE administration [ see Use in Specific Populations (8.2)] .
Thyroid Dysfunction
Advise parents/caregivers about the risk of developing thyroid dysfunction after OMNIPAQUE administration. Advise parents/caregivers about when to seek medical care for their child to monitor for thyroid function [see Warnings and Precautions (5.9)].
NOVAPLUS ®
Distributed by GE Healthcare Inc., Marlborough, MA 01752 U.S.A.
Product of Norwegian Origin.
OMNIPAQUE is a trademark of GE HealthCare or one of its subsidiaries.
GE is a trademark of General Electric Company used under trademark license.
NOVAPLUS is a registered trademark of Vizient, Inc.
© 2023 GE HealthCare
PRINCIPAL DISPLAY PANEL - 50 mL Bottle Box Label - 1412
520-Y
Contains
10 x 50 mL
Bottles
NDC 0407-1412-38
Omnipaque™
(iohexol)
Injection
240
mgI/mL
50 mL
NOVAPLUS ®
Single-Dose Bottle.
Sterile Aqueous Solution
For Injection or Oral Use.
PRINCIPAL DISPLAY PANEL - 50 mL Bottle Box Label - 1413
530-Y
Contains
10 x 50 mL
Bottles
NDC 0407-1413-86
Omnipaque™
(iohexol)
Injection
300
mgI/mL
50 mL
NOVAPLUS ®
Single-Dose Bottle.
Sterile Aqueous Solution
For Injection or Oral Use.
PRINCIPAL DISPLAY PANEL - 50 mL Bottle Box Label - 1414
540-Y
Contains
10 x 50 mL
Bottles
NDC 0407-1414-82
Omnipaque™
(iohexol)
Injection
NOT FOR
INTRATHECAL
USE
350
mgI/mL
50 mL
NOVAPLUS ®
Single-Dose Bottle.
Sterile Aqueous Solution
For Injection or Oral Use.
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.