Design and created by Guideline Central in participation with the American Society of Addiction Medicine.

Benzodiazepine Tapering
American Society of Addiction Medicine
Publication Date: March 4, 2025
Key Points
Background
- Benzodiazepines (BZDs) are approved by the US Food and Drug Administration (FDA) to treat a wide range of conditions, including anxiety and panic disorders, social phobia, insomnia, and seizures and are commonly prescribed.
- Use of these medications is associated with increased risk for adverse events, including falls, motor vehicle accidents, cognitive impairment, delirium, overdose, and death, particularly when BZDs are used in combination with central nervous system (CNS) depressants such as alcohol or opioids.
- When BZDs are used regularly, abrupt discontinuation (i.e., stopping the medication without a taper) or precipitous dose decreases can lead to serious and potentially life-threatening withdrawal symptoms (See Table 3).
- Physical dependence is a biological phenomenon that develops in response to repeated use of a medication and is distinct from BZD use disorder. Nearly all patients who regularly use BZD for more than a month will develop physical dependence. Only 1.5% of people who use BZD meet criteria for BZD use disorder.
- While there are some limited indications for longer term BZD use (e.g., severe treatment-resistant generalized anxiety disorder (GAD) or bipolar spectrum disorders, complex seizure disorders, spasticity, sleep disorders involving abnormal movements), it is generally recommended to limit BZD use to 2-4 weeks
Key Takeaways
BZDs should not be discontinued abruptly in patients who are likely to be physically dependent on the medication and at risk for withdrawal (See Table 2); rather, their medication dosage should be tapered gradually over a period of time under clinical supervision.
The following are ten key takeaways of this Guideline for adult patients who have been taking BZDs regularly and may be at risk for physical dependencea:
- Clinicians should base clinical recommendations regarding continued BZD prescribing on ongoing assessment of the risks and benefits of continued BZD use as well as those of tapering/ discontinuation (See Table 1). Tapering is generally indicated when the risks of continuing BZD medication outweigh the benefits.
- Clinicians should conduct more frequent risk–benefit assessments of continued BZD prescribing for patients who:
- Are concomitantly taking opioid medication.
- Have a substance use disorder (SUD).
- Have additional risk factors for adverse effects, such as co-occurring physical conditions (e.g., obstructive sleep apnea) or mental health conditions (e.g., bipolar spectrum disorder).
- Clinicians should use caution if utilizing urine drug screen immunoassays for BZDs due to known limitations.
- Clinicians should consider the maternal–fetal dyad when assessing the risks and benefits of continued BZD prescribing in patients who are pregnant.
- Clinicians should taper BZDs in most older adults (i.e., ≥65 years) unless there are compelling reasons for continuation.
- Clinicians should conduct more frequent risk–benefit assessments of continued BZD prescribing for patients who:
- Clinicians should consider approaches to BZD tapering in collaboration with patients and their care partners utilizing shared decision-making strategies.
- Clinicians should not discontinue BZDs abruptly in patients who are likely to be physically dependent and at risk for withdrawal symptoms (See Recommendation 2).
- Physical dependence can develop within weeks and is heterogeneous across patients (See Table 2).
- Although most patients can complete BZD tapering in outpatient settings, clinicians should consider inpatient or medically managed residential care when patient presentation indicates significant risk that cannot be safely managed in outpatient care.
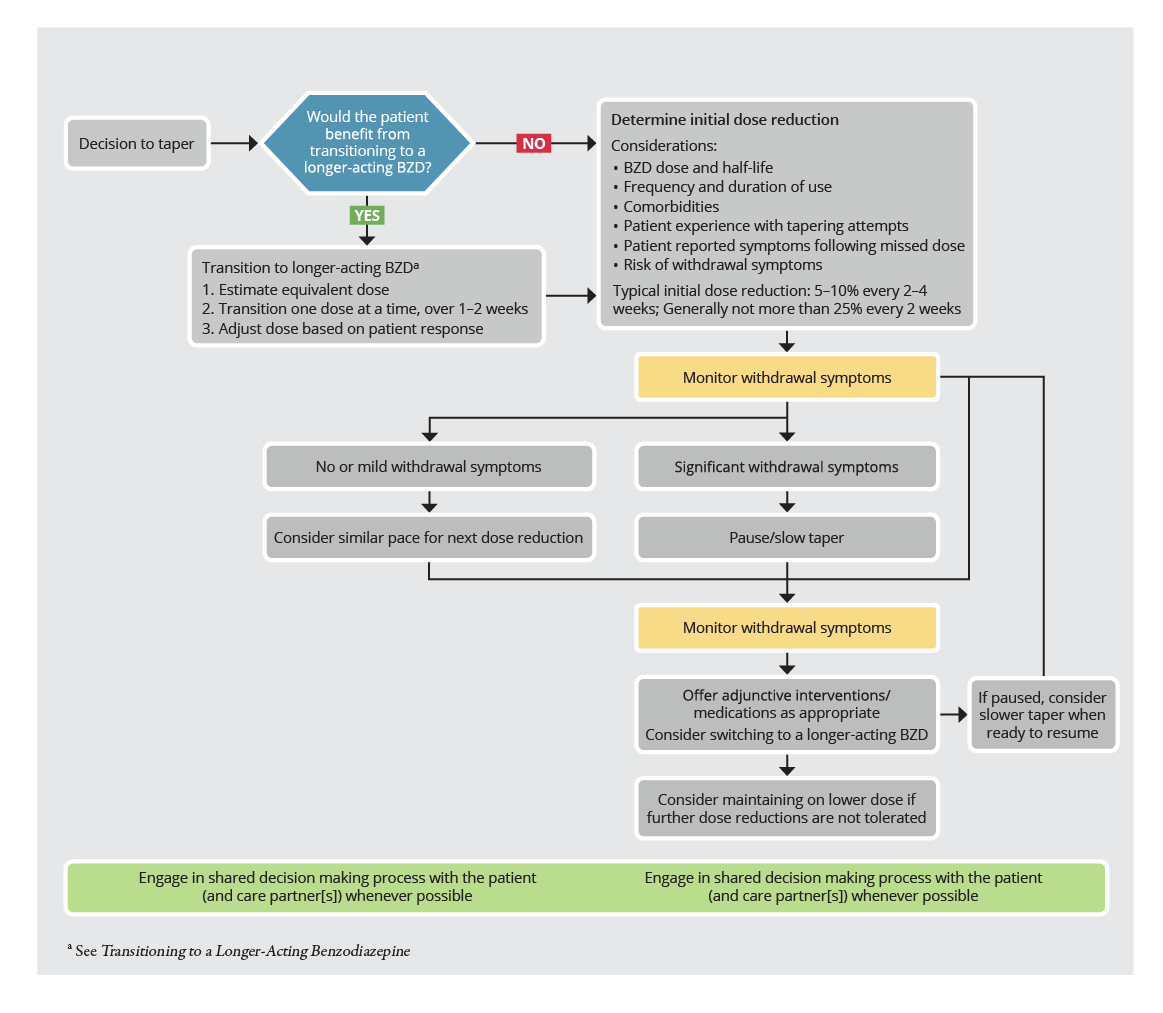
- Clinicians should design the tapering strategy to minimize harms from both continued BZD use and the tapering process, such as withdrawal symptoms and recurrence of symptoms for which the BZD was originally prescribed. The initial pace of the BZD taper should generally include dose reductions of 5% to 10% every 2–4 weeks. The taper should typically not exceed 25% every 2 weeks.
- Patients who have been taking lower doses for a relatively short period of time (e.g., <3 months) may be able to taper more quickly.
- The goal of tapering may be discontinuing the BZD medication or reducing the BZD dose to the point where the risks no longer outweigh the benefits.
- Clinicians should tailor tapering strategies to each individual patient and adjust tapering based on patient response.
- Clinicians can consider transitioning patients without contraindications to a comparable dose of a longer-acting BZD medication for the taper (See Benzodiazepine Dose Equivalents, Table 7).
- Clinicians should monitor patients for the emergence of BZD withdrawal signs and symptoms with each dose reduction. If significant signs or symptoms emerge, the taper should be slowed or paused (See Table 3).b
- Some patients experience significant withdrawal symptoms, even with gradual tapering, and should be offered slower tapering as needed.
- In some cases, maintainingc a patient on a lower dose may be sufficient to reduce the current risks such that they no longer outweigh the benefits.
- Clinicians should offer patients undergoing BZD tapering adjunctive psychosocial interventions (e.g., cognitive behavioral therapy [CBT], cognitive behavioral therapy for insomnia [CBT-I]) to support successful tapering (See Table 9 and Recommendation 10).
- Clinicians should provide concurrent treatment for any co-occurring physical health conditions and psychiatric disorders, including SUDs, that could interfere with the BZD taper.
- Clinicians should employ harm reduction strategies—such as providing opioid overdose reversal medication (e.g., naloxone) to those concomitantly taking opioids or otherwise at risk for opioid overdose, connecting patients to local resources, and providing patient education—based on each individual patient’s risks.
- It may take months to years to fully taper off BZDs, particularly if patients have been taking a high dose for an extended period of time.
b Pausing a BZD taper refers to temporarily halting dose reductions until withdrawal symptoms subside.
c Maintaining a BZD taper refers to discontinuing dose reductions with no plan to further reduce the dose.
Guiding Principles
- The recommendations in this Guideline are intended to support patient-centered care. Many complex factors influence decision making related to BZD tapering, and there is significant heterogeneity in patient response to tapering. This Guideline should be implemented to allow flexibility in response to diverse clinical circumstances.
- Healthcare systems, payers, policymakers, and clinicians should avoid misapplying this Guideline beyond its intended use in ways that may lead to unintentional harms for patients.
- Clinicians should develop tapering strategies collaboratively with patients, tailoring strategies to each patient’s risks, needs, and preferences and adjusting strategies based on a patient’s response.
- Healthcare systems and policymakers should carefully consider how to best leverage existing healthcare resources to meet the needs of the potentially large population for whom BZD tapering may be indicated.
- Physical dependence is an expected result of ongoing use of prescribed BZDs and distinct from SUD. Clinicians should not presume that patients with physical dependence have an SUD. Patients with SUD should be managed appropriately (see Recommendations for Patients with Benzodiazepine and Other Substance Use Disorders), with referrals for specialty treatment as necessary.
Recommendations
Considerations for Tapering Benzodiazepines
Table 1. Potential Benefits and Risks of Continued BZD Use and BZD Taperinga
| BZD use | BZD taper | |
|---|---|---|
| Potential benefits |
| |
| Potential risks |
|
|
a Clinicians should consider the likelihood of each benefit and risk for the individual patient. The narrative notes risk/hazard ratios available in the published literature.
b Including compassionate use for end of life or palliative care.
c Including risks associated with counterfeit BZDs from the illicit drug market, such as contamination with highly potent synthetic opioids (HPSOs) (e.g., fentanyl) and novel synthetic substances.
Partnering With Patients
Level of Care Considerations
Tapering Process
Adjunctive Interventions
Table 2. Risk for Clinically Significant BZD Withdrawala
| Duration of use | Frequency of use | Total daily dose | Risk for clinically significant withdrawalb |
|---|---|---|---|
| Any | ≤3 days per week | Any | Rarec |
| <1 month | ≥4 days per week | Any | Lower risk, but possible |
| 1–3 months | ≥4 days per week | Lowd | Lower risk, but possible |
| 1–3 months | ≥4 days per week | Moderate to highf | Yes, with greater risk with increasing dose and duration |
| ≥3 months | ≥4 days per week | Any | Yes, with greater risk with increasing dose and duration |
b Many factors influence the risk of physical dependence and withdrawal syndrome, including but not limited to age, co-occurring physical and mental health conditions, other substance use, and prior history of withdrawal.
c Half-lives are unknown for some novel synthetic benzodiazepines available in the illicit market.
d A low daily dose is estimated as 10 mg diazepam equivalents or less (e.g., ≤0.5 mg clonazepam, ≤2 mg lorazepam, ≤1 mg alprazolam). See Table 8 for dose equivalents.
e A moderate daily dose is estimated as 10–15 mg diazepam equivalents (e.g., 0.5–1.5 mg clonazepam, 2–3 mg lorazepam, 1–2 mg alprazolam). See Table 8 for dose equivalents.
f A high daily dose is estimated as more than 15 mg diazepam equivalents (e.g., >1.5 mg clonazepam, >3 mg lorazepam, >2 mg alprazolam). See Table 8 for dose equivalents.
Table 3. Common Benzodiazepine Withdrawal Signs and Symptomsa
- General
- Elevated blood pressure
- Headaches
- Sweating, night sweats
- Affective
- Anxiety, panic attacks
- Depression, dysphoria
- Irritability, agitation, aggression
- Cardiovascular
- Chest pain
- Palpitations
- Tachycardia
- Gastrointestinal
- Abdominal cramps
- Diarrhea
- Nausea and vomiting
- Neurological
- Cognitive impairment (e.g., poor memory, reduced concentration)
- Confusion, deliriumb
- Perceptual disturbance
- Seizuresb
- Sensory hypersensitivity (i.e., to light, sound, taste, and smell)
- Tingling, numbness, altered sensation
- Tinnitus
- Neuromuscular
- Akathisia, restlessness
- Depersonalization, derealization
- Psychosis (e.g., paranoia)b
- Suicidality and self-harm
- Neuropsychiatric
- Akathisia, restlessness
- Depersonalization, derealization
- Psychosis (e.g., paranoia)b
- Suicidality and self-harm
- Sleep
- Hypersomnia
- Insomnia
- Nightmares
b Typically associated with abrupt cessation of high doses of BZDs.
Patients Co-prescribed Benzodiazepines and Opioids
Patients with Benzodiazepine and Other Substance Use Disorders
Patients with Co-occurring Psychiatric Disorders
Older Adults
Patients Who Are Pregnant or Lactating
Table 4. Benzodiazepine Tapering Considerations by Pregnancy Trimester
| 1st trimester | 2nd trimester | 3rd trimester | Postpartum | |
|---|---|---|---|---|
| Potential Fetal Effects of BZDs | Minimal evidence of fetal malformations Increased risk of preterm birth | Increased risk of preterm birth, low birth weight, cesarean delivery, ventilatory support | Concern for withdrawal and potential fetal effects if high doses are used during lactation | |
| Potential Effects of Pregnancy on BZD Pharmacokinetics | Increased volume of distribution and CYP2C19, CYP3A4, and CYP2C9 metabolism (resulting in decreased effect) Decreased CYP1A2 and CYP2C19 activity | Increased volume of distribution and CYP2C19, CYP3A4, and CYP2C9 metabolism (resulting in decreased effect) Decreased CYP1A2 and CYP2C19 activity | Increased volume of distribution and CYP2C19, CYP3A4, and CYP2C9 metabolism (resulting in decreased effect) Decreased CYP1A2 and CYP2C19 activity | Reversal of pregnancy changes may increase effect |
| Causes of Insomnia | Nausea, urinary frequency, back pain | Fetal movements, heartburn, leg cramps, shortness of breath | Fetal movements, heartburn, leg cramps, shortness of breath | Infant care, pain |
| Considerations for Tapering BZDs | If alternative planned (e.g., SSRI) start alternative early to allow 6–8 weeks for effect before tapering BZD. Per above: BZD effect may decrease even before taper | Lowest dose possible to avoid neonatal withdrawal | Monitor sleep closely | |
| Alternative Medication for Insomnia | Diphenhydramine | Antihistamines, trazodone | Antihistamines, trazodone | |
| Alternative Medication for Acute Anxiety | Hydroxyzinea | Hydroxyzine | Hydroxyzine | Hydroxyzine |
| Alternative Medication for Severe Chronic Anxiety | SSRI | SSRI | SSRI | Sertraline has lowest relative infant dose (RID) |
b Possible increase in persistent pulmonary hypertension of the newborn, with a number needed to harm of 1,000.
Table 5. Relative Infant Dose (RID) of BZD Medications
| Medicationa | RID |
|---|---|
| Alprazolam | 2–9% |
| Chlordiazepoxide | Unknown |
| Clonazepam | 2.5–4.6% |
| Clorazepate | Unknown, shares metabolite with diazepam |
| Diazepam | Up to 11% |
| Estazolam | Unknown |
| Flurazepam | Unknown |
| Lorazepamc | 0.7–4.4% |
| Oxazepam | 10–33% |
| Quazepam | 0.2–2.5% |
| Temazepam | Dose dependent 0–10% |
| Triazolam | Unknown |
b For optimal safety, the target RID is <10%.
c Lorazepam is generally preferred in pregnancy and lactation due to its lack of active metabolites and low RID.
Management of Severe or Complicated Withdrawal
Table 6. Sample Residential (Level 3.7) Protocol for Phenobarbital Taper
- Day 1
- 64.8 mg initial dose and then 32.4 mg every 4 hours
- If the patient is experiencing significant withdrawal symptoms, clinicians may add a 32.4 mg dose
- 226.8 mg total scheduled daily dose, with a maximum total daily dose of 330 mg
- Day 2
- 32.4 mg every 4 hours
- If the patient is experiencing significant withdrawal symptoms, clinicians may add a 32.4 mg dose
- 194.4 mg total scheduled daily dose, with a maximum total daily dose of 300 mg
- Day 3
- 32.4 mg every 6 hours
- If the patient is experiencing significant withdrawal symptoms, clinicians may add a 32.4 mg dose
- 129.6 mg total scheduled daily dose, with a maximum total daily dose of 240 mg
- Day 4
- 32.4 mg every 8 hours
- If the patient is experiencing significant withdrawal symptoms, clinicians may add a 32.4 mg dose
- 97.2 mg total scheduled daily dose, with a maximum total daily dose of 180 mg
- Day 5
- 32.4 mg every 12 hours
- If the patient is experiencing significant withdrawal symptoms, clinicians may add a 32.4 mg dose
- 64.8 mg total scheduled daily dose, with a maximum total daily dose of 150 mg
- Day 6
- The patient may be discharged (or, for patients with SUD, transitioned to a less intensive level of addiction care) when the total daily dose is <60 mg within 24 hours
Sample Hospital (Level 4) Protocol for Phenobarbital Taper
Disclaimer: This is an example protocol that is currently used in a hospital setting for tapering with phenobarbital. It should not be interpreted as an exact recommended protocol.
Administer a test dose of 64.8 mg oral phenobarbital.
Assess patients 1 hour after administering the test dose to ensure they are not oversedated or intoxicated.
If patient tolerates the test dose, continue with the following phenobarbital taper schedule:
- 129.6 mg oral phenobarbital every 4 hours for a daily total of 6 doses
- 129.6 mg oral phenobarbital every 6 hours for a daily total of 4 doses
- 129.6 mg oral phenobarbital every 8 hours for a daily total of 3 doses
If patient exhibits any signs or symptoms of oversedation or intoxication, hold the next scheduled dose.
After 72 hours, patient should be safe for discharge (or, for patients with SUD, transitioned to a less intensive level of addiction care) without additional phenobarbital or BZD doses.
Additional Tables and Figures
Benzodiazepine Dose Equivalents
Table 7. Benzodiazepine Approximate Dose Equivalents to 10 mg Oral Diazepama
| Benzodiazepine | Anatomical Therapeutic Chemical classification system (ATC) Therapeutic Class | US Department of Veterans Affairs (VA)/US Department of Defense (DoD) SUD CPG (2021) | Ashton Manual (2002)b |
|---|---|---|---|
| Alprazolam | Anxiolytic | 1 | 0.5 |
| Chlordiazepoxide | Anxiolytic | 25 | 25 |
| Clonazepam | Antiepileptic | 1 | 0.5 |
| Clorazepate | Anxiolytic | 15 | 15 |
| Diazepam | Anxiolytic | 10 | 10 |
| Estazolam | Sedative–Hypnotic | 1 | 1 - 2 |
| Flurazepam | Sedative–Hypnotic | 15 | 15 - 30 |
| Lorazepam | Anxiolytic | 2 | 1 |
| Oxazepam | Anxiolytic | 30 | 20 |
| Quazepam | Sedative–Hypnotic | 10 | 20 |
| Temazepam | Sedative–Hypnotic | 15 | 20 |
| Triazolam | Sedative–Hypnotic | 0.25 | 0.5 |
b Same equivalents in Ashton, H. Benzodiazepine Equivalence Table [Online]. Revised April 2007. https://www.benzo.org.uk/bzequiv.htm and Ashton CH. The diagnosis and management of benzodiazepine dependence. Curr Opin Psychiatry. 2005;18(3):249–255. doi:10.1097/01.yco.0000165594.60434.84.
Conversion to diazepam equivalents is not straightforward and depends on patient factors such as age, metabolism, and other medications. Some patients may require higher doses than the reported equivalent, while others may require lower doses. Transition to an alternative BZD may be more successful if the doses are slowly transitioned over 1–2 weeks rather than 1–2 days.
Determining an equivalent BZD dose to begin tapering is complicated when patients are taking BZDs from the illicit drug market. In general, clinicians should titrate the BZD dose to the minimum dose necessary to control a patient’s withdrawal symptoms and taper from that point.
Table 8. Pharmacokinetic Properties of Benzodiazepines
| Benzodiazepine | Time to peak plasma level (h; via oral) | Relative lipid solubilitya | Onset of action (min)b | Elimination half-life (h)c | Metabolismd |
|---|---|---|---|---|---|
| Alprazolam | 1–2 h (tablet or orally disintegrating tablet [ODT]) 5–11 h extended release (XR) | Moderate | 15–30 | 6–12 | CYP3A4 |
| Chlordiazepoxide | 0.5–4 h | Moderate | 15–30 | 5–10 36–200 (AM) | CYP3A4 |
| Clonazepam | 1–2 h | Low | 15–30 | 18–50 | CYP3A4 |
| Clorazepatee | 0.5–2 h | High | 15 | CYP2C19 CYP3A4 | |
| Diazepam | 0.5–2 h | High | ≤15 | 20–100 36–200 (AM) | CYP1A2 CYP2C9 CYP2C19 CYP3A4 |
| Estazolam | 2 h | Low | 30–60 | 10–24 | CYP3A4 |
| Flurazepam | 0.5–2 h | High | ≤15 | 40–250 (AM) | CYP2C19 CYP3A4 |
| Lorazepam | 2–4 h | Moderate | 15–30 | 10–20 | Glucuronide conjugation |
| Oxazepam | 2–4 h | Low | 30–60 | 4–15 | Glucuronide conjugation |
| Quazepam | 2 h | High | 15 | 39 73 (AM) | CYP2C9 CYP2C19 CYP3A4 |
| Temazepam | 2–3 h | Moderate | 30–60 | 10–20 | Glucuronide conjugation |
| Triazolam | 1–2 h | Moderate | 15–30 | 1.5–5 | CYP3A4 |
a Increased lipid solubility results in more rapid onset of CNS activity but can also result in rapid redistribution into adipose tissue resulting in a shorter duration of action even in agents with long elimination half-life (e.g., diazepam)
b Rapid onset of action is associated with high lipid solubility and increased potential for misuse.
c Agents with moderate to high lipid solubility will have shorter duration of action with single or intermittent doses than suggested by the elimination half-life as these medications distribute rapidly into adipose tissue. With initial dosing, multiple daily doses may be needed to maintain effect. With chronic use and repeated dosing, accumulation is more likely to occur with these agents, especially those with long elimination half-lives (e.g., diazepam).
d Agents metabolized via glucuronide conjugation do not have pharmacokinetic interactions and are considered to be safer in older adults and patients with hepatic impairment.
e Hydrolized to nordiazepam in the stomach.
Table 9. Adjunctive Psychosocial Interventions
| Intervention | Brief description | Papers/resources | |
|---|---|---|---|
| Behavioral interventions | CBT | Cognitive behavioral therapy is a structured psychological treatment that helps to change thoughts, feelings, and behaviors, to treat a variety of problems. | CBT for panic (Otto et al [2010]; Otto et al [1993]; Spiegel et al [1994]) CBT for BZD withdrawal (O’Connor et al [2008]; Oude Voshaar et al [2003]) CBT for GAD (Gosselin et al [2006]) Digital CBT (Klein et al [2023]) |
| Behavioral interventions | CBT-I | Cognitive behavioral therapy for insomnia is a structured psychological treatment that helps to change thoughts, feelings, and behaviors that are contributing to insomnia. | Coteur et al (2022); Morin et al (2004); Baillargeon et al (2003) |
| Behavioral interventions | Behavior modification | Behavior modification is a psychotherapeutic intervention used to eliminate or reduce unwanted behavior. | Pottie et al (2018) |
| Behavioral interventions | Mental health counseling | A variety of psychotherapy approaches are used in practice. Although CBT and behavior modification have the most evidence as adjunctive interventions for BZD withdrawal, other methods may be as or even more effective for specific patients. In general, any mental health provider who is comfortable addressing the reason for a patient’s initial BZD prescription and managing symptoms that may develop during the withdrawal process (e.g., anxiety, insomnia) will likely be helpful for patients. | American Counseling Association National Association of Social Workers National Alliance on Mental Illness |
| Lifestyle factors | Sleep hygiene | Sleep hygiene refers to environment and behaviors that are conducive to optimizing restorative sleep. These may include avoiding caffeine, stimulants, alcohol near bedtime. Along with setting up a night routine and sleep schedule that is conducive to good sleep. | Lähteenmäki et al (2013); Coteur et al (2023) |
| Lifestyle factors | Exercise and physical activity | Gentle exercise (e.g., walking or swimming) may be helpful. The Ashton Manual recommends regular moderate enjoyable exercise during a benzodiazepine taper. | Reconnexion. The Benzodiazepine Toolkit (2018:54) The Ashton Manual (2002) |
| Lifestyle factors | Diet | Staying well-hydrated, eating a well balanced diet, and eliminating caffeine (including energy drinks) and alcohol may be helpful. | Reconnexion. The Benzodiazepine Toolkit (2018:53) The Ashton Manual (2002) |
| Lifestyle factors | Mindfulness | Mindfulness is a cognitive skill, usually developed through meditation. | Barros et al (2022) |
| Complementary health approaches | Acupuncture | Yeung described acupuncture as the insertion of “fine needles at special acupoints on the body according to the traditional Chinese meridian theory. The inserted acupuncture needles can be connected by an electric-stimulator to deliver electric-stimulation and is termed as electroacupuncture.” | Electroacupuncture (Yeung et al [2019]) |
| Complementary health approaches | Progressive muscle relaxation | Progressive muscle relaxation involves alternately tensing then relaxing muscles, one by one. | Otto et al (2010) |
| Complementary health approaches | Anxiety management training (AMT) | In AMT, patients are “asked to imagine unpleasant events which they had experience, concentrate on early signs of distress and counteract them with relaxation.” | Elsesser et al (1996) |
| Peer specialist services | Peer support | Individuals who typically have lived experience in BZD tapering, mental health, and/or substance use provide support one-on-one or in group settings, either in person or virtually, to support people going through BZD tapering. | National Institutes for Health and Care Excellence (2022) Lynch et al (2022) |
Table 10. Medications for Anxiety-Related Symptomsa
| Medicationa | Class / mechanism | Considerations for useb | Other population considerations | |
|---|---|---|---|---|
| Acute anxiety | Clonidineb | Central alpha-2 agonist | Monitor blood pressure; avoid in hypotension If used as a scheduled medication, taper to discontinue | |
| Acute anxiety | Gabapentinb | Gamma aminobutyric acid (GABA) analogue | Risk for misuse Risk associated with combining with other medications, particularly opioids | Avoid in patients with history of sedative use disorder |
| Acute anxiety | Hydroxyzinec | Antihistamine | Avoid in patients with history of QTc prolongation | Avoid in older adults |
| Acute anxiety | Propranololb | Beta-blocker | Contraindicated in bradycardia, greater than first-degree block; avoid in uncontrolled bronchial asthma May be scheduled or dosed as needed for situational anxiety | |
| Chronic anxiety (GAD, panic, PTSD, social anxiety) | Buspironed | 5HT1A receptor agonist | Not effective as a prn agent Only effective for GAD | |
| Chronic anxiety (GAD, panic, PTSD, social anxiety) | Selective serotonin reuptake inhibitors (SSRIs)e | Antidepressantf | May be anxiogenic upon initiation and dose increase; start at a low dose and titrate slowly Variable interactions with other medications | |
| Chronic anxiety (GAD, panic, PTSD, social anxiety) | Serotonin norepinephrine reuptake inhibitor (SNRIs)e | Antidepressantf | May be anxiogenic upon initiation and dose increase; start at a low dose and titrate slowly May increase blood pressure Caution in uncontrolled hypertension | |
| Chronic anxiety (GAD, panic, PTSD, social anxiety) | Mirtazapinec | Serotonin and norepinephrine modulator | Not FDA approved for treatment of anxiety disorders May be anxiolytic upon initiation More sedating than SSRIs and SNRIs upon initiation | |
| Chronic anxiety (GAD, panic, PTSD, social anxiety) | Prazosinb | Central alpha-1 antagonist | Approved for hypertension; may be used off-label for PTSD related nightmares but not other symptoms of anxiety Monitor blood pressure; avoid in hypotension |
b Not FDA approved for anxiety disorders. The CGC noted that gabapentin has potential for misuse and, therefore, while it may be useful in certain circumstances, should not be considered prior to other potential adjunctive medications.
c FDA approved.
d FDA approved for GAD only.
e Variably approved for GAD, panic disorder, PTSD, and social anxiety disorder.
f Antidepressants have boxed warnings regarding suicidality, especially in adolescents and emerging adults
Table 11. Medications for Insomnia-Related Symptoms
| Medicationa | Class / mechanism | Considerations for useb | Other population considerations |
|---|---|---|---|
| Doxepinc | Antihistaminic tricyclic antidepressant | American Academy of Sleep Medicine (AASM) approved for sleep maintenance insomnia Avoid in patients with suicidal ideation and behavior due to risk for overdose | Caution in older adults, coronary artery disease, arrhythmia |
| Diphenhydramined | Antihistamine | AASM does not recommend for sleep onset or sleep maintenance insomnia | Avoid in older adults, may have paradoxical effects in children |
| Doxylamined | Antihistamine | Avoid in older adults, may have paradoxical effects in children | |
| Hydroxyzinee | Antihistamine | Avoid in patients with history of QTc prolongation | Avoid in older adults |
| Melatonind | Sedative- Hypnotic | AASM does not recommend for sleep onset or sleep maintenance insomnia | Avoid during pregnancy and breastfeeding; insufficient safety evidence |
| Ramelteonc | Agonist of melatonin receptors 1 and 2 | AASM approved for sleep onset insomnia Prone to significant interactions with cytochrome P450 (CYP) inhibitors and inducers | |
| Trazodonee | Antidepressant | Start with lower doses to avoid orthostasis in older adults AASM does not recommend for sleep onset or sleep maintenance insomnia | Use with caution in older adults |
b Use in individual patients should always include review of medical and medication history and individual prescribing information to assess for any relative/absolute contraindications.
c FDA approved.
d FDA approved, available over the counter.
e Not FDA approved for insomnia.
Figure 1. Determine Whether and Where to Taper
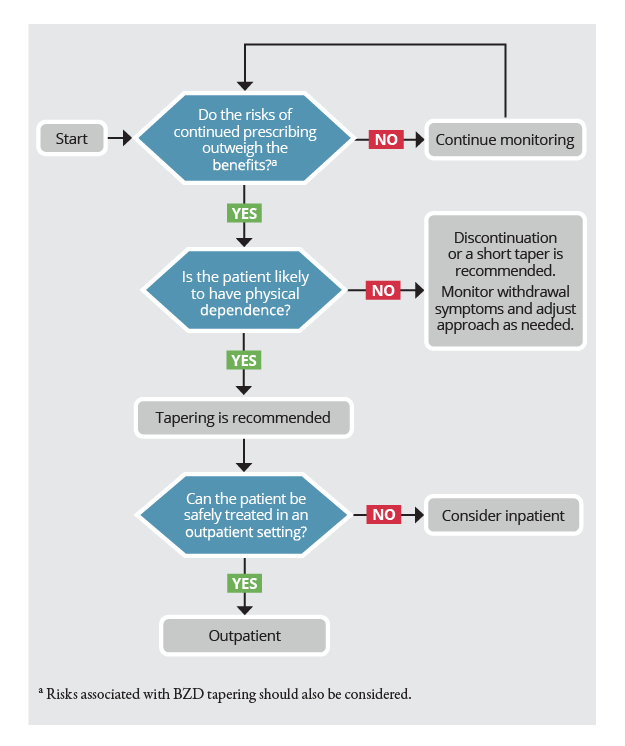
Figure 2. Taper Management — Outpatient
Recommendation Grading
| Certainty of Evidence | Strength of Recommendation | ||
|---|---|---|---|
| H | High certainty | S | Strong Recommendation |
| M | Moderate certainty | C | Conditional Recommendation |
| L | Low certainty | W | Weak Recommendation |
| VL | Very low certainty | ||
| CC | Clinical consensus |
Abbreviations
- AASM
American Academy of Sleep Medicine
- AMT
anxiety management training
- ATC
Anatomical Therapeutic Chemical classification system
- BZD
benzodiazepine(s)
- CBT
- cognitive behavioral therapy
- CBT-I
cognitive behavioral therapy for insomnia
- CDC
- US Centers for Disease Control and Prevention
- CGC
Clinical Guideline Committee
- CNS
- central nervous system
- CPG
- clinical practice guideline
- CYP
- cytochrome P450
- DoD
US Department of Defense
- FDA
- U.S. Food and Drug Administration
- GABA
- gamma-aminobutyric acid
- GAD
- generalized anxiety disorder
- GRADE
- Grading of Recommendations Assessment, Development, and Evaluation
- HPSO
highly potent synthetic opioid
- NSDUH
National Survey on Drug Use and Health
- ODT
orally disintegrating tablet
- PDMP
- Prescription Drug Monitoring Program
- PTSD
- posttraumatic stress disorder
- RID
relative infant dose
- SNRI
serotonin norepinephrine reuptake inhibitor
- SSRI
- selective serotonin reuptake inhibitor
- SUD
- substance use disorder
- VA
US Department of Veterans Affairs
- XR
extended release
Source Citation
The Joint Clinical Practice Guideline on Benzodiazepine Tapering. Available at: https://www.asam.org/quality-care/clinical-guidelines/benzodiazepine-tapering.
This clinical practice guideline has been endorsed by the American Academy of Neurology, the American Academy of Physician Associates, the American Association of Nurse Practitioners, the American Association of Psychiatric Pharmacists, the American College of Medical Toxicology, the American College of Obstetricians and Gynecology, the American Geriatrics Society, and the American Society of Addiction Medicine.
Expert Authors/Reviewers
Guideline Committee Members
Emily Brunner, MD, DFASAM (Chair); Chwen-Yuen A. Chen, MD, FACP, FASAM; Tracy Klein, PhD, FNP, ARNP, FAANP, FRE, FAAN; Donovan Maust, MD, MS; Maryann Mazer-Amirshahi, PharmD, MD, PhD, MPH, FACMT, FASAM; Marcia Mecca, MD; Deanna Najera, MPAS, MS, PA-C, DFAAPA; Chinyere Ogbonna, MD, MPH; Kiran F. Rajneesh, MD, MS, FAAN; Elizabeth Roll, MD; Amy E. Sanders, MD, MS, MPhil, FAAN; Brett Snodgrass, DNP, FNP-C, ACHPN, FAANP; Amy VandenBerg, PharmD, BCPP; Tricia Wright, MD, MS, FACOG, DFASAM
Disclaimer
This resource is for informational purposes only, intended as a quick-reference tool based on the cited source guideline(s), and should not be used as a substitute for the independent professional judgment of healthcare providers. Practice guidelines are unable to account for every individual variation among patients or take the place of clinician judgment, and the ultimate decision concerning the propriety of any course of conduct must be made by healthcare providers after consideration of each individual patient situation. Guideline Central does not endorse any specific guideline(s) or guideline recommendations and has not independently verified the accuracy hereof. Any use of this resource or any other Guideline Central resources is strictly voluntary.
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.