Design and created by Guideline Central in participation with the American College of Cardiology, American College of Clinical Pharmacy, American Heart Association, and Heart Rhythm Society.



Management of Patients with Atrial Fibrillation
American College of Cardiology
American Heart Association
Heart Rhythm Society
American College of Clinical Pharmacy
Publication Date: November 30, 2023
Overview
Top 10 Take-Home Messages for Atrial Fibrillation
- Stages of atrial fibrillation (AF): The previous classification of AF, which was based only on arrhythmia duration, although useful, tended to emphasize therapeutic interventions. The new proposed classification, using stages, recognizes AF as a disease continuum that requires a variety of strategies at the different stages, from prevention, lifestyle and risk factor modification (LRFM), screening, and therapy.
- AF risk factor modification and prevention: This guideline recognizes lifestyle and risk factor modification as a pillar of AF management to prevent onset, progression, and adverse outcomes. The guideline emphasizes risk factor management throughout the disease continuum and offers more prescriptive recommendations, accordingly, including management of obesity, weight loss, physical activity, smoking cessation, alcohol moderation, hypertension, and other comorbidities.
- Flexibility in using clinical risk scores and expanding beyond CHA2DS2-VASc for prediction of stroke and systemic embolism: Recommendations for anticoagulation are now made based on yearly thromboembolic event risk using a validated clinical risk score, such as CHA2DS2-VASc. However, patients at an intermediate annual risk score who remain uncertain about the benefit of anticoagulation can benefit from consideration of other risk variables to help inform the decision, or the use of other clinical risk scores to improve prediction, facilitate shared decision making, and incorporate into the electronic medical record.
- Consideration of stroke risk modifiers: Patients with AF at intermediate to low (<2%) annual risk of ischemic stroke can benefit from consideration of factors that might modify their risk of stroke, such as the characteristics of their AF (eg, burden), nonmodifiable risk factors (sex), and other dynamic or modifiable factors (blood pressure control) that may inform shared decision-making (SDM) discussions.
- Early rhythm control: With the emergence of new and consistent evidence, this guideline emphasizes the importance of early and continued management of patients with AF that should focus on maintaining sinus rhythm and minimizing AF burden.
- Catheter ablation of AF receives a Class 1 indication as first-line therapy in selected patients: Recent randomized studies have demonstrated the superiority of catheter ablation over drug therapy for rhythm control in appropriately selected patients. In view of the most recent evidence, we upgraded the Class of Recommendation.
- Catheter ablation of AF in appropriate patients with heart failure (HF) with reduced ejection fraction (EF) receives a Class 1 indication: Recent randomized studies have demonstrated the superiority of catheter ablation over drug therapy for rhythm control in patients with heart failure and reduced ejection failure. In view of the data, we upgraded the Class of Recommendation for this population of patients.
- Recommendations have been updated for device-detected AF: In view of recent studies, more prescriptive recommendations are provided for patients with device-detected AF that consider the interaction between episode duration and the patient's underlying risk for thromboembolism. This includes considerations for patients with AF detected via implantable devices and wearables.
- Left atrial appendage occlusion (LAAO) devices receive higher level Class of Recommendation: In view of additional data on safety and efficacy of left atrial appendage occlusion devices, the Class of Recommendation has been upgraded to 2a compared to the 2019 AF Focused Update for use of these devices in patients with long-term contraindications to anticoagulation.
- Recommendations are made for patients with AF identified during medical illness or surgery (precipitants): Emphasis is made on the risk of recurrent AF after AF is discovered during noncardiac illness or other precipitants, such as surgery.
2. Background and Pathophysiology
2.1. Epidemiology
- Atrial fibrillation is the most sustained common arrhythmia, and its incidence and prevalence are increasing in the United States and globally (Figure 1; Figure 2; Figure 3).
- AF is associated with a 1.5- to 2-fold increased risk of death.
Introduction
Colors in tables and figures correspond to Class of Recommendations and Level of Evidence tables.
Figure 1. Temporal Trends in Counts and Age-Standardized Rates of AF-Prevalent Cases by Social Demographic Index (SDI) Quintile for Both Sexes Combined, 1990 to 2017

SDI was made up of the geometric mean of three common indicators: the lag distributed income per capita, mean educational achievement for those aged 15 years or older, and total fertility rate under 25 years. SDI ranged from 0 to 1, where 0 represents the theoretical minimum level of development, whereas 1 represents the theoretical maximum level of development.
Modified from Dai H, et al. by permission of Oxford University Press on behalf of the European Society of Cardiology. Copyright 2020, Oxford University Press.
Figure 2. Prevalence of AF Among Medicare Beneficiaries, 1993–2007

Reproduced with permission from Piccini JP, et al. [Circulation: Cardiovascular Quality and Outcomes. 2012;5:85–93] Copyright 2012, American Heart Association, Inc.
Figure 2. Prevalence of AF Among Medicare Beneficiaries, 1993–2007 (cont'd)

Reproduced with permission from Piccini JP, et al. [Circulation: Cardiovascular Quality and Outcomes. 2012;5:85–93] Copyright 2012, American Heart Association, Inc.
Figure 3. Age-Standardized Global Prevalence Rates of AF and Atrial Flutter (AFL) per 100,000, Both Sexes, 2020

Modifed with permission from Tsao CW, et al. Copyright 2023, American Heart Association, Inc. Modifed Source: Institute for Health Metrics Evaluation. Used with permission. All rights reserved.
2.1.2. Risk Factors and Associated Heart Disease
Table 3. Risk Factors for Diagnosed AF
| Condition | Study type | Effect on risk of AF | Summary risk of incident AF | Effect of LRFM | |
|---|---|---|---|---|---|
| Risk factors | Advancing age |
|
| ↑ risk | Not applicable (N/A) |
| Risk factors | Advancing age |
|
| ↑ risk | Not applicable (N/A) |
| Risk factors | Smoking |
|
| ↑ risk | N/A |
| Risk factors | Smoking |
|
| ↑ risk | N/A |
| Risk factors | Smoking |
|
| ↑ risk | N/A |
| Risk factors | Physical activity |
|
| U curve: Sedentary lifestyle and elite/extreme exercise: ↑ risk | Exercise: ↓ AF burden, recurrence, symptoms; ↑ quality of life (QOL), functional capacity |
| Risk factors | Alcohol |
|
| ↑ risk |
|
| Risk factors | Alcohol |
|
| ↑ risk |
|
| Risk factors | Alcohol |
|
| ↑ risk |
|
| Risk factors | Adiposity markers: weight, body mass index (BMI), obesity |
|
| ↑ risk |
|
| Risk factors | Adiposity markers: weight, body mass index (BMI), obesity |
|
| ↑ risk |
|
| Risk factors | Adiposity markers: weight, body mass index (BMI), obesity |
|
| ↑ risk |
|
| Risk factors | Height |
|
| ↑ risk | N/A |
| Risk factors | Height |
|
| ↑ risk | N/A |
| Risk factors | Height |
|
| ↑ risk | N/A |
| Risk factors | Hypertension and blood pressure (BP) |
|
|
|
|
| Risk factors | Hypertension and blood pressure (BP) |
|
|
|
|
| Risk factors | Hypertension and blood pressure (BP) |
|
|
|
|
| Risk factors | Hypertension and blood pressure (BP) |
|
|
|
|
| Risk factors | Resting heart rate |
|
|
| N/A |
| Risk factors | Resting heart rate |
|
|
| N/A |
| Risk factors | Diabetes mellitus (DM) |
|
| ↑ risk |
|
| Risk factors | Diabetes mellitus (DM) |
|
| ↑ risk | Optimal glycemic control preablation may ↓ AF recurrence postablation |
| Risk factors | Diabetes mellitus (DM) |
|
| ↑ risk | Optimal glycemic control preablation may ↓ AF recurrence postablation |
| Cardiovascular disease | HF or coronary artery disease (CAD) |
|
| ↑ risk | N/A |
| Cardiovascular disease | HF |
|
| ↑ risk | N/A |
| Cardiovascular disease | HF |
|
| ↑ risk | N/A |
| Cardiovascular disease | HF |
|
| ↑ risk | N/A |
| Cardiovascular disease | CAD |
|
| ↑ risk | N/A |
| Cardiovascular disease | CAD |
|
| ↑ risk | N/A |
| Cardiovascular disease | CAD |
|
| ↑ risk | N/A |
| Cardiovascular disease | Valvular heart disease (VHD) |
|
| ↑ risk | N/A |
| Cardiovascular disease | Valvular heart disease (VHD) |
|
| ↑ risk | N/A |
| Cardiovascular disease | Valvular heart disease (VHD) |
|
| ↑ risk | N/A |
| Cardiovascular disease | Cardiac surgery |
|
| ↑ risk |
|
| Cardiovascular disease | Cardiac surgery |
|
| ↑ risk |
|
| Other conditions | Chronic kidney disease (CKD) |
|
| ↑ ↔ risk | N/A |
| Other conditions | Chronic kidney disease (CKD) |
|
| ↑ ↔ risk | N/A |
| Other conditions | Obstructive sleep apnea |
|
| ↑ risk |
|
| Other conditions | Obstructive sleep apnea |
|
| ↑ risk |
|
| Other conditions | Thyroid disease |
|
| ↑ risk | |
| Other conditions | Thyroid disease |
|
| ↑ risk | |
| Other conditions | Sepsis |
|
| ↑ risk | N/A |
| Other conditions | Sepsis |
|
| ↑ risk | N/A |
| Electrocardiogram (ECG) markers | PR interval |
|
|
| N/A |
| Electrocardiogram (ECG) markers | PR interval |
|
|
| N/A |
| Electrocardiogram (ECG) markers | Left ventricular (LV) hypertrophy |
|
| ↑ risk | N/A |
| Electrocardiogram (ECG) markers | Left ventricular (LV) hypertrophy |
|
| ↑ risk | N/A |
| Biomarkers | Natriuretic peptides |
|
| ↑ ↔ risk | N/A |
| Biomarkers | Natriuretic peptides |
|
| ↑ ↔ risk | N/A |
| Biomarkers | Inflammatory markers |
|
|
| N/A |
| Biomarkers | Inflammatory markers |
|
|
| N/A |
| Biomarkers | Lp(a) |
|
| ↑ risk | N/A |
| Biomarkers | Lp(a) |
|
| ↑ risk | N/A |
| Imaging markers | Left atrial size or function |
|
| ↑ LA size, emptying fraction: ↑ risk | Surgical LA reduction in conjunction with cardiac surgery or surgical AF ablation in patients with persistent AF may ↑ rates of sinus rhythm |
| Imaging markers | Left atrial size or function |
|
| ↑ LA size, emptying fraction: ↑ risk | Surgical LA reduction in conjunction with cardiac surgery or surgical AF ablation in patients with persistent AF may ↑ rates of sinus rhythm |
| Imaging markers | LV wall thickness |
|
| ↑ risk | N/A |
| Imaging markers | LV wall thickness |
|
| ↑ risk | N/A |
| Social determinants of health | Education |
|
| Variable ↑ ↓ risk | N/A |
| Social determinants of health | Education |
|
| Variable ↑ ↓ risk | N/A |
| Social determinants of health | Income |
|
| Variable ↑ ↓ risk | N/A |
| Social determinants of health | Socioeconomic status |
|
| Low SES: ↑ ↔ risk | N/A |
| Social determinants of health | Socioeconomic status |
|
| Low SES: ↑ ↔ risk | N/A |
| Genetics | Family history/ heritability |
|
| ↑ risk | N/A |
| Genetics | Family history/ heritability |
|
| ↑ risk | N/A |
| Genetics | Genome-wide association study (GWAS) |
|
| ↑ risk | N/A |
Population attributable fraction: the proportional disease incidence in the population that is estimated to be due to the risk factor.
Statistically significant associations reported, unless otherwise indicated.
2.2. Atrial Arrhythmia Classification and Definitions
Figure 4. AF Stages: Evolution of Atrial Arrhythmia Progression

(Original figure created by the Atrial Fibrillation Guideline Writing Committee.)
Figure 5. Pillars for AF Management
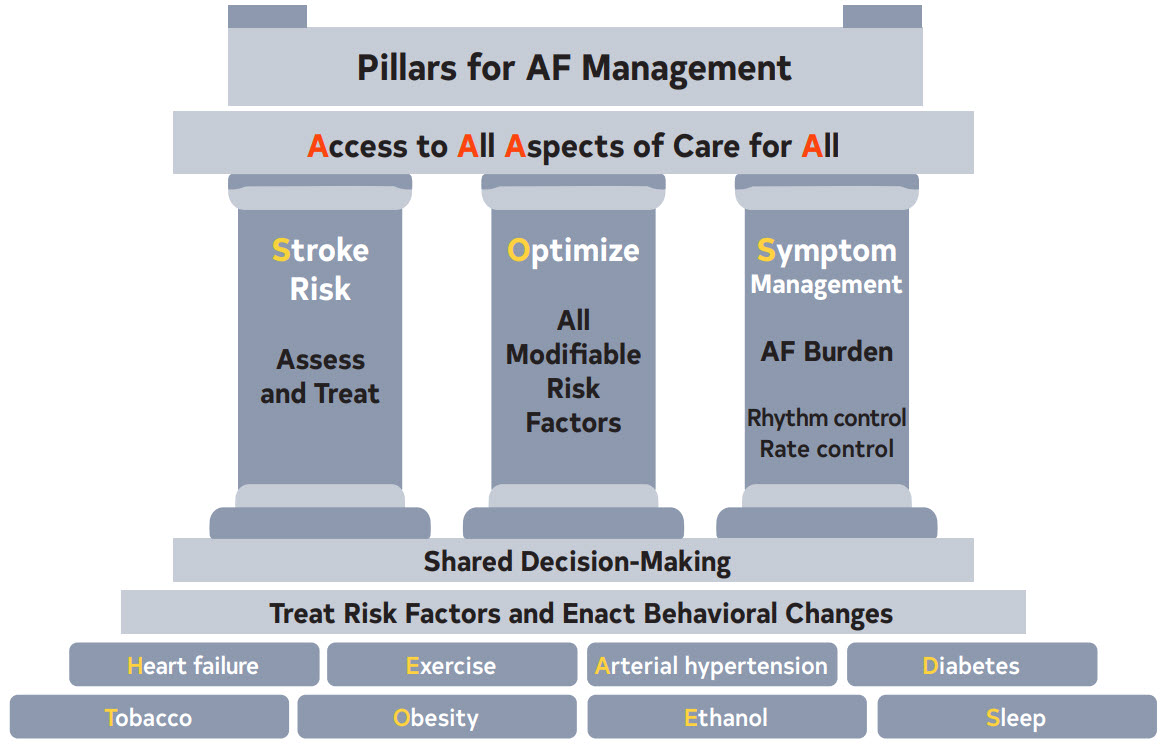
Table 4. Definitions
| Term | Definition |
|---|---|
| Atrial fibrillation |
|
| Clinical AF | With the increasing availability of wearable devices and other continuous monitoring technologies, the distinction between clinical and subclinical AF has become increasingly blurred thus the writing group felt the term clinical AF has become less useful. Yet, the term was kept since most of the evidence from randomized trials that have led to guideline recommendations for the treatment of AF refer to “clinical AF.” These trials required ECG documentation of the arrhythmia for inclusion and a majority of patients presented for clinical evaluation and/or therapy of the arrhythmia. |
| Subclinical AF | Subclinical AF refers to this arrhythmia identified in individuals who do not have symptoms attributable to AF and in whom there are no prior ECGs documenting AF. This includes AF identified by implanted devices (pacemakers, defibrillators, or implantable loop recorders) or wearable monitors. |
| Atrial high-rate episodes (AHRE) | These are defined as atrial events exceeding the programmed detection rate limit set by the device. These are recorded by implanted devices but require visual inspection to confirm AF and exclude other atrial arrhythmias, artifact or oversensing. |
| AF burden | AF burden encompasses both frequency and duration and refers to the amount of AF that an individual has. AF burden has been defined differently across studies. For the purpose of this guideline, AF burden will be defined as the durations of an episode or as a percentage of AF duration during the monitoring period depending on how it was defined in the individual studies. |
| First detected AF | The first documentation of AF, regardless of prior symptoms |
| Paroxysmal AF | AF that is intermittent and terminates within ≤7 days of onset |
| Persistent AF | AF that is continuous and sustains for >7 days and requires intervention. Of note, patients with persistent AF who with therapy become paroxysmal should still be defined as persistent as this reflects their original pattern and is a more useful to predict outcomes and define substrate. |
| Long-standing persistent AF | AF that is continuous for >12 months in duration |
| Permanent AF | A term that is used when the patient and clinician make a joint decision to stop further attempts to restore and/or maintain sinus rhythm Acceptance of AF represents a therapeutic decision and does not represent an inherent pathophysiological attribute of AF |
| Terms considered obsolete | |
| Chronic AF | This historical term has had variable definitions and should be abandoned. It has been replaced by the “paroxysmal, persistent, long-standing persistent and permanent” terminology. |
| Valvular and Nonvalvular AF | The distinction between “valvular” and “non-valvular” AF remains a matter of debate. Their definitions may be confusing. Recent trials comparing vitamin K antagonists with non-vitamin K antagonist oral anticoagulants (OACs) in AF were performed among patients with so-called “non-valvular’’ AF. These trials have all allowed native valvular heart disease other than mitral stenosis (mostly moderate and severe) and prosthetic heart valves to be included. We should no longer consider the classification of AF as ‘‘valvular’’ or “non-valvular” for the purpose of defining the etiology of AF, since the term was specific for eligibility of stroke risk reduction therapies. Valvular and nonvalvular terminology should be abandoned. |
| Lone AF | This term has been used in the past to identify AF in younger patients without structural heart disease who are at a lower risk for thromboembolism. This term does not enhance patient care, is not currently used and should be abandoned. |
2.2.2. Associated Arrhythmias
Figure 6. Types of Atrial Flutter and Macroreentrant Atrial Tachycardia

The typical, reverse typical, and the lower-loop flutter all have the low right atrial isthmus incorporated in the flutter circuit. Other macroreentrant flutters include scar-mediated reentrant tachycardia and left mitral isthmus flutter. Modified with permission from Wellens HJJ.
Copyright 2002, American Heart Association, Inc. Illustration courtesy of Dr F. Cosio.
2.3. Mechanisms and Pathophysiology
Figure 7. Mechanisms and Pathways Leading to AF

2.3.3. Role of the Autonomic Nervous System (ANS)
Figure 8. Contemporary Summary of the Role of the ANS in AF

2.5. Addressing Health Inequities and Barriers to AF Management
3. Shared Decision-Making in AF Management
Table 5. Table of Publicly Available Decision Aids
| Agency | Website Link | Focus Area |
|---|---|---|
| American College of Cardiology Colorado Program for Patient Centered Decisions | https://patientdecisionaid.org/icd/atrial-fibrillation/ | Stroke risk reduction therapies |
| Anticoagulation Choice Decision Aid | https://anticoagulationdecisionaid.mayoclinic.org/ | Stroke risk reduction therapies |
| Ottawa Hospital Research Institute Developer Healthwise | https://decisionaid.ohri.ca/AZlist.html | AF ablation Stroke risk reduction |
| Stanford | https://afibguide.com/ | Stroke risk reduction therapies |
Management
4. Clinical Evaluation
4.1. Risk Stratification and Population Screening
Table 6. CHARGE-AF Risk Score for Detecting Incident AF*
| Variable (X) | Estimated β coefficient (SE) | HR (95% CI) |
|---|---|---|
| Age (per 5-y increment) | 0.508 (0.022) | 1.66 (1.59–1.74) |
| White Race | 0.465 (0.093) | 1.59 (1.33–1.91) |
| Height (per 10-cm increment) | 0.248 (0.036) | 1.28 (1.19–1.38) |
| Weight (per 15-kg increment) | 0.115 (0.033) | 1.12 (1.05–1.20) |
| Systolic BP (per 20-mm Hg increment) | 0.197 (0.033) | 1.22 (1.14–1.30) |
| Diastolic BP (per 10-mm Hg increment) | -0.101 (0.032) | 0.90 (0.85–0.96) |
| Smoking (current versus former/never) | 0.359 (0.063) | 1.42 (1.25–1.60) |
| Diabetes (yes) | 0.237 (0.073) | 1.27 (1.64–2.48) |
| Myocardial infarction (yes) | 0.496 (0.089) | 1.64 (1.38–1.96) |
Table 6 does not encompass all complications.
Table 7. C2HEST Risk Score for Detecting Incident AF*
| Acronym | Risk Factor | Points |
|---|---|---|
| C2 | CAD/chronic obstructive pulmonary disease (COPD) | 1–2 |
| H | Hypertension | 1 |
| E | Elderly (age ≥75 y) | 2 |
| S | Systolic heart failure | 2 |
| T | Thyroid disease (hyperthyroidism) | 1 |
The C2HEST score: C2, coronary artery disease or chronic obstructive pulmonary disease [1 point each]; H, hypertension [1 point]; E, elderly [age ≥75 y, 2 points]; S, systolic HF [2 points]; T, thyroid disease [hyperthyroidism, 1 point])
4.2. Basic Evaluation
4.2.1. Basic Clinical Evaluation
4.2.2. Rhythm Monitoring Tools and Methods
5. Lifestyle and Risk Factor Modification for AF Management
5.1. Primary Prevention
5.2. Secondary Prevention: Management of Comorbidities and Risk Factors
5.2.1. Weight Loss in Individuals Who Are Overweight or Obese
5.2.2. Physical Fitness
5.2.3. Smoking Cessation
5.2.4. Alcohol Consumption
5.2.5. Caffeine Consumption
5.2.8. Treatment of Hypertension
5.2.9. Sleep
5.2.10. Comprehensive Care
6. Prevention of Thromboembolism
6.1. Risk Stratification Schemes
Table 8. Three Validated Risk Models for Stroke
| Risk Factor | CHA2DS2-VASc | ATRIA | GARFIELD |
|---|---|---|---|
| Age ≥85 y | 6 | 0.98 | |
| Age ≥75 y | 2 | 5 | 0.59 |
| Age 65–74 y | 1 | 3 | 0.20 |
| Female sex | 1 | 1 | |
| Hypertension | 1 | 0.16 | |
| Renal disease | 1 | 0.35 | |
| Diabetes | 1 | 1 | 0.21 |
| Current smoking | 0.48 | ||
| Congestive heart failure | 1 | 1 | 0.23 |
| Prior stroke or transient ischemic attack (TIA) | 2 | 2–8* | 0.80 |
| Vascular disease | 1 | 0.20 | |
| Dementia | 0.51 | ||
| Prior bleeding | 0.30 | ||
| Proteinuria | 1 | ||
| Low risk score | 0 | 0–5 | 0—0.89 |
| Intermediate risk score | 1 | 6 | 0.90—1.59 |
| High risk score | ≥2 | 7–15 | ≥1.60 |
| C-index (11) | — | ||
| C-index (13) | 0.67 | — | 0.71 |
Figure 9. Rates of Stroke by Stroke Risk Score Levels in Different Cohorts
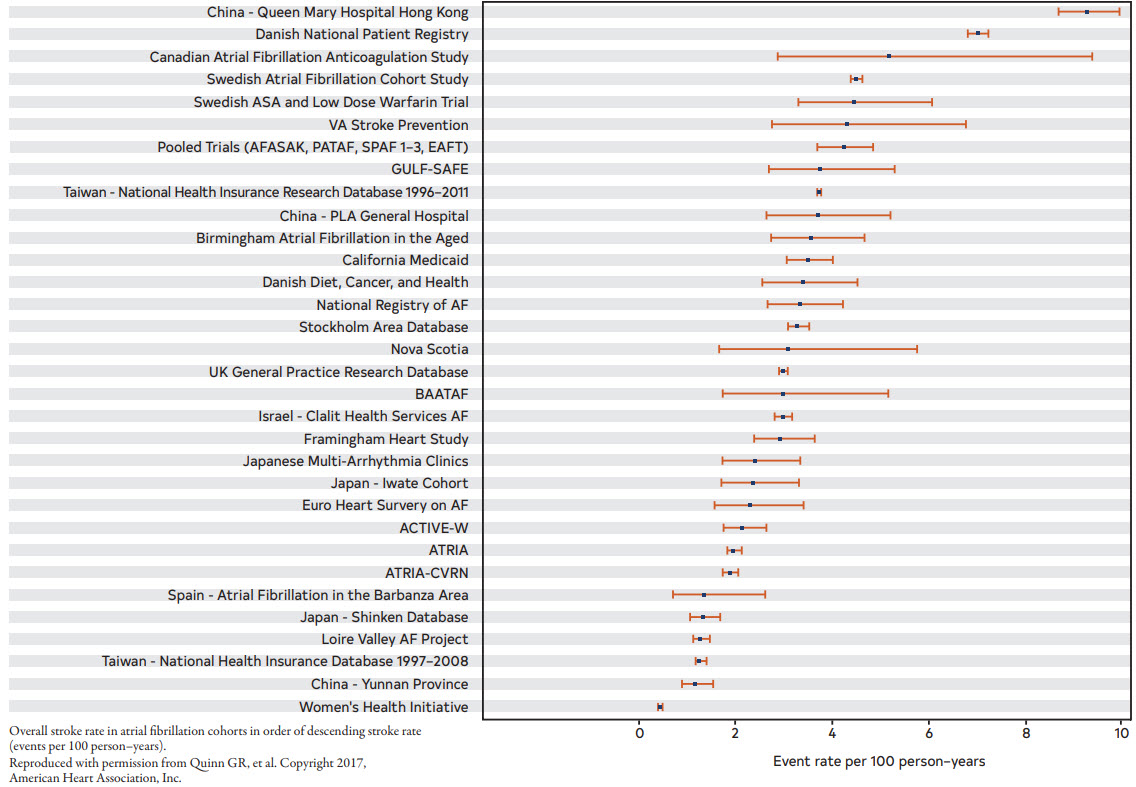
Table 9. Some Best Known Published Clinical Scores With Potential Advantages
| Year of Publication Score Name | Score Components | Potential Advantages | No. of Validation Studies | Hyperlink to Online Score Calculator if Available |
|---|---|---|---|---|
| 2001 Congestive heart failure, hypertension, age >75 years, diabetes mellitus, stroke/transient ischemia attack/ thromboembolism (CHADS2) | Congestive heart failure (CHF), hypertension, age (≥65 y = 1 point, ≥75 y = 2 points), diabetes, stroke/TIA (2 points) | CHADS2 was superior to existing risk classification schemes. AFI scheme: C-statistic = 0.68 (0.65–0.71) SPAF-III scheme: C-statistic = 0.74 (0.71–0.76) CHADS2 score: C-statistic = 0.82 (0.80–0.84) | 46 | https://www.mdcalc.com/calc/40/chads2-score-atrial-fibrillation-stroke-risk |
| 2010 CHA2DS2-VASc | CHF, hypertension, age ≥75 y, diabetes, stroke or TIA, vascular disease, age 65–74 y, female sex | Most commonly used and studied, superior to CHADS2. C-statistic = 0.606 (0.513–0.699) for CHA2DS2-VASs versus 0.561 (0.450–0.672) for CHADS2 Improved compared with original CHADS2 score. | 82 | https://www.mdcalc.com/calc/801/cha2ds2-vasc-score-atrial-fibrillation-stroke-risk#next-steps |
| 2013 ATRIA | Age (65–74 y = 3 points, 75–84 y = 5 points, ≥85 y = 6 points), hypertension, diabetes, CHF, proteinuria, GFR <45 mL/min/1.73 m2, sex | Includes more age categories, renal function, and proteinuria. More patients were classified as low or high risk but not as well tested in general. | 11 | https://www.mdcalc.com/calc/1842/atria-stroke-risk-score |
| 2017 GARFIELD-AF | Web-based, uses routinely collected clinical data, and includes a total of 16 questions | Web-based tool for predicting stroke and mortality, includes the effect of the different anticoagulants, bleeding risk and mortality to facilitate shared decision-making on the potential benefits/risks of anticoagulation | 4 | https://af.garfieldregistry.org/garfield-af-risk-calculator |
| 2016 MCHA2DS2-VASc | Expanded lower threshold for age to 50 y (1 point for age 50–74 y) | Validated in Asian cohort. Can further identify Asian AF patients who may derive benefits from stroke prevention. In one study, MCHA2DS2-VASc was superior to CHA2DS2-VASc. C-statistics = 0.708 (0.703–0.712) vs. 0.689 (0.684–0.694) | 1 |
Table 10. Risk Factor Definitions for CHA2DS2-VASc Score as in the Original Article
| C | Heart Failure | The presence of signs and symptoms of either right (elevated central venous pressure, hepatomegaly, dependent edema) or left ventricular failure (exertional dyspnea, cough, fatigue, orthopnea, paroxysmal nocturnal dyspnea, cardiac enlargement, rates, gallop rhythm, pulmonary venous congestion) or both, confirmed by noninvasive or invasive measurements demonstrating objective evidence of cardiac dysfunction. |
|---|---|---|
| H | Hypertension | A resting blood pressure >140 mm Hg systolic and/or >90 mm Hg diastolic on at least 2 occasions or current antihypertensive pharmacological treatment. |
| A2 | Age, additional risk/point | Age ≥75 y |
| D | Diabetes | Fasting plasma glucose level ≥7.0 mmol/L (126 mg/dL) or treatment with hypoglycemic agent and/or insulin. |
| S2 | Thromboembolism | Either an ischemic stroke, transient ischemic attack, peripheral embolism, or pulmonary embolism. |
| V | Vascular Disease | Coronary artery disease (prior myocardial infarction, angina pectoris, percutaneous coronary intervention (PCI), or coronary artery bypass surgery) or peripheral vascular disease (the presence of any of the following: intermittent claudication, previous surgery or percutaneous intervention on the abdominal aorta or the lower extremity vessels, abdominal or thoracic vascular surgery, arterial and venous thrombosis.) |
| A | Age standard risk/weight | Age 65–74 y |
| Sc | Sex Category | Female sex |
Table 11. Factors That Increase the Risk of Stroke
- Higher AF burden/Long duration
- Persistent/permanent AF versus paroxysmal
- Obesity (body mass index ≥30 kg/m2)
- HCM
- Poorly controlled hypertension
- Estimated glomerular filtration rate (eGFR) (<45 mL/h)
- Proteinuria (>150 mg/24 h or equivalent)
- Enlarged LA volume (≥73 mL) or diameter (≥4.7 cm)
Table 12. Thromboembolic Event Rates by Point Score for ATRIA, CHADS2, and CHA2DS2-VASc Risk Scores*
| Points | ATRIA Events | ATRIA Person-years | ATRIA Rate per 100 Person-years | CHADS2† Events | CHADS2† Person-years | CHADS2† Rate per 100 Person-years | CHA2DS2-VASc‡ Events | CHA2DS2-VASc‡ Person-years | CHA2DS2-VASc‡ Rate per 100 Person-years |
|---|---|---|---|---|---|---|---|---|---|
| 0 | 2 | 2652 | 0.08 | 22 | 6126 | 0.36 | 1 | 2493 | 0.04 |
| 1 | 12 | 2819 | 0.43 | 121 | 10, 084 | 1.20 | 21 | 3806 | 0.55 |
| 2 | 14 | 1419 | 0.99 | 253 | 9757 | 2.59 | 46 | 5560 | 0.83 |
| 3 | 13 | 1780 | 0.73 | 178 | 4782 | 3.72 | 121 | 7305 | 1.66 |
| 4 | 19 | 2960 | 0.64 | 81 | 1309 | 6.19 | 193 | 6898 | 2.80 |
| 5 | 36 | 3614 | 0.99 | 19 | 450 | 4.23 | 175 | 4057 | 4.31 |
| 6 | 83 | 4346 | 1.91 | 11 | 101 | 10.84 | 85 | 1783 | 4.77 |
| 7 | 119 | 4768 | 2.50 | — | — | — | 24 | 498 | 4.82 |
| 8 | 151 | 3913 | 3.86 | — | — | — | 14 | 179 | 7.82 |
| 9 | 104 | 2400 | 4.33 | — | — | — | 5 | 30 | 16.62 |
| 10 | 75 | 1181 | 6.35 | — | — | — | — | — | — |
| 11 | 31 | 501 | 6.18 | — | — | — | — | — | — |
| 12 | 20 | 183 | 10.95 | — | — | — | — | — | — |
| 13 | 4 | 53 | 7.52 | — | — | — | — | — | — |
| 14 | 2 | 12 | 16.36 | — | — | — | — | — | — |
| 15 | 0 | 7 | 0 | — | — | — | — | — | — |
| All | 685 | 32, 609 | 2.10 | — | — | — | — | — | — |
† The CHADS2 score assigns points as follows: 1 point each for the presence of congestive heart failure, hypertension, age ≥75 y, and diabetes mellitus and 2 points for history of stroke/transient ischemic attack.
‡ The CHA2DS2-VASc score assigns points as follows: 1 point each for congestive heart failure/left ventricular dysfunction, hypertension, diabetes mellitus, vascular disease, age 65 to 74 y, and female sex, and 2 points each for age ≥75 years and stroke/transient ischemic attack/thromboembolism.
Reproduced with permission from Singer DE, et al. Copyright 2013, The Authors.
Published on behalf of the American Heart Association, Inc., by Wiley-Blackwell.
6.2. Risk-Based Selection of Oral Anticoagulation: Balancing Risks and Benefits
6.3. Oral Anticoagulants
6.3.1. Antithrombotic Therapy
Figure 10. Antithrombotic Options in Patients with AF
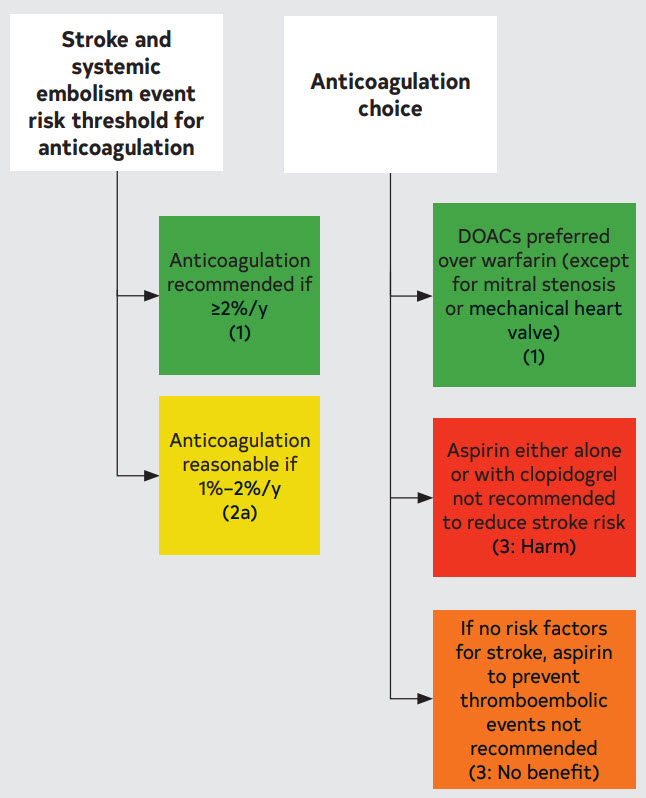
Table 13. OACs Pharmacokinetic Characteristics and Dosing
| Name (Class) | Warfarin (Vitamin K antagonist (VKA)) | Dabigatran (Direct Thrombin Inhibitor) | Rivaroxaban (Factor Xa Inhibitor) | Apixaban (Factor Xa Inhibitor) | Edoxaban (Factor Xa Inhibitor) |
|---|---|---|---|---|---|
| Metabolism | S-isomer: CYP2C9 R-isomer: CYP2C19, CYP3A4 | Minimal | CYP3A4/5 | CYP3A4 | Minimal CYP3A4 |
| P-glycoprotein substrate | No | Yes | Yes | Yes | Yes |
| Excretion | 0% renal; very little warfarin excreted unchanged in urine | 80% renal | 66% renal, 28% feces | 27% renal, 73% biliary and intestinal | 50% renal, 50% liver and biliary/intestinal |
| Half-life | 20–60 h | 12–17 h | 5–9 h | 12 h | 10–14 h |
| Renal dosing adjustment based on actual body weight | N/A |
|
|
|
|
| Drug interaction management based on concomitant therapy of CYP3A4 inhibitors/p-glycoprotein inhibitors | Adjust dose based on INR trends | CrCl 30–50 mL/ min with concomitant use of dronedarone or systemic ketoconazole: 75 mg twice daily CrCl < 30 mL/min: avoid dabigatran use concomitantly with dronedarone or systemic ketoconazole | Avoid rivaroxaban use with concomitant therapy of combined p-glycoprotein and strong CYP3A4 inhibitors (eg, systemic ketoconazole and ritonavir) No dose adjustment required with clarithromycin Avoid rivaroxaban use in patients with CrCl 15 to < 80 mL/min receiving combined p-glycoprotein and moderate CYP3A4 inhibitors (eg, erythromycin) | In patients receiving apixaban 5 mg twice daily, reduce dose to 2.5 mg twice daily when combined p-glycoprotein and strong CYP3A4 inhibitors (eg, itraconazole, systemic ketoconazole, ritonavir) are concomitantly used If patients already receiving apixaban 2.5 mg twice daily, avoid apixaban use if combined p-glycoprotein and strong CYP3A4 inhibitors are concomitantly used | No dose adjustment is required |
| Drug interaction management based on concomitant therapy of p-glycoprotein/ CYP3A4 inducers (eg, carbamazepine, phenytoin, rifampin, St. John’s wort) | Adjust dose based on INR trends | Avoid use | Avoid use | Avoid use | Avoid use with rifampin. No study evaluated the effect of other p-glycoprotein/ CYP3A4 inducers on edoxaban drug levels |
| Appropriate use based on liver function (Child-Pugh score)† Child-Pugh A (mild) | Not mentioned in the labeling | No dose adjustment needed | No dose adjustment needed | No dose adjustment needed | No dose adjustment needed |
| Child-Pugh B (moderate) | Not mentioned in the labeling | Use with caution | Avoid use | Use with caution | Use with caution |
| Child-Pugh C (severe) | Not mentioned in the labeling | Avoid use | Avoid use | Avoid use | Avoid use |
†Child-Pugh scoring: the severity of liver disease, primarily cirrhosis. Child-Pugh A (mild): 5 to 6 points; Child-Pugh B (moderate): 7 to 9 points; Child-Pugh C (severe): 10 to 15 points. The score is based on the 5 variables: encephalopathy (none=1 point, grade 1 and 2=2 points, grade 3 and 4=3 points); ascites (none=1 point, slight=2 points, moderate=3 points); total bilirubin (<2 mg/mL=1 point, 2-3 mg/mL=2 points, >3 mg/mL=3 points); albumin (>3.5 mg/mL=1 point, 2.8-3.5 mg/mL=2 points, <2.8 mg/mL=3 points); INR (<1.7=1 point, 1.7-2.2=2 points, >2.2=3 points).
Information obtained from manufacturer package inserts. Adapted with permission from pgs. 28–31 of Kido K. Copyright 2021, American College of Clinical Pharmacy
Figure 11. DOAC Laboratory Monitoring

* HAS-BLED scoring (low risk=score 0, moderate risk=score 1–2, high risk=score ≥3): uncontrolled hypertension (systolic blood pressure >160 mm Hg)=1 point; abnormal renal (serum creatinine >2.26 mg/dL, dialysis, or kidney transplant) or hepatic function (bilirubin >2 times upper limit normal, alanine aminotransferase/aspartate aminotransferase/alkaline phosphatase >3 times upper limit normal, or cirrhosis)=1 or 2 points; stroke (hemorrhagic or ischemic)=1 point; bleeding history or predisposition=1 point; labile INR (time in therapeutic range <60%)=1 point; elderly age >65 years=1 point; drugs (antiplatelet agents or nonsteroidal anti-inflammatory drugs) or excessive alcohol intake (8 units/week)=1 or 2 points.
Child-Pugh scoring: the severity of liver disease, primarily cirrhosis in patients with documented liver disease. Child-Pugh A (mild): 5 to 6 points; Child-Pugh B (moderate): 7 to 9 points; Child-Pugh C (severe): 10 to 15 points. The score is based on the 5 variables: encephalopathy (none=1 point, grade 1 and 2=2 points, grade 3 and 4=3 points); ascites (none=1 point, slight=2 points, moderate=3 points); total bilirubin (<2 mg/mL=1 point, 2–3 mg/mL=2 points, >3 mg/mL=3 points); albumin (>3.5 mg/mL=1 point, 2.8–3.5 mg/mL=2 points, <2.8 mg/mL=3 points); INR (<1.7=1 point, 1.7–2.2=2 points, >2.2=3 points).
6.3.1.1. Considerations in Managing Anticoagulants
6.4. Silent AF and Stroke of Undetermined Cause
6.4.1. Oral Anticoagulation for Device-Detected Atrial High-Rate Episodes Among Patients Without a Prior Diagnosis of AF
Figure 12. Consideration of Oral Anticoagulation for Device-Detected AHREs According to Patient Stroke Risk by CHA2DS2-VASc Score and Episode Duration

A potential approach to patients with SCAF could consider both patient risk (as gauged by the CHA2DS2-VASc score) and SCAF burden/duration.
Circle A indicates patients at low risk or with short and infrequent AHREs do not require anticoagulation; Circle B, patients with intermediate risk and AHREs lasting >6 min to 24 h are an uncertain population but are currently under study in 2 prospective randomized controlled trials; and Circle C, patients at high risk with longer episodes could be considered reasonable candidates for anticoagulation, although the precise threshold for SCAF duration remains uncertain.
Reproduced with permission from Noseworthy PA, et al. Copyright 2019 American Heart Association, Inc.
Modified from Freedman B et al. Copyright 2017 Springer Nature Limited.
6.5.1. Percutaneous Approaches to Occlude the Left Atrial Appendage (LAA)
Table 14. Situations in Which Long-Term Anticoagulation Is Contraindicated and Situations When It Remains Reasonable
- Severe bleeding due to a nonreversible cause involving the gastrointestinal, pulmonary, or genitourinary systems
- Spontaneous intracranial/intraspinal bleeding due to a nonreversible cause
- Serious bleeding related to recurrent falls when cause of falls is not felt to be treatable
Long-Term Anticoagulation is Still Reasonable
- Bleeding involving the gastrointestinal, pulmonary, or genitourinary systems that is treatable
- Bleeding related to isolated trauma
- Bleeding related to procedural complications
6.5.2. Cardiac Surgery — LAA Exclusion/Excision
6.6 Active Bleeding on Anticoagulant Therapy and Reversal Drugs
Table 15. Reversal Agents for Oral Anticoagulants
| Idarucizumab | Andexanet alfa | 4-Factor PCC | Activated PCC | |
|---|---|---|---|---|
| Class | Humanized monoclonal antibody fragment binding to dabigatran and neutralizing anticoagulation effects | A recombinant modified human factor Xa protein binding and sequestering the factor Xa inhibitors | Prothrombin complex concentrate: coagulation factors II, VII, IX, and X Anticoagulation proteins C and S | Nonactivated factors II, IX, and X Activated VII |
| US Food and Drug Administration (FDA) indications | Reversal of dabigatran effects
| Reversal of apixaban or rivaroxaban
| The urgent reversal for acute major bleeding or need for an urgent surgery/invasive procedure in patients receiving vitamin K antagonists | Control and prevention of bleeding episodes, perioperative management, prophylaxis to prevent or reduce bleeding frequency in hemophilia A and B patients |
| Off-label indications | N/A | Edoxaban-associated life-threatening bleeding | Reversal of factor Xa inhibitors in patients requiring urgent procedure or with life-threatening bleeding | Dabigatran- associated life-threatening bleeding |
| Dosing | 5-g (2 separate vials of 2.5 g/vial) intravenous infusion over 5 minutes. Additional 5 g may be given if reappearance of bleeding with elevated coagulation parameters have been observed or patients require second emergent surgery/procedure and elevated coagulation parameters | Low-dose regimen: 400-mg bolus at a target rate of 30 mg/min followed by 4 mg/min for up to 120 min High-dose regimen: 800-mg bolus at a target rate of 30 mg/min followed by 8 mg/min for up to 120 min The recommended dosing is based on apixaban or rivaroxaban, dose, and time since the patient’s last dose of apixaban or rivaroxaban | Warfarin reversal based on pretreatment INR (units of factor IX): 1. INR 2 to <4: 25 units/kg (up to 2500 units) 2. INR 4 to 6: 35 units/kg (up to 3500 units) 3. INR 6<: 50 units/kg (up to 5000 units) Oral factor Xa inhibitors: 2000 units once or 25 to 50 units/kg | Dabigatran- associated life-threatening bleeding: 50 units/kg once |
| Onset | Within 5 min | Within 2 min | Within 10 min | Within 30 min |
| Duration | 12–24 h | 2 h | 8 h | 12 h |
| Monitoring | Coagulation parameters (activated partial thromboplastin time [aPTT], diluted thrombin time, or ecarin clotting time) between 12–24 h to assess redistribution of dabigatran from peripheral to plasma | Current commercial anti-Xa activity assays are not suitable for measuring factor Xa activities after andexanet alfa use | Warfarin reversal: Repeat INR within 30 min after the administration | N/A |
| Others | Risk of serious reactions (hypoglycemia, hypophosphatemia, metabolic acidosis, increase in uric acid, acute liver failure) in patients with hereditary fructose intolerance (due to sorbitol excipient 4 g in each 5 g of idarucizumab) No procoagulant effect based on endogenous thrombin potential | No FDA indication for other factor Xa inhibitors other than apixaban or rivaroxaban Andexanet alfa may interfere with the anticoagulation effect of heparin US Black Box warning: Serious and life-threatening adverse events (arterial and venous thromboembolism, myocardial infarction, ischemic stroke, cardiac arrest, sudden deaths) | May not be indicated for patients with thromboembolic events in the prior 3 mon It includes heparin Administer intravenous vitamin K 10 mg over 10–20 min in addition to 4-factor PCC | It does not include heparin Coagulation parameters do not correlate with the drug’s efficacy Not effective to reverse factor Xa inhibitors |
Figure 13. Active Bleeding Associated with Oral Anticoagulant
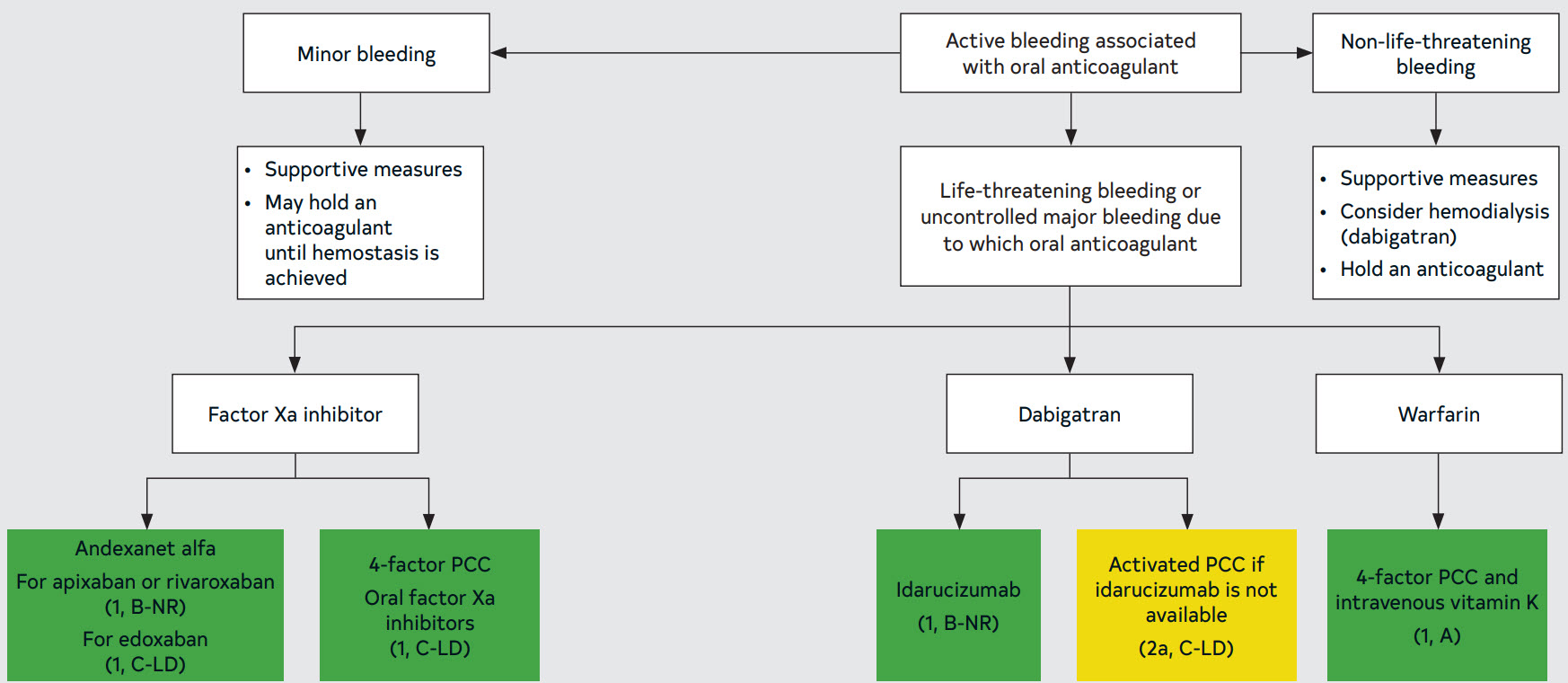
Figure 14. Forms of ICH, Classified by Mechanism
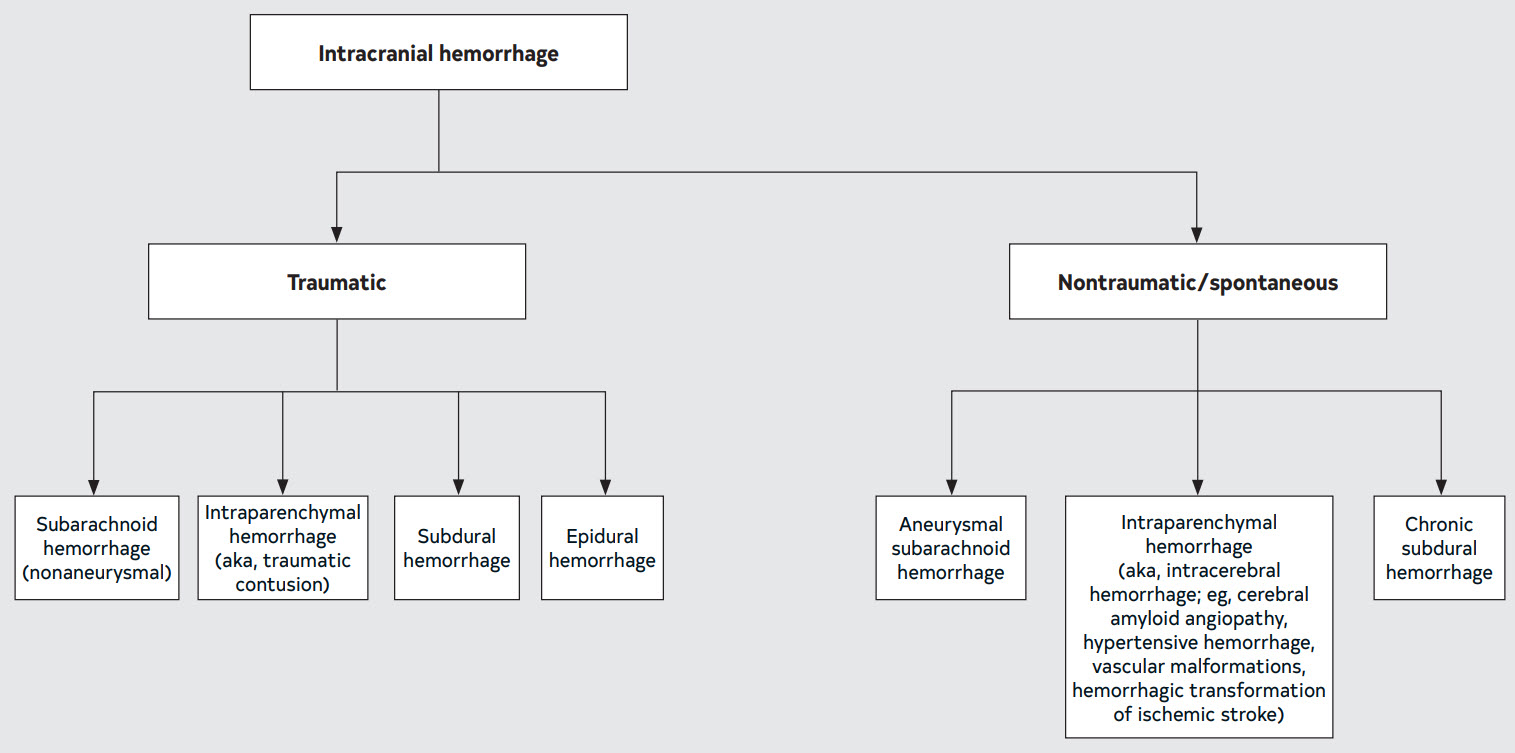
6.6.1. Management of Patients with AF and Intracranial Hemorrhage
Table 16. Bleeding Events (Precent/Year) in Direct Oral Anticoagulant Pivotal Clinical Trials
| Study | RE-LY (n=18,113) | ARISTOTLE (n=18,201) | ENGAGE-AF TIMI 48 (n=21,105) | ROCKET-AF (n=14,264) |
|---|---|---|---|---|
| Major bleeding |
|
| Rivaroxaban 3.6% vs. warfarin 3.4% (HR, 1.04, 95% CI, 0.90–1.20; P=0.58) | |
| GI bleeding |
|
|
| |
| Intracranial bleeding |
|
|
|
|
Adapted with permission from pgs. 32-33 of Kido K.22 Copyright 2021 American College of Clinical Pharmacy.
Table 17. Risk Factors for Thromboembolic Complications and Recurrent ICH
- Mechanical heart valve
- Rheumatic valve disease
- Previous history of stroke/ thromboembolism
- Hypercoagulable state (eg, active malignancy, genetic thrombophilia)
- High CHA2DS2-VASc score (>5)
Factors Associated With High Risk of Recurrent ICH
- Suspected cerebral amyloid angiopathy
- Lobar intraparenchymal hemorrhage (IPH)
- Older age
- >10 cerebral microbleeds on MRI
- Disseminated cortical superficial siderosis on MRI
- Poorly controlled hypertension
- Previous history of spontaneous ICH
- Genetic/acquired coagulopathy
- Untreated symptomatic vascular malformation or aneurysm
6.7. Periprocedural Management
Figure 15. Flowchart: Management of Periprocedural Anticoagulation in Patients With AF
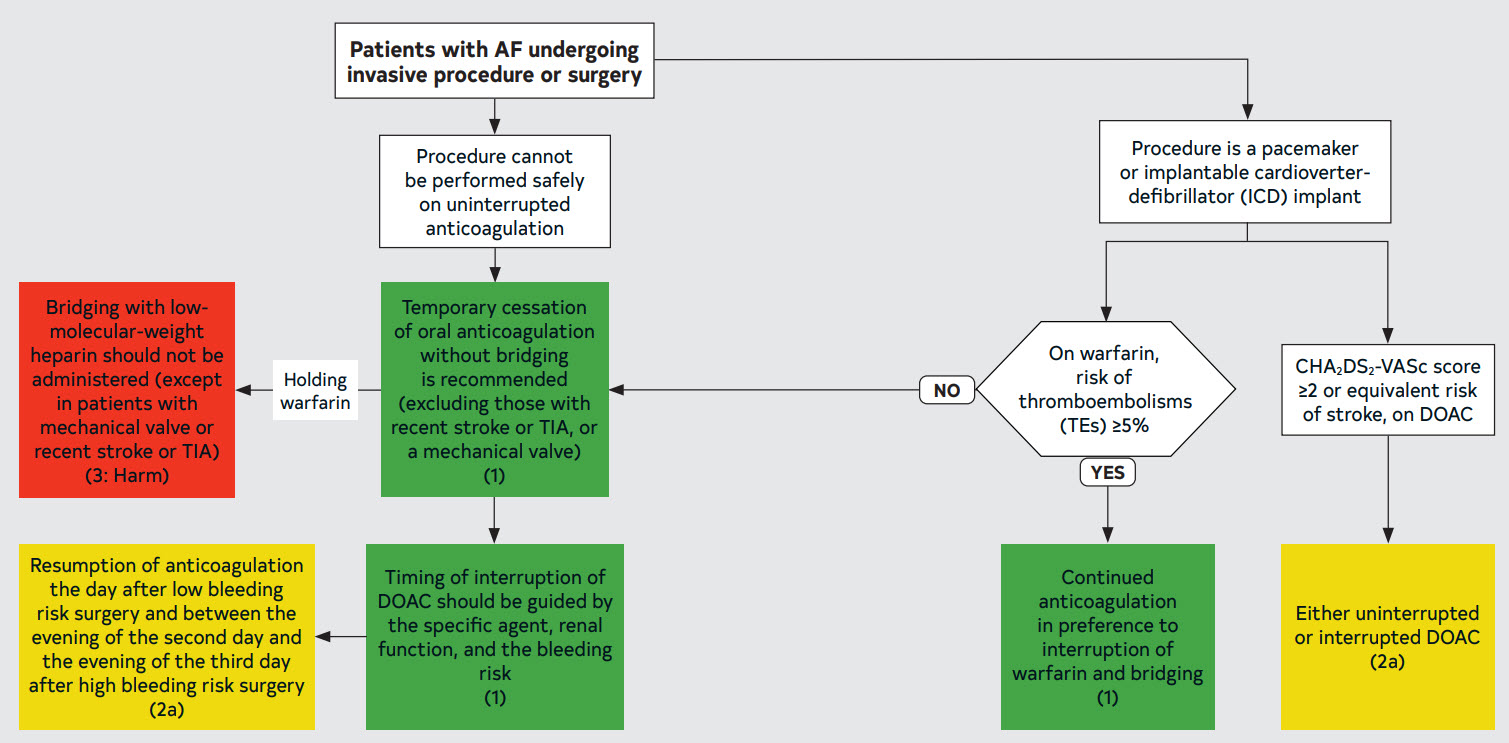
Table 18. Timing of Discontinuation of Oral Anticoagulants in Patients With AF Scheduled to Undergo an Invasive Procedure or Surgery in Whom Anticoagulation is to Be Interrupted
| Anticoagulant | Low Bleeding Risk Procedure | High Bleeding Risk Procedure |
|---|---|---|
| Apixaban (CrCl >25 mL/min)* | 1 d† | 2 d |
| Dabigatran (CrCl >50 mL/min) | 1 d | 2 d |
| Dabigatran (CrCl 30-50 mL/min) | 2 d | 4 d |
| Edoxaban (CrCl >15 mL/min) | 1 d | 2 d |
| Rivaroxaban (CrCl >30 mL/min) | 1 d | 2 d |
| Warfarin | 5 d for a target INR <1.5 2-3 d for a target INR <2 | 5 d |
* For patients on DOAC with creatinine clearance lower than the values in the table, few clinical data exist: consider holding for an additional 1 to 3 days, especially for high bleeding risk procedures.
† The number of days is the number of full days before the day of surgery in which the patient does not take any dose of anticoagulant. The drug is also not taken the day of surgery. For instance, in the case of holding a twice daily drug for 1 day, if the drug is taken at 8 pm, and surgery is at 8 am, at the time of surgery it will be 36 hours since the last dose was taken.
6.8. Anticoagulation in Specific Populations
6.8.1. AF Complicating Acute Coronary Syndrome or Percutaneous Coronary Intervention
6.8.2. Chronic Coronary Disease (CCD)
6.8.3. Peripheral Artery Disease (PAD)
6.8.4. Chronic Kidney Disease/Kidney Failure
Table 19. Recommended Doses of Currently Approved DOACs According to Renal Function
| DOAC | CrCl (mL/min) >95 mL/min | CrCl (mL/min) 51–95 | CrCl (mL/min) 31–50 | CrCl (mL/min) 15–30 | CrCl (mL/min) <15 or on dialysis |
|---|---|---|---|---|---|
| Apixaban | 5 or 2.5 mg twice daily* | 5 or 2.5 mg twice daily* | 5 or 2.5 mg twice daily* | 5 or 2.5 mg twice daily* | 5 or 2.5 mg twice daily* |
| Dabigatran | 150 mg twice daily | 150 mg twice daily | 150 mg twice daily | 75 mg twice daily | Contraindicated |
| Edoxaban | Contraindicated | 60 mg once daily | 30 mg once daily | 30 mg once daily | Contraindicated |
| Rivaroxaban | 20 mg once daily | 20 mg once daily | 15 mg once daily | 15 mg once daily | 15 mg once daily† |
Note that other, nonrenal considerations such as drug interactions may also apply.
The gray area indicates doses not studied in the pivotal clinical trials of these agents.
* If at least 2 of the following are present: serum creatinine ≥1.5 mg/dL, age ≥80 years, or body weight ≤60 kg, the recommended dose is 2.5 mg twice daily. The ARISTOTLE trial excluded patients with either a creatinine of >2.5 mg/dl or a calculated CrCl <25 mL/min.
† Rivaroxaban is not recommended for other indications in patients with a CrCl <15 mL/min, but such a recommendation is not made for the AF indication. However, pharmacokinetic data are limited.
6.8.5. AF in Valvular Heart Disease
6.8.6. Anticoagulation of Typical Atrial Flutter
- “Typical” AFL is defined as either typical counterclockwise AFL when the macroreentrant circuit is dependent on the CTI using the isthmus from the patient’s right to left or typical clockwise AFL when the macroreentrant circuit is dependent on the CTI and uses this isthmus from the patient’s left to right.
- “Atypical” AFL is not dependent on the CTI and may arise from a macroreentrant circuit in the LA, such as perimitral or LA roof flutter or could be dependent on scar from prior ablation or surgery.
7. Rate Control
Figure 16. Anticoagulation for Typical (CTI-Dependent) AFL

† For example, left atrial enlargement, inducible AF, chronic obstructive pulmonary disease, concomitant heart failure.
7.1. Broad Considerations for Rate Control
Table 20. Clinical Presentations and Objectives of Heart Rate Control
| Presentation | Objective |
|---|---|
| Symptomatic atrial fibrillation | To reduce symptoms |
| Tachycardia-induced cardiomyopathy | To improve heart function or reduce the risk of recurrent cardiomyopathy |
| Implantable cardioverter-defibrillator use | To reduce risk of inappropriate shock |
| Cardiac resynchronization therapy (CRT) use | To enhance biventricular pacing (BiVP), likelihood of myocardial recovery, and/or preservation of function |
| Tachycardia-bradycardia form of sick sinus syndrome among those with a pacemaker | To reduce the risk of hospitalization |
Table 21. Pharmacological Agents for Rate Control in Patients With Atrial Fibrillation - Beta Blockers
| Intravenous Administration | Oral Maintenance Dose | Elimination Half-Life | Notes | |
|---|---|---|---|---|
| Metoprolol tartrate | 2.5–5 mg bolus over 2 min; up to 3 doses | 25–200 mg twice daily | 3–4 h | |
| Metoprolol succinate | N/A | 50–400 mg daily | 3–7 h | |
| Atenolol | N/A | 25–100 mg daily | 6–7 h | Predominantly renally eliminated |
| Bisoprolol | N/A | 2.5–10 mg daily | 9–12 h | |
| Carvedilol | N/A | 3.125–25 mg twice daily | 7–10 h | |
| Esmolol | 500 µg/kg bolus over 1 min; then 50–300 µg/kg/min | N/A | 9 min | |
| Nadolol | N/A | 10–240 mg daily | 20–24 h | |
| Propranolol | 1 mg over 1 min; repeat as needed every 2 min; up to 3 doses | 10–40 mg 3–4 times daily | IV: 2.4 h Oral: 3–6 h Extended release (ER:) 8–20 h |
Table 21. Pharmacological Agents for Rate Control in Patients With Atrial Fibrillation - Nondihydropyridine Calcium Channel Blockers
| Intravenous Administration | Oral Maintenance Dose | Elimination Half-Life | Notes | |
|---|---|---|---|---|
| Diltiazem | 0.25 mg/kg (actual body weight) IV over 2 min May repeat 0.35 mg/kg over 2 min; then 5–15 mg/h continuous infusion | 120–360 mg daily (ER) | IV: 3–5 h Oral immediate release: 3–4.5 h ER: 4–9.5 h | Avoid in heart failure with reduced ejection fraction (HFrEF) |
| Verapamil | 5–10 mg over ≥2 min (may repeat twice); then 5 mg/h continuous infusion (max 20 mg/h) | 180–480 mg daily (ER) | IV: 6–8 h Oral: 2–7 h ER: 12–17 h | Avoid in HFrEF |
Table 21. Pharmacological Agents for Rate Control in Patients With Atrial Fibrillation - Digitalis Glycoside
| Intravenous Administration | Oral Maintenance Dose | Elimination Half-Life | Notes | |
|---|---|---|---|---|
| Digoxin | 0.25–0.5 mg over several min; repeat doses of 0.25 mg every 6 h (maximum 1.5 mg/24 h) | 0.0625–0.25 mg daily | 1–2 d | Renally eliminated Increased mortality at plasma concentrations exceeding 1.2 ng/mL |
Table 21. Pharmacological Agents for Rate Control in Patients With Atrial Fibrillation - Other
| Intravenous Administration | Oral Maintenance Dose | Elimination Half-Life | Notes | |
|---|---|---|---|---|
| Amiodarone | 150–300 mg IV over 1 h, then 10–50 mg/h over 24 h | 100–200 mg daily (generally IV form used for rate control) | IV: 9–36 d Oral: 26–107 d | Loading dose 6–10 g administered over 2–4 wk; can combine IV and oral dosing to complete |
7.2.1. Acute Rate Control
Figure 17. Acute Rate Control in AF With Rapid Ventricular Response (RVR)

7.2.2. Long-Term Rate Control
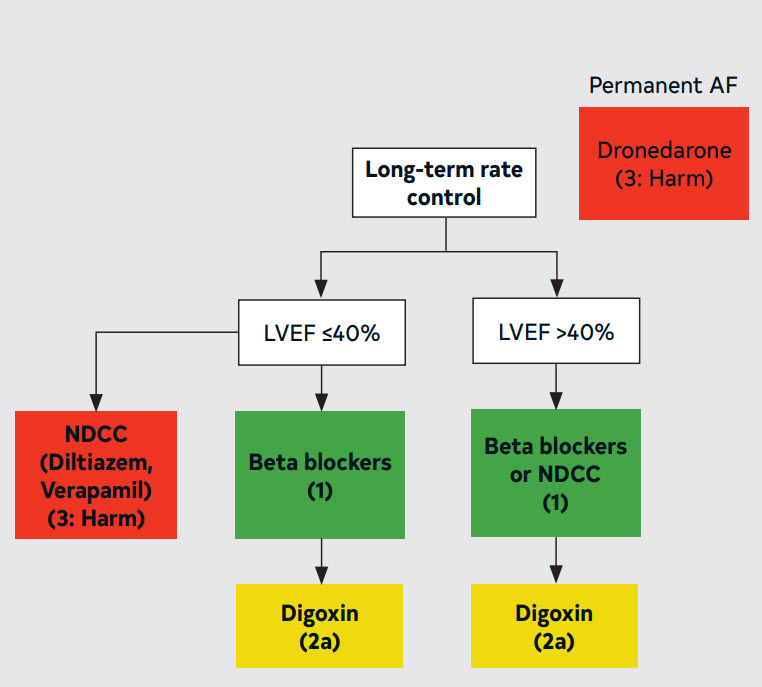
Figure 18. AF Long-Term Rate Control
7.3. Atrioventricular Nodal Ablation (AVNA)
8. Rhythm Control
8.1. Goals of Therapy With Rhythm Control
Figure 19. Patient and Clinical Considerations for Choosing Between Rhythm Control and Rate Control

Figure 20. Flowchart for Treatment Choices When Required to Decrease AF Burden

Flowchart outlining overall strategy and treatment options for patient with AF in whom rhythm-control therapy is required.
8.2. Electrical and Pharmacological Cardioversion
8.2.1. Prevention of Thromboembolism in the Setting of Cardioversion
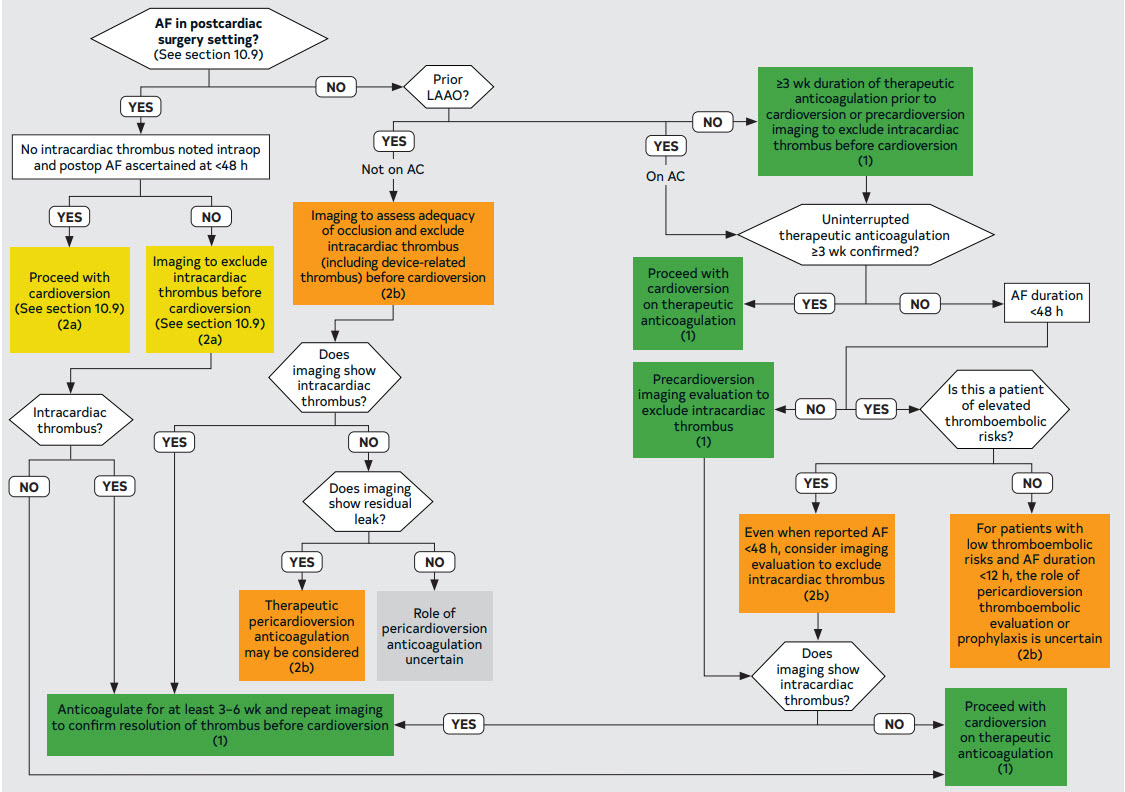
Figure 21. Patients With Hemodynamically Stable AF Planned for Cardioversion
8.2.2. Electrical Cardioversion
8.2.3. Pharmacological Cardioversion
Table 22. Drugs for Pharmacological Conversion of Atrial Fibrillation to Sinus Rhythm
| Drug | Route of Administration | Loading Dose | Maintenance Dose | Approximate Time to Conversion to Sinus Rhythm | Primary Route(s) of Elimination | Elimination Half-Life | Major Adverse Effects |
|---|---|---|---|---|---|---|---|
| Amiodarone | IV | 5–7 mg/kg or 300 mg* | 1200–3000 mg via continuous infusion over 24 h | 8–12 h | Liver metabolism Biliary excretion | 9–36 d | Bradycardia Hypotension QT prolongation Phlebitis Torsades de pointes (TdP) |
| Flecainide | Oral† | N/A | 200 mg if <70 kg, 300 mg if >70 kg, single dose | 3–8 h | Liver (70%) Kidney (30%)† | 12–27 h | Atrial flutter AV block Dizziness Dyspnea Exacerbation of HFrEF Headache Nausea QT prolongation Ventricular tachycardia (VT) Visual disturbances |
| Ibutilide | IV | ≥60 kg: 1 mg over 10 min <60 kg: 0.01 mg/kg over 10 min If arrhythmia does not terminate within 10 min after the end of the first infusion, may administer a second dose, equal to the first dose | N/A | 30–90 min | Liver | 2–12 h | Nonsustained VT QT prolongation TdP |
| Procainamide | IV | 1 g over 30 min | 2 mg/min continuous infusion over 1 hour | 30–60 min | Liver (16–33%) Kidney (50–65%)‡ | 3–4 h (parent) 7 h (NAPA) | Agranulocytosis AV block Exacerbation of HFrEF Hypotension Neutropenia QT prolongation Rash Thrombocytopenia TdP |
| Propafenone | Oral | N/A | 450 mg if <70 kg, 600 mg if >70 kg, single dose | 3–8 h | Liver | 9 h | Atrial flutter AV block Dizziness Dyspnea Exacerbation of HFrEF Nausea Taste disturbances VT Visual disturbances |
† Flecainide is available in an intravenous dosage form in Europe.
‡ % of a dose excreted unchanged in urine.
Figure 22. Treatment Algorithm for Pharmacological Conversion of AF to Sinus Rhythm

† Amiodarone requires several hours for efficacy; ibutilide is generally effective in 30–90 minutes, but carries a higher risk of QT interval prolongation and torsades de pointes.
‡ Recommend avoidance of intravenous procainamide for patients initially treated with amiodarone or ibutilide, to avoid excessive QT interval prolongation and torsades de pointes; rather, procainamide may be considered for patients for whom amiodarone and ibutilide are not considered optimal as first-line drugs.
§ First dose should be administered in a facility that can provide continuous ECG monitoring and cardiac resuscitation, due to the potential for proarrhythmia or post-conversion bradycardia.
8.3. AADs for Maintenance of Sinus Rhythm
8.3.1. Specific Drug Therapy for Long-Term Maintenance of Sinus Rhythm
Figure 23. Treatment Algorithm for Drug Therapy for Maintenance of Sinus Rhythm

Table 23. Specific Drug Therapy for Maintenance of Sinus Rhythm in Patients With Atrial Fibrillation
| Drug | Loading Dose | Maintenance Dose | Primary Route(s) of Elimination | Elimination Half-Life | Mechanism of Action | Major Adverse Effects | Important Pharmacokinetic Drug Interactions |
|---|---|---|---|---|---|---|---|
| Amiodarone | Total loading dose 6–10 g, given 400–800 mg daily in 2–4 divided doses for 1–4 wk | 200 mg once daily | Liver metabolism Biliary excretion | 14–59 d | Inhibits IKr, IKs, INa, IKur, Ito, ICa-L, IKAch Noncompetitive betablocker | AV block Bradycardia Corneal microdeposits Elevation in transaminases Hepatotoxicity Hyperthyroidism Hypothyroidism Nausea QT prolongation Peripheral neuropathy Photosensitivity Pulmonary fibrosis Skin pigmentation (blue-grey) TdP | Moderate* inhibitor of CYP2C9, weak† inhibitor of CYP2D6 Some inhibition of CYP3A Increases plasma concentrations of warfarin, lovastatin,‡ simvastatin,§ cyclosporine Inhibits p-gp Increases plasma concentrations of digoxin |
| Dofetilide | N/A | CrCl > 60 mL/min: 500 µg twice daily CrCl 40–60 mL/min: 250 µg twice daily CrCl 20–40 mL/min: 125 µg twice daily CrCl < 20 mL/min: Contraindicated | Kidney | 10 h | Inhibits IKr and augments late INa | QT prolongation TdP | Dofetilide is renally excreted via the renal cation transport system. The following drugs inhibit renal cation transport, increase plasma dofetilide concentrations, and are contraindicated in patients taking dofetilide: Cimetidine Dolutegravir Ketoconazole Megestrol Prochlorperazine Trimethoprim (alone or in combination with sulfamethoxazole) Verapamil In addition, hydrochlorothiazide (alone or in combination with triamterene) increases plasma dofetilide concentrations and should not be coadministered with dofetilide |
| Dronedarone | N/A | 400 mg twice daily | Liver metabolism | 13–19 h | Inhibits IKr, IKs, INa, IKur, Ito, ICa-L, IKAch Noncompetitive betablocker | Abdominal pain Asthenia Bradycardia Diarrhea Nausea and vomiting QT prolongation Rash TdP | Dronedarone is a substrate for CYP3A and is a moderate inhibitor of CYP3A and CYP2D6 Dronedarone is also a substrate for, and inhibitor of, p-gp Dronedarone may increase plasma concentrations of: Dabigatran Digoxin Simvastatin|| Sirolimus Tacrolimus Warfarin The following drugs may increase plasma dronedarone concentrations: Grapefruit juice The following drugs may decrease plasma dronedarone concentrations: CYP 3A inducers including St. John’s wort, rifampin, and phenytoin |
| Flecainide | N/A | 50–300 mg/d PO divided q 8–12 h | Liver (70%) Kidney (30%)¶ | 12–27 h | Inhibits INa | Atrial flutter AV block Dizziness Dyspnea Exacerbation of HFrEF Headache Nausea QT prolongation VT Visual disturbances | Flecainide is a substrate for CYP2D6 The following drugs may increase plasma flecainide concentrations: Amiodarone Duloxetine Fluoxetine Paroxetine |
| Propafenone | N/A | 150–300 mg/ PO q 8 h, ER 225–425 PO q 12 h | Liver | 9 h | Inhibits INa | Atrial flutter Bradycardia AV block Dizziness Dyspnea Exacerbation of HFrEF Nausea Taste disturbances VT Visual disturbances | Propafenone is a substrate for CYP 2D6 The following drugs may increase plasma propafenone concentrations: Fluoxetine Paroxetine Propafenone may increase plasma digoxin concentrations Propafenone may increase plasma warfarin concentrations |
| Sotalol | CrCl >60 mL/min: 40–80 mg twice daily for 3 d CrCl: 40–60 mL/min: 80 mg once daily for 3 d CrCl <40 mL/min: Contraindicated | CrCl >60 mL/min: 80–160 mg twice daily CrCl: 40-60 mL/min: 80–160 mg once daily CrCl <40 mL/min: Contraindicated | Kidney | 12 h | Inhibits IKr Betablocker d-Sotalol augments late INa | AV block Bradycardia Bronchospasm Diarrhea Exacerbation of HFrEF Fatigue Nausea and vomiting QT prolongation TdP | None |
† Mild inhibitor: causes a ≥1.25-fold but <2-fold increase in AUC or a 20% to 50% decrease in clearance.
‡ Lovastatin doses should not exceed 40 mg daily in patients taking amiodarone.
§ Simvastatin doses should not exceed 20 mg daily in patients taking amiodarone.
|| Simvastatin doses should not exceed 10 mg daily in patients taking dronedarone.
¶ % of a dose excreted unchanged in urine.
8.3.2. Inpatient Initiation of Antiarrhythmic Agents
Table 24. Recommended Monitoring for Patients Taking Oral Amiodarone
| Adverse Effect | Baseline Testing | Initial Follow-Up Testing | Additional Follow-Up Testing |
|---|---|---|---|
| Hypo- or hyperthyroidism | TSH (T4 and T3 if TSH abnormal) | 3–6 mo | Every 6 mo |
| Hepatotoxicity | AST, ALT | 3–6 mo | Every 6 mo |
| QT interval prolongation | ECG | Annually | — |
| Interstitial lung disease | Chest x-ray: Recommended CT chest: not recommended | Chest x-ray: Unexplained cough or dyspnea or other signs/symptoms suspicious for interstitial lung disease | CT chest: As indicated to follow-up ongoing symptoms or chest x-ray findings |
| Corneal microdeposits (epithelial keratopathy) | Not recommended | Development of visual abnormalities which may indicate optic neuropathy | — |
| Dermatologic (blue-grey skin discoloration), photosensitivity | Not recommended | Physical examination annually | Development of skin discoloration, severe sunburn |
| Neurological | Not recommended | Physical examination annually | Development of peripheral neuropathy or other neurological abnormalities |
Table 25. Recommended Monitoring for Patients Taking Other Antiarrhythmic Drugs
| Drug | Baseline Testing | Follow-Up Testing | Additional Follow-Up Testing |
|---|---|---|---|
| Dofetilide | 12-lead ECG Continuous ECG monitoring during 3-d hospitalization for dofetilide initiation Serum potassium and magnesium concentration Serum creatinine for estimation of CrCl | In 3–6 mo: 12-lead ECG Serum potassium and magnesium concentration Serum creatinine for estimation of CrCl | Every 3–6 mo (more frequently for patients concomitantly taking other QT interval-prolonging drugs or with changing kidney function): 12-lead ECG Serum potassium and magnesium concentration Serum creatinine for estimation of CrCl |
| Dronedarone | 12-lead ECG AST† ALT† | Within first 6 mo: AST† ALT† | — |
| Ibutilide | 12-lead ECG Determination of serum potassium and magnesium concentrations and correction of hypokalemia and/or hypomagnesemia is recommended prior to initiation of the infusion | Continuous ECG monitoring for assessment of QTc interval duration is recommended for at least 4 h following infusion or until the QTc has returned to baseline to minimize the risk of ibutilide-associated TdP | — |
| Procainamide | 12-lead ECG Blood pressure | ECG monitoring for assessment of rhythm, QRS width and QTc interval is recommended during the infusion to minimize the risk of procainamide-associated ventricular proarrhythmia, including TdP Blood pressure monitoring is recommended during the infusion to detect clinically relevant hypotension | — |
| Sotalol | 12-lead ECG Continuous ECG monitoring during 3-d hospitalization for sotalol initiation Serum potassium and magnesium concentration Serum creatinine for estimation of CrCl | In 3-6 mo: 12-lead ECG Serum potassium and magnesium concentration Serum creatinine for estimation of CrCl | Every 3–6 mo (more frequently for patients concomitantly taking other QT interval-prolonging drugs or with changing kidney function): 12-lead ECG Serum potassium and magnesium concentration Serum creatinine for estimation of CrCl |
† To facilitate early detection of potential dronedarone-associated hepatotoxicity.
8.3.4. Upstream Therapy
8.4. AF Catheter Ablation
8.4.1. Patient Selection
8.4.2. Techniques and Technologies for AF Catheter Ablation
8.4.3. Management of Recurrent AF After Catheter Ablation
8.4.4. Anticoagulation Therapy Before and After Catheter Ablation
8.4.5. Complications Following AF Catheter Ablation
Table 26 . Complications After Atrial Fibrillation Catheter Ablation
| Complication | Frequency of Complication | Timing of Complication | Signs and Symptoms | Diagnosis | Treatment |
|---|---|---|---|---|---|
| LA-esophageal fistula | 0.2% | 1–4 wk | Chest pain, pain with swallowing, fever, stroke symptoms | CT scan of chest | Surgery |
| Cardiac perforation w/ tamponade | 0.4%–1.5% | During procedure | Hypotension | Echocardiography | Pericardiocentesis |
| CVA/TIA | 0.1%–1.0% | During procedure and up to 1 wk | Neurological findings | MRI or CT scan | Anticoagulate when safe |
| Pulmonary vein (PV) stenosis | 0.1%–0.8% | Months | Dyspnea, hemoptysis | MRI or CT Scan | Stent |
| Phrenic nerve paralysis | 0.2%–0.4% | During procedure | Dyspnea | Fluoroscopy | Time |
| Vascular access complications | 1%–7% | During procedure and up to 1 mon | Pain, swelling at access site | Ultrasound or CT scan | Observation |
| Vascular access complications requiring surgery | 0.1%–0.3% | During procedure and up to 1 mon | Pain and swelling at access site | Ultrasound or CT scan | Surgery |
| Death | 0.1%–0.4% | During procedure | |||
| Pneumonia | 0.4%–1.0% | Days | Cough, fever | Chest x-ray | Antibiotics |
8.5. Role of Pacemakers and Implantable Cardioverter-Defibrillators for the Prevention and Treatment of AF
8.6. Surgical Ablation
9. Management of Patients With HF
9.1. General Considerations for AF and HF
9.2. Management of AF in Patients With HF*
† Consider the risk of cardioversion and stroke when using amiodarone as a rate-control agent.
10. AF and Specific Patient Groups
Figure 24. Management of Patients with HF and AF
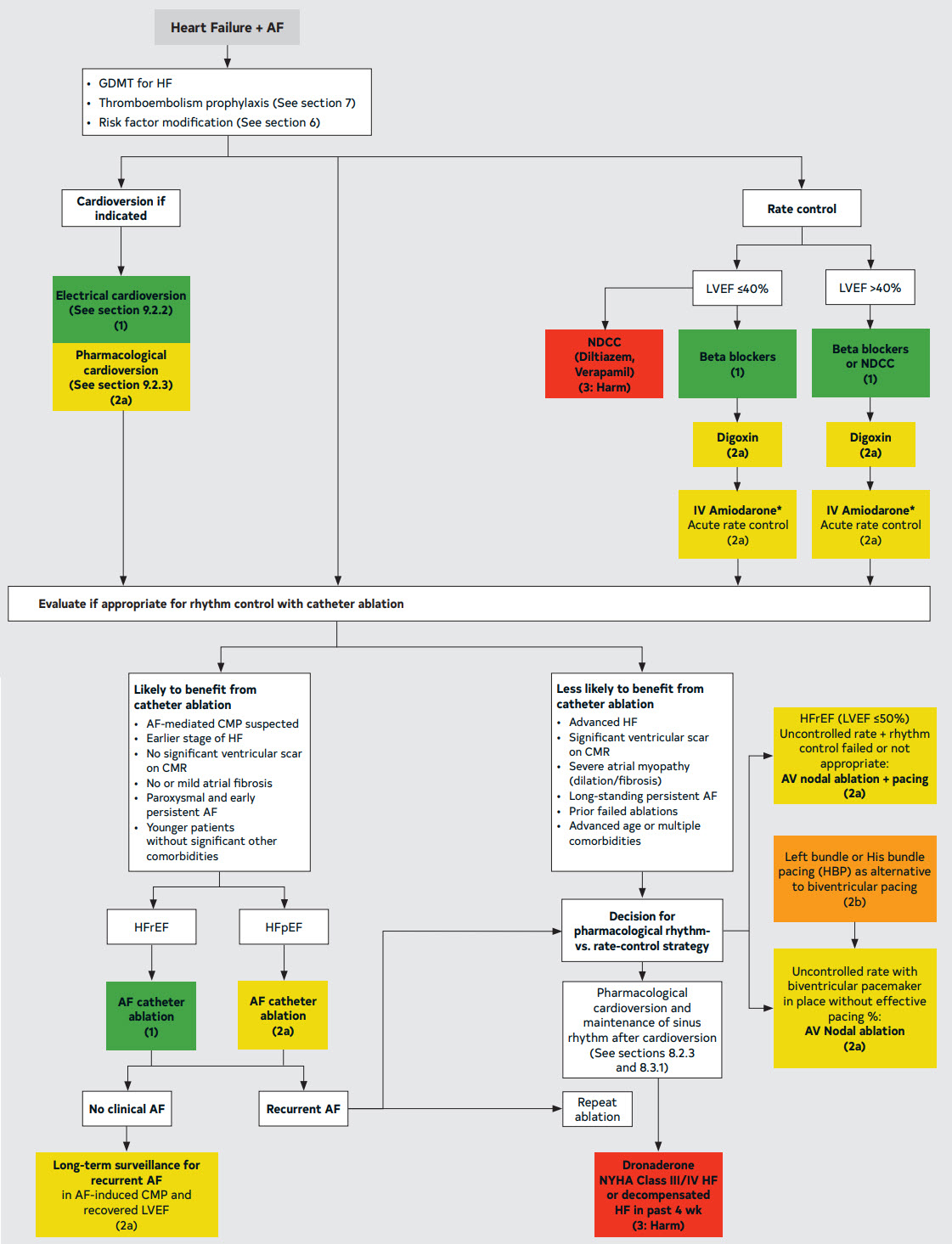
10.1. Management of Early Onset AF, Including Genetic Testing
10.2. Athletes
10.3. Management Considerations in Patients With AF and Obesity
10.4. Anticoagulation Considerations in Patients With Class III Obesity
10.5. AF and VHD
10.6. Wolff-Parkinson-White (WPW) and Pre-Excitation Syndromes
10.7. HCM
10.8. Adult Congenital Heart Disease (ACHD)
10.9. Prevention and Treatment After Cardiac Surgery
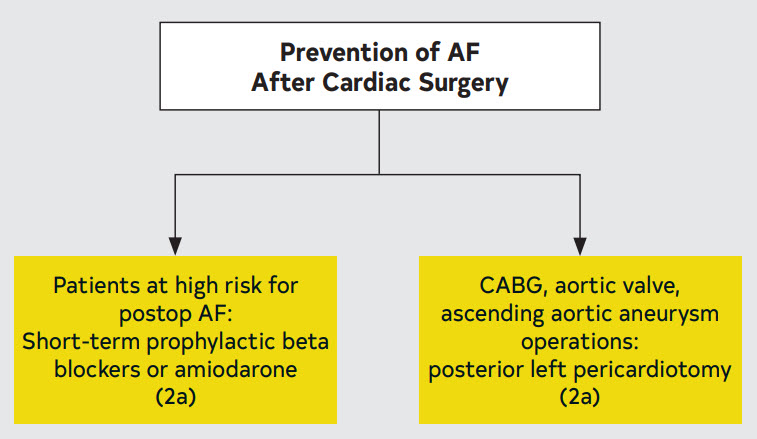
10.9.1. Prevention of AF After Cardiac Surgery
Figure 25. Prevention of AF After Cardiac Surgery
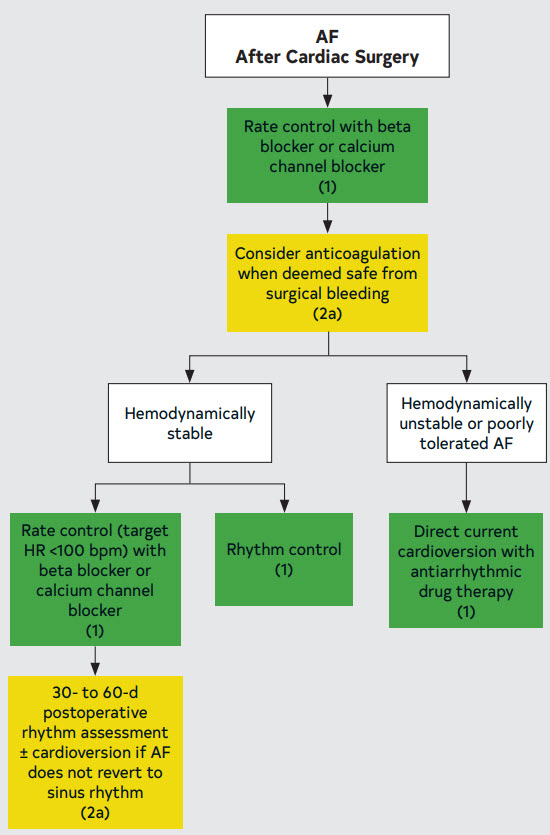
10.9.2. Treatment of AF After Cardiac Surgery
Figure 26. Treatment of AF After Cardiac Surgery
10.10. Acute Medical Illness or Surgery (Including AF in Critical Care)
Figure 27. Unadjusted Cumulative Risk of AF Recurrence

(A) Overall risk of recurrent AF among individuals with and without acute precipitants. (B) Overall risk of recurrent AF among individuals with infection, cardiac surgery, and noncardiothoracic surgery, as compared with no precipitant. These 3 precipitants were selected for display because the risk of recurrent AF was significantly lower as compared with the referent group without precipitants in multivariable adjusted models. Individuals with other AF precipitants were excluded from this plot for clarity. CT, cardiothoracic.
Reproduced with permission from Wang EY, et al. Copyright 2020, American Heart Association, Inc
Figure 28. Acute Medical or Surgical Illness

10.11. Hyperthyroidism
10.12. Pulmonary Disease
10.13. Pregnancy
Table 28. Anticoagulation Strategies During Pregnancy - Antenatal Options
Table 29. Medical Cancer Therapy Associated With Increased Risk of AF (>1%)
| Cancer Therapy | Frequency Reported in Clinical Trials and Observational Studies Common: Incidence 1%–10% | Frequency Reported in Clinical Trials and Observational Studies Frequent: >10% | Comments |
|---|---|---|---|
| Anthracyclines Doxorubicin, epirubicin, idarubicin, mitoxantrone | × | AF may be a secondary result of anthracycline cardiotoxicity; studies in different populations demonstrate variable risk of AF | |
| Antimetabolites | × | × | |
| Clofarabine combined with cytarabine | × | ||
| 5 fluorouracil (5FU) | × | ||
| Cepecitabine | |||
| Gemcitabine | |||
| Alkylating agents Cyclophosphamide Melphalan + stem cell transplantation | × | ×* | * Stem cell transplantation is associated with an increased risk of AF, and the risk may be higher with melphalanassociated regimens |
| Immunomodulatory drugs | × | Given rates reported from patients with multiple myeloma, AF due to underlying cardiac AL amyloid may contribute | |
| Lenalidomide | × | ||
| Interleukin-2 | |||
| Tyrosine kinase inhibitors (TKIs) | ׆ | ׆ | † Reported AF rates with ibrutinib have varied across trials (4%–18%), partly related to varying duration of follow-up and patient factors. Second-generation BTKis have more selective Bruton’s tyrosine kinase (BTK) activity and are associated with a lower incidence of AF than ibrutinib Based on US FDA adverse event reporting system |
| Ibrutinib (Bruton’s kinase inhibitor [BTKi]) | × | ||
| Acalbrutinib (2ndgeneration BTKi) | × | ||
| Zanubrutinib (2ndgeneration BTKi) | × | ||
| Ponatinib (BCRABL TKI) and other TKIs (eg, trametinib, osimertinib, nilotinib, ribociclib) | × | ||
| VEGF inhibitor Sorafenib in combination with 5FU | |||
| BRAF inhibitor Vemurafenib | |||
| Chimeric antigen receptor (CAR) T-cell therapy Tisagenlecleucel Axicabtagene ciloleucel | × | ||
| Monoclonal antibodies Rituximab | × |
10.14. Cardio-Oncology and Anticoagulation Considerations
Table 30. Special Considerations for Anticoagulation in Patients With AF on Active Cancer Treatment
| Increased bleeding risk | High bleeding risk estimators (eg, HAS-BLED) Thrombocytopenia (platelet <50,000/uL) Intracranial malignancy Gastrointestinal malignancy History of major bleeding Severe kidney dysfunction (eGFR <30 mL/min/1.73m2) |
|---|---|
| Drug interactions | P-glycoprotein inducers or inhibitors CYP 3A4 inducers or inhibitors |
10.15. CKD and Kidney Failure
10.16. Anticoagulation Use in Patients With Liver Disease
The score is based on the 5 variables: encephalopathy (none=1 point, grade 1 and 2=2 points, grade 3 and 4= 3 points); ascites (none=1 point, slight=2 points, moderate=3 points); total bilirubin (<2 mg/mL=1 point, 2–3 mg/mL=2 points, >3 mg/mL=3 points); albumin (>3.5 mg/mL=1 point, 2.8–3.5 mg/mL=2 points, <2.8 mg/mL=3 points); INR (<1.7=1 point, INR 1.7–2.2=2 points, INR >2.2=3 points).
Class of Recommendations and Level of Evidence
| Class (Strength) of Recommendation |
|---|
| CLASS 1 (STRONG) Benefit >>> Risk |
Suggested phrases for writing recommendations:
|
| CLASS 2a (MODERATE) Benefit >> Risk |
Suggested phrases for writing recommendations:
|
| CLASS 2b (WEAK) Benefit ≥ Risk |
Suggested phrases for writing recommendations:
|
| CLASS 3: No Benefit (MODERATE) (Generally, LOE A or B use only) Benefit = Risk |
Suggested phrases for writing recommendations:
|
| CLASS 3: Harm (STRONG) Risk > Benefit |
Suggested phrases for writing recommendations:
|
| Level (Quality) of Evidence‡ |
|---|
| LEVEL A |
|
| LEVEL B-R (Randomized) |
|
| LEVEL B-NR (Nonrandomized) |
|
| LEVEL C-LD (Limited Data) |
|
| LEVEL C-EO (Expert Opinion) |
|
COR and LOE are determined independently (any COR may be paired with any LOE).
A recommendation with LOE C does not imply that the recommendation is weak. Many important clinical questions addressed in guidelines do not lend themselves to clinical trials. Although RCTs are unavailable, there may be a very clear clinical consensus that a particular test or therapy is useful or effective.
* The outcome or result of the intervention should be specified (an improved clinical outcome or increased diagnostic accuracy or incremental prognostic information).
† For comparative-effectiveness recommendations (COR I and IIa; LOE A and B only), studies that support the use of comparator verbs should involve direct comparisons of the treatments or strategies being evaluated.
‡ The method of assessing quality is evolving, including the application of standardized, widely used, and preferably validated evidence grading tools; and for systematic reviews, the incorporation of an Evidence Review Committee.
COR indicates Class of Recommendation; EO, expert opinion; LD, limited data; LOE, Level of Evidence; NR, nonrandomized; R, randomized; RCT, randomized controlled trial.
Abbreviations
- ACE
- angiotensin-converting enzyme
- ACS
- acute coronary syndrome
- AF
- atrial fibrillation
- ARB
- angiotensin receptor blocker
- AV
- atrioventricular
- BID
- two times a day
- bpm
- beats per minute
- CAD
- coronary artery disease
- CKD
- chronic kidney disease
- COPD
- chronic obstructive pulmonary disease
- COR
- Class of Recommendation
- CPR
- cardiopulmonary resuscitation
- CrCl
- creatinine clearance
- CT
- computed tomography
- ECG
- electrocardiogram
- EF
- ejection fraction
- ER
- extended release
- GI
- gastrointestinal
- HCM
- hypertrophic cardiomyopathy
- HCTZ
- hydrochlorthiazide
- HF
- heart failure
- HFpEF
- heart failure with preserved ejection fraction
- INR
- international normalized ratio
- IV
- intravenous
- LA
- left atrium/atrial
- LAA
- left atrial appendage
- LMWH
- low molecular weight heparin
- LOE
- Level of Evidence
- LV
- left ventricular
- LVEF
- left ventricular ejection fraction
- MRI
- magnetic resonance imaging
- N/A
- not applicable
- NSAIDs
- Non-Steroidal Anti-Inflammatory Drugs
- PAD
- peripheral artery disease
- QD
- once daily
- QID
- four times a day
- RA
- right atrium/atrial
- RAAS
- renin-angiotensin-aldosterone system
- RV
- right ventricular
- RVR
- rapid ventricular response
- TE
- thromboembolic events
- TEE
- transesophageal echocardiography
- TTE
- transthoracic echocardiogram
- UFH
- unfractionated heparin
- VHD
- valvular heart disease
- WPW
- Wolff-Parkinson-White
Source Citation
Joglar JA, Chung MK, Armbruster AL, Benjamin EJ, Chyou JY, Cronin EM, Deswal A, Eckhardt L, Goldberger ZD, Gopinathannair R, Gorenek B, Hess PL, Hlatky M, Hogan G, Ibeh C, Indik JH, Kido K, Kusumoto F, Link MS, Linta KT, Marcus GM, McCarthy PM, Patel N, Patton KK, Perez MV, Piccini JP, Russo AM, Sanders P, Streur MM, Thomas KL, Times SS, Tisdale JE, Valente AM, Van Wagoner DR. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. [published online ahead of print Nov 30, 2023].
J Am Coll Cardiol. doi: 10.1016/j.jacc.2023.08.017
Copublished in Circulation. doi: 10.1161/CIR.0000000000001193
Disclaimer
This resource is for informational purposes only, intended as a quick-reference tool based on the cited source guideline(s), and should not be used as a substitute for the independent professional judgment of healthcare providers. Practice guidelines are unable to account for every individual variation among patients or take the place of clinician judgment, and the ultimate decision concerning the propriety of any course of conduct must be made by healthcare providers after consideration of each individual patient situation. Guideline Central does not endorse any specific guideline(s) or guideline recommendations and has not independently verified the accuracy hereof. Any use of this resource or any other Guideline Central resources is strictly voluntary.
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.